Global Medical Device Analytical Testing Outsourcing Market Analysis By Services (Extractable and leachable, Material characterization, Physical testing, Bioburden testing, Sterility testing), and By Region (U.S., Canada, Mexico, Rest of North America, UK, France, Germany, Italy, Spain, Benelux Union, Nordic Countries , Rest of Europe, China, Japan, India, South Korea, Singapore, Philippines, Indonesia, Thailand, Malaysia, Vietnam, Rest of Southeast Asia, Brazil, Argentina Rest of Latin America, Saudi Arabia, UAE, Egypt, Kuwait, Southern Africa, Rest of Middle East & Africa) – Global Insights, Growth, Size, Comparative Analysis, Trends and Forecast, 2019 - 2027
Industry Trends
Global Medical Device Analytical Testing Outsourcing Market was valued at US$ 3189.32 million in 2018 and is expected to reach US$ 7458.94 million by 2027, growing at an estimated CAGR of 9.8% over the forecast period.
Medical Device Analytical Testing Outsourcing Market is witnessing a notable growth. Implantable Medical Devices (IMDs) are progressively being utilized to improve wellbeing results for patients. IMD planners need to adjust multifaceted nature, unwavering quality, power utilization, and expenses. Subsequently, organizations move their concentration to development instead of non-center exercises and in this way re-appropriate these exercises to dispatch compelling gadgets. Open heart medical procedure is perilous and intrusive. Achievements in clinical hardware are empowering heart experts to find new surgeries. For instance, mitral and tricuspid valve substitution offers access to the heart by a catheter in the skin. It can diminish recuperation time and re-affirmations and spare lives. A few associations are now battling to feature their creative heart medical procedure items to the market.
Unlawful utilization of medications can influence lives, wreck networks, and put a strain on open administrations. In the U.S., the FDA is trying to handle substance maltreatment through the innovative utilization of clinical gadget innovation. VR treatment and the utilization of transcranial attractive incitement gadgets are a portion of the affirmed plans for handling chronic drug use. It won't take long for clinical gadgets to be incorporated as a standard choice for the treatment of psychological wellness sicknesses, for example, sorrow. The National Healthcare System (NHS) in the United Kingdom has manufactured a cerebrum incitement gadget that can be accessible from the drug store utilizing remedy. Individuals experiencing psychological well-being sicknesses much of the time have diminished movement in the left frontal flap, which controls dynamic, character, and passionate articulation. The Flow headset is a wearable gadget that can be connected to the head with the assistance of anodes and gives electrical incitement to that piece of the cerebrum. Meetings typically last about 30 minutes. Scientists are likewise trying a little gadget that can be embedded in the mind, that directs cerebrum capacities and beats of electrical incitement when a patient beginnings feeling low.
Anti-toxin opposition is a difficult issue that makes amazing anti-infection agents pointless to fend off organisms. There is no single explanation behind the issue, yet one of the conceivable outcomes can be the admission of anti-infection agents at an exorbitant rate. Specialists normally take that course if they need patients to begin on treatment while they hang tight for lab results to turn out. Regardless, masters at Penn State University fabricated a clinical gadget that identifies the nearness of microscopic organisms in a brief timeframe and as needs be characterizes it. This has caused it feasible for specialists to just to recommend the anti-microbial that is required, along these lines helping cut down on anti-infection obstruction. All these factors has positively impacted the Medical Device Analytical Testing Outsourcing Market.
Wearable devices are very noticeable at this moment. Researches suggest that wearables will increase by 26% all-inclusive, with shipments arriving at 225 million out of 2019. As wearables become typical, their clinical applications will likewise turn out to be increasingly overwhelming. Examiners have defined a "microgel" covering for implantable clinical gadgets that releases focused on measures of anti-infection agents if microscopic organisms approach the inserts. Each micro gel has a network-like structure of bits that are multiple times smaller than the width of a strand of human hair. Experts accept this leap forward could essentially decrease the quantity of post-careful contaminations experienced by patients. Expecting this is the situation, this would assist them with chopping down various intricacies, and wouldn't expect them to get anti-microbial medicines to battle existing diseases.
Gadgets associated with the IoT are giving clinical gadget producers an expansive cluster of new informational indexes with imminent applications for their organizations. Associations can utilize these estimations to include various highlights and improve execution based on client communications recorded by the gadget, rather than depending on old strategies, for example, overviews. This can incite a predominant shopper involvement with an undeniably quiet determined industry. The FDA is blasting the way by building a national framework for gathering ongoing information and clinical evidence on everything from contraptions utilized in spinal fixes to tendon conclusion methods. The revelations may well characterize the association among patients and clinical gadgets all through the coming years.
Apple was permitted two De Novos to tackle its ECG checking capacities on the Apple Watch. Bose, then again, is additionally chipping away at advances to improve portable amplifiers. In the not so distant future, non-clinical gadget organizations chipping away at clinical innovations will turn out to be increasingly ordinary, with Apple and Bose working together with various associations that recognize chances to use their innovation in the clinical gadget industry.
Specialists intently watch the clinical gadget and gear market to study foreseen changes. As per reports, incomes for clinical gadgets this year will outflank 2018's numbers. As of late, the FDA divulged a snappier pathway for the endorsement of clinical gadgets, which is a major driver of development in the clinical gadget and hardware advertise.
Services Insights
Material characterization dominated the Medical Device Analytical Testing Outsourcing Market in 2015 and is likely to grow at a lucrative rate during the forecast period. Different tests that are comprehensive of cleaning, reprocessing, technique advancement and approval, quickened steadiness testing, biocompatibility, approval testing for bundling materials (holder conclusion saturation), and transport solidness are additionally foreseen to observe remunerating development.
Leachable additionally may emerge from auxiliary bundling parts, for example, foil pockets for low-thickness polyethylene (LDPE) ampoules. LDPE is a semi-penetrable polymer, so it permits mixes in the pocket material to go through the LDPE ampoule and relocate into the medication item definition put away in the ampoule. Ink print or names as a component of essential or optional bundling are additionally normal wellsprings of leachable. Medication item conveyance frameworks, for example, IV sacks, organization sets, sedate conveyance siphons and mouthpieces, and so on., have brief span presentation to the medication item definitions or the patients, yet they are as yet wellsprings of conceivable leachable.
Regional Insights
North America is likely to witness lucrative growth in Medical Device Analytical Testing Outsourcing Market which is attributed to the fact that it is one of the top manufacturing hubs of highly reliable, complex, and high-end medical devices. The regulatory authorities also perform routine post-market surveillance by levying a fee from the manufacturer. Receipt of any complaints about the drawbacks of the product entails its withdrawal from the market, thereby proving the stringent nature of these procedures.
BHP Europe Laboratory Services is a free research facility inside Bemis Healthcare Packaging Europe. They offer an exhaustive scope of testing administrations including Aging, Material Testing, Transportation, Package and Analytical testing, fundamental for item approval. Notwithstanding giving excellent testing and investigation, the group of specialists has significant involvement with prompting, critical thinking, and counseling on all parts of bundling guidelines and norms. Numerous makers are running behind with their MDR usage. Contingent upon what number of items they have to test and who is completing trying, the procedure can take months or even years. Numerous little and medium-size organizations are not prepared because of restricted assets, absence of administrative information, or just not realizing where to begin. Organizations with a bigger volume of items might be more in danger, particularly on the off chance that they haven't started organizing items. In any case, those with fewer items and less ground to make up should as of now be progressing in the direction of MDR consistency. If they're not, it's an ideal opportunity to put the pedal to the metal. Rise of customized medication, sedate gadget mix, man-made brainpower, wearables and expanded spotlight on constant patient observing brought about complex clinical gadgets biological system. Ongoing patient observation, remote patient checking, and ceaseless patient checking are the central regions of intrigue with regards to interminable illness the executives, for example, diabetes and cardiovascular ailments.
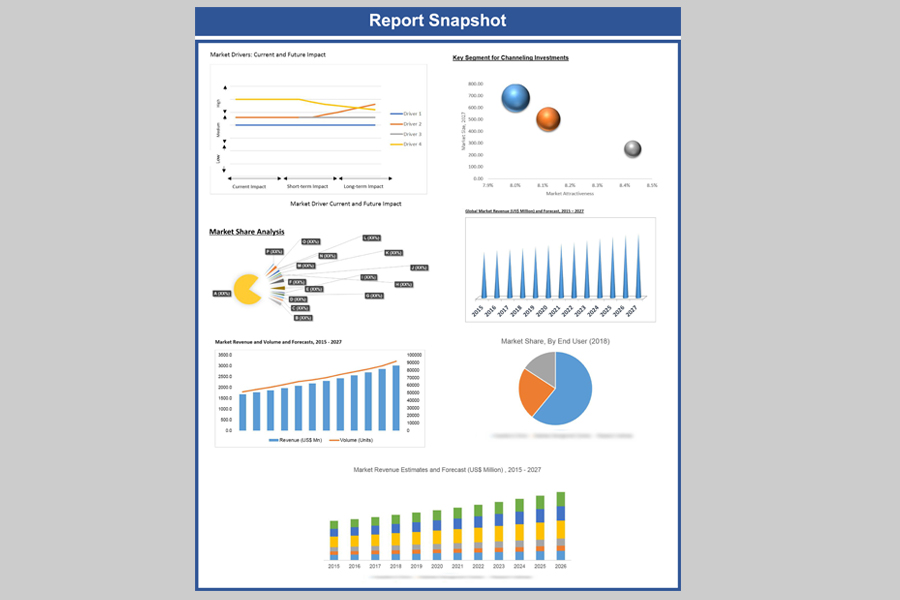
Global Medical Device Analytical Testing Outsourcing in Healthcare Market Revenue & Forecast, (US$ Million), 2015 – 2027
Competitive Landscape
In this complex ecosystem, equipment must deliver accurate results, have durability and be easy to operate. In order to match those parameters, devices must now undergo shear testing under accelerated conditions to obtain marketing approval and have a competitive advantage, thereby, fuelling the Medical Device Analytical Testing Outsourcing Market. Having these tests in-house is a matter of time, labour and cost; outsourcing these tests helps to overcome these constraints.
SGS SA; Toxikon, Inc.; Eurofins Scientific; Pace Analytical Services, LLC; Intertek Group plc; WuXi AppTec.; NORTH AMERICAN SCIENCE ASSOCIATES INC.; Envigo; Charles River Laboratories International Inc.; and Medical Device Testing Services are the key players in Medical Device Analytical Testing Outsourcing Market.
Service portfolio expansion along with regional penetration is anticipated to positively affect the players operating in Medical Device Analytical Testing Outsourcing Market. For Instance, in December 2016, company acquired Exova’s environment testing laboratories in Eastern Canada. This initiative is expected to help the company enhance geographical footprints in North America.
Global Medical Device Analytical Testing Outsourcing Market:
- By Services
- Extractable and leachable
- Material characterization
- Physical testing
- Bioburden testing
- Sterility testing
- By Geography
- North America
- US
- Canada
- Mexico
- Rest of North America
- Europe
- France
- The UK
- Spain
- Germany
- Italy
- Nordic Countries
- Denmark
- Finland
- Iceland
- Sweden
- Norway
- Benelux Union
- Belgium
- The Netherlands
- Luxembourg
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- New Zealand
- Australia
- South Korea
- Southeast Asia
- Indonesia
- Thailand
- Malaysia
- Singapore
- Rest of Southeast Asia
- Rest of Asia Pacific
- Middle East and Africa
- Saudi Arabia
- UAE
- Egypt
- Kuwait
- South Africa
- Rest of Middle East & Africa
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- North America
Table of Contents
![]()
1.
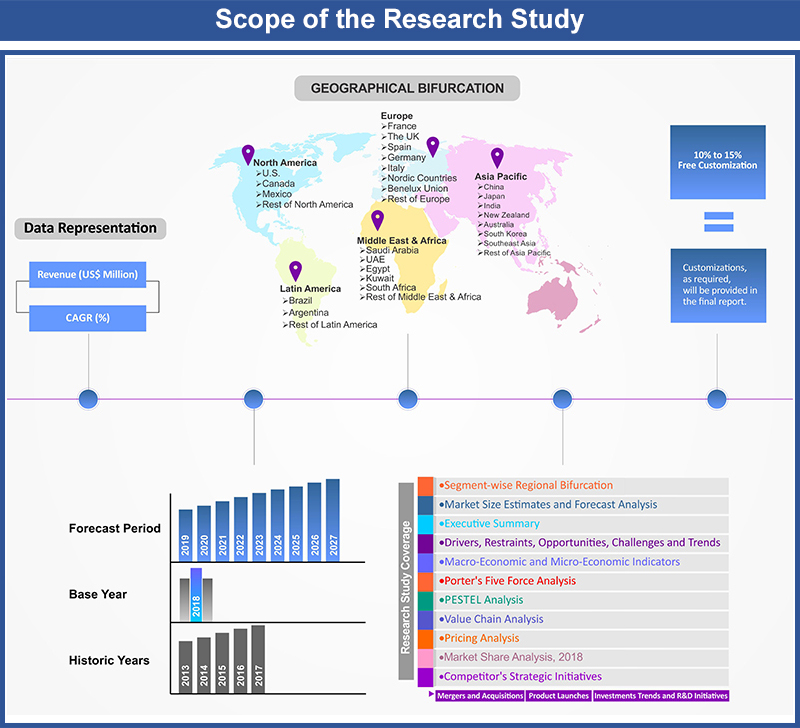
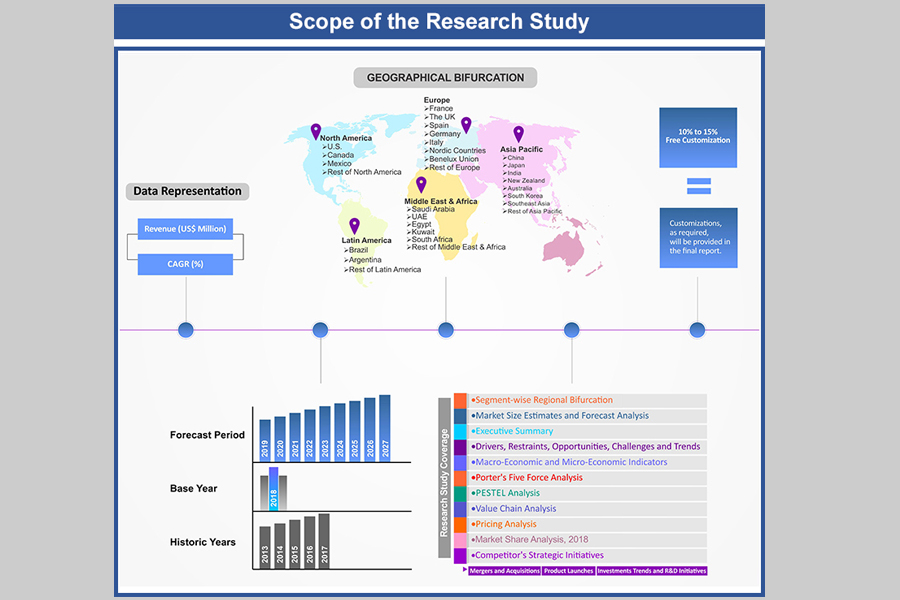
Market Scope
1.1. Market
Segmentation
1.2. Years
Considered
1.2.1. Historic
Years: 2013 - 2017
1.2.2. Base
Year: 2018
1.2.3. Forecast
Years: 2019 – 2027
2.
Key Target Audiences
3.
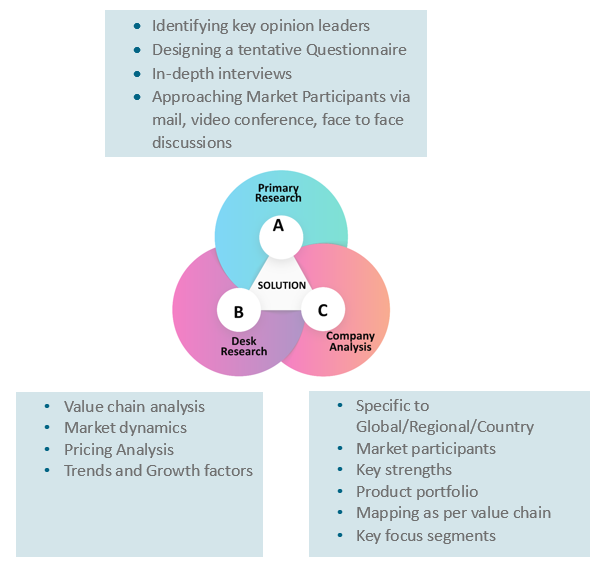
Research Methodology
3.1. Primary
Research
3.1.1. Research
Questionnaire
3.1.2. Global
Percentage Breakdown
3.1.3. Primary
Interviews: Key Opinion Leaders (KOLs)
3.2. Secondary
Research
3.2.1. Paid
Databases
3.2.2. Secondary
Sources
3.3. Market
Size Estimates
3.3.1. Top-Down
Approach
3.3.2. Bottom-Up
Approach
3.4. Data
Triangulation Methodology
3.5. Research
Assumptions
4.
Recommendations and
Insights from AMI’s Perspective**
5.
Holistic Overview of
Medical Device Analytical Testing Outsourcing Market
6.
Market Synopsis: Medical Device Analytical Testing Outsourcing
Market
7.
Medical Device Analytical
Testing Outsourcing Market Analysis: Qualitative Perspective
7.1. Introduction
7.1.1. Product
Definition
7.1.2. Industry
Development
7.2. Market
Dynamics
7.2.1. Drivers
7.2.2. Restraints
7.2.3. Opportunities
7.3. Trends in
Medical Device Analytical Testing Outsourcing Market
7.4. Market
Determinants Radar Chart
7.5. Macro-Economic
and Micro-Economic Indicators: Medical Device Analytical Testing Outsourcing
Market
7.6. Porter’s
Five Force Analysis
8.
Global Medical Device
Analytical Testing Outsourcing Market Analysis and Forecasts, 2019 – 2027
8.1. Overview
8.1.1. Global
Medical Device Analytical Testing Outsourcing Market Revenue (US$ Mn)
8.2. Global
Medical Device Analytical Testing Outsourcing Market Revenue (US$ Mn) and
Forecasts, By Services
8.2.1. Extractable
and leachable
8.2.1.1. Definition
8.2.1.2. Market Estimation and Penetration, 2013 – 2018
8.2.1.3. Market Forecast, 2019 – 2027
8.2.1.4. Compound Annual Growth Rate (CAGR)
8.2.1.5. Regional Bifurcation
8.2.1.5.1. North America
8.2.1.5.1.1. Market
Estimation, 2013 – 2018
8.2.1.5.1.2. Market
Forecast, 2019 – 2027
8.2.1.5.2. Europe
8.2.1.5.2.1. Market
Estimation, 2013 – 2018
8.2.1.5.2.2. Market
Forecast, 2019 – 2027
8.2.1.5.3. Asia Pacific
8.2.1.5.3.1. Market
Estimation, 2013 – 2018
8.2.1.5.3.2. Market
Forecast, 2019 – 2027
8.2.1.5.4. Middle East and Africa
8.2.1.5.4.1. Market
Estimation, 2013 – 2018
8.2.1.5.4.2. Market
Forecast, 2019 – 2027
8.2.1.5.5. Latin America
8.2.1.5.5.1. Market
Estimation, 2013 – 2018
8.2.1.5.5.2. Market
Forecast, 2019 – 2027
8.2.2. Material
characterization
8.2.2.1. Definition
8.2.2.2. Market Estimation and Penetration, 2013 – 2018
8.2.2.3. Market Forecast, 2019 – 2027
8.2.2.4. Compound Annual Growth Rate (CAGR)
8.2.2.5. Regional Bifurcation
8.2.2.5.1. North America
8.2.2.5.1.1. Market
Estimation, 2013 – 2018
8.2.2.5.1.2. Market
Forecast, 2019 – 2027
8.2.2.5.2. Europe
8.2.2.5.2.1. Market
Estimation, 2013 – 2018
8.2.2.5.2.2. Market
Forecast, 2019 – 2027
8.2.2.5.3. Asia Pacific
8.2.2.5.3.1. Market
Estimation, 2013 – 2018
8.2.2.5.3.2. Market
Forecast, 2019 – 2027
8.2.2.5.4. Middle East and Africa
8.2.2.5.4.1. Market
Estimation, 2013 – 2018
8.2.2.5.4.2. Market
Forecast, 2019 – 2027
8.2.2.5.5. Latin America
8.2.2.5.5.1. Market
Estimation, 2013 – 2018
8.2.2.5.5.2. Market
Forecast, 2019 – 2027
8.2.3. Physical
testing
8.2.3.1. Definition
8.2.3.2. Market Estimation and Penetration, 2013 – 2018
8.2.3.3. Market Forecast, 2019 – 2027
8.2.3.4. Compound Annual Growth Rate (CAGR)
8.2.3.5. Regional Bifurcation
8.2.3.5.1. North America
8.2.3.5.1.1. Market
Estimation, 2013 – 2018
8.2.3.5.1.2. Market
Forecast, 2019 – 2027
8.2.3.5.2. Europe
8.2.3.5.2.1. Market
Estimation, 2013 – 2018
8.2.3.5.2.2. Market
Forecast, 2019 – 2027
8.2.3.5.3. Asia Pacific
8.2.3.5.3.1. Market
Estimation, 2013 – 2018
8.2.3.5.3.2. Market
Forecast, 2019 – 2027
8.2.3.5.4. Middle East and Africa
8.2.3.5.4.1. Market
Estimation, 2013 – 2018
8.2.3.5.4.2. Market
Forecast, 2019 – 2027
8.2.3.5.5. Latin America
8.2.3.5.5.1. Market
Estimation, 2013 – 2018
8.2.3.5.5.2. Market
Forecast, 2019 – 2027
8.2.4. Bioburden
testing
8.2.4.1. Definition
8.2.4.2. Market Estimation and Penetration, 2013 – 2018
8.2.4.3. Market Forecast, 2019 – 2027
8.2.4.4. Compound Annual Growth Rate (CAGR)
8.2.4.5. Regional Bifurcation
8.2.4.5.1. North America
8.2.4.5.1.1. Market
Estimation, 2013 – 2018
8.2.4.5.1.2. Market
Forecast, 2019 – 2027
8.2.4.5.2. Europe
8.2.4.5.2.1. Market
Estimation, 2013 – 2018
8.2.4.5.2.2. Market
Forecast, 2019 – 2027
8.2.4.5.3. Asia Pacific
8.2.4.5.3.1. Market
Estimation, 2013 – 2018
8.2.4.5.3.2. Market
Forecast, 2019 – 2027
8.2.4.5.4. Middle East and Africa
8.2.4.5.4.1. Market
Estimation, 2013 – 2018
8.2.4.5.4.2. Market
Forecast, 2019 – 2027
8.2.4.5.5. Latin America
8.2.4.5.5.1. Market
Estimation, 2013 – 2018
8.2.4.5.5.2. Market
Forecast, 2019 – 2027
8.2.5. Sterility
testing
8.2.5.1. Definition
8.2.5.2. Market Estimation and Penetration, 2013 – 2018
8.2.5.3. Market Forecast, 2019 – 2027
8.2.5.4. Compound Annual Growth Rate (CAGR)
8.2.5.5. Regional Bifurcation
8.2.5.5.1. North America
8.2.5.5.1.1. Market
Estimation, 2013 – 2018
8.2.5.5.1.2. Market
Forecast, 2019 – 2027
8.2.5.5.2. Europe
8.2.5.5.2.1. Market
Estimation, 2013 – 2018
8.2.5.5.2.2. Market
Forecast, 2019 – 2027
8.2.5.5.3. Asia Pacific
8.2.5.5.3.1. Market
Estimation, 2013 – 2018
8.2.5.5.3.2. Market
Forecast, 2019 – 2027
8.2.5.5.4. Middle East and Africa
8.2.5.5.4.1. Market
Estimation, 2013 – 2018
8.2.5.5.4.2. Market
Forecast, 2019 – 2027
8.2.5.5.5. Latin America
8.2.5.5.5.1. Market
Estimation, 2013 – 2018
8.2.5.5.5.2. Market
Forecast, 2019 – 2027
8.3. Key
Segment for Channeling Investments
8.3.1. By
Services
9.
North America Medical
Device Analytical Testing Outsourcing Market Analysis and Forecasts, 2019 -
2027
9.1. Overview
9.1.1. North
America Medical Device Analytical Testing Outsourcing Market Revenue (US$ Mn)
9.2. North
America Medical Device Analytical Testing Outsourcing Market Revenue (US$ Mn)
and Forecasts, By Services
9.2.1. Extractable
and leachable
9.2.2. Material
characterization
9.2.3. Physical
testing
9.2.4. Bioburden
testing
9.2.5. Sterility
testing
9.3. North
America Medical Device Analytical Testing Outsourcing Market Revenue (US$ Mn)
and Forecasts, By Country
9.3.1. U.S
9.3.1.1. U.S Medical Device Analytical Testing Outsourcing Market
Revenue (US$ Mn) and Forecasts, By Services
9.3.1.1.1. Extractable and leachable
9.3.1.1.2. Material characterization
9.3.1.1.3. Physical testing
9.3.1.1.4. Bioburden testing
9.3.1.1.5. Sterility testing
9.3.2. Canada
9.3.2.1. Canada Medical Device Analytical Testing Outsourcing Market
Revenue (US$ Mn) and Forecasts, By Services
9.3.2.1.1. Extractable and leachable
9.3.2.1.2. Material characterization
9.3.2.1.3. Physical testing
9.3.2.1.4. Bioburden testing
9.3.2.1.5. Sterility testing
9.3.3. Mexico
9.3.3.1. Mexico Medical Device Analytical Testing Outsourcing Market
Revenue (US$ Mn) and Forecasts, By Services
9.3.3.1.1. Extractable and leachable
9.3.3.1.2. Material characterization
9.3.3.1.3. Physical testing
9.3.3.1.4. Bioburden testing
9.3.3.1.5. Sterility testing
9.3.4. Rest of
North America
9.3.4.1. Rest of North America Medical Device Analytical Testing
Outsourcing Market Revenue (US$ Mn) and Forecasts, By Services
9.3.4.1.1. Extractable and leachable
9.3.4.1.2. Material characterization
9.3.4.1.3. Physical testing
9.3.4.1.4. Bioburden testing
9.3.4.1.5. Sterility testing
9.4. Key
Segment for Channeling Investments
9.4.1. By
Country
9.4.2. By
Services
10.
Europe Medical Device
Analytical Testing Outsourcing Market Analysis and Forecasts, 2019 - 2027
10.1. Overview
10.1.1. Europe
Medical Device Analytical Testing Outsourcing Market Revenue (US$ Mn)
10.2. Europe
Medical Device Analytical Testing Outsourcing Market Revenue (US$ Mn) and
Forecasts, By Services
10.2.1. Extractable
and leachable
10.2.2. Material
characterization
10.2.3. Physical
testing
10.2.4. Bioburden
testing
10.2.5. Sterility
testing
10.3. Europe
Medical Device Analytical Testing Outsourcing Market Revenue (US$ Mn) and
Forecasts, By Country
10.3.1. France
10.3.1.1. France Medical Device Analytical Testing Outsourcing Market
Revenue (US$ Mn) and Forecasts, By Services
10.3.1.1.1. Extractable and leachable
10.3.1.1.2. Material characterization
10.3.1.1.3. Physical testing
10.3.1.1.4. Bioburden testing
10.3.1.1.5. Sterility testing
10.3.2. The UK
10.3.2.1. The UK Medical Device Analytical Testing Outsourcing Market
Revenue (US$ Mn) and Forecasts, By Services
10.3.2.1.1. Extractable and leachable
10.3.2.1.2. Material characterization
10.3.2.1.3. Physical testing
10.3.2.1.4. Bioburden testing
10.3.2.1.5. Sterility testing
10.3.3. Spain
10.3.3.1. Spain Medical Device Analytical Testing Outsourcing Market
Revenue (US$ Mn) and Forecasts, By Services
10.3.3.1.1. Extractable and leachable
10.3.3.1.2. Material characterization
10.3.3.1.3. Physical testing
10.3.3.1.4. Bioburden testing
10.3.3.1.5. Sterility testing
10.3.4. Germany
10.3.4.1. Germany Medical Device Analytical Testing Outsourcing Market
Revenue (US$ Mn) and Forecasts, By Services
10.3.4.1.1. Extractable and leachable
10.3.4.1.2. Material characterization
10.3.4.1.3. Physical testing
10.3.4.1.4. Bioburden testing
10.3.4.1.5. Sterility testing
10.3.5. Italy
10.3.5.1. Italy Medical Device Analytical Testing Outsourcing Market
Revenue (US$ Mn) and Forecasts, By Services
10.3.5.1.1. Extractable and leachable
10.3.5.1.2. Material characterization
10.3.5.1.3. Physical testing
10.3.5.1.4. Bioburden testing
10.3.5.1.5. Sterility testing
10.3.6. Nordic
Countries
10.3.6.1. Nordic Countries Medical Device Analytical Testing Outsourcing
Market Revenue (US$ Mn) and Forecasts, By Services
10.3.6.1.1. Extractable and leachable
10.3.6.1.2. Material characterization
10.3.6.1.3. Physical testing
10.3.6.1.4. Bioburden testing
10.3.6.1.5. Sterility testing
10.3.6.2. Nordic Countries Medical Device Analytical Testing Outsourcing
Market Revenue (US$ Mn) and Forecasts, By Country
10.3.6.2.1. Denmark
10.3.6.2.2. Finland
10.3.6.2.3. Iceland
10.3.6.2.4. Sweden
10.3.6.2.5. Norway
10.3.7. Benelux
Union
10.3.7.1. Benelux Union Medical Device Analytical Testing Outsourcing
Market Revenue (US$ Mn) and Forecasts, By Services
10.3.7.1.1. Extractable and leachable
10.3.7.1.2. Material characterization
10.3.7.1.3. Physical testing
10.3.7.1.4. Bioburden testing
10.3.7.1.5. Sterility testing
10.3.7.2. Benelux Union Medical Device Analytical Testing Outsourcing
Market Revenue (US$ Mn) and Forecasts, By Country
10.3.7.2.1. Belgium
10.3.7.2.2. The Netherlands
10.3.7.2.3. Luxembourg
10.3.8. Rest of
Europe
10.3.8.1. Rest of Europe Medical Device Analytical Testing Outsourcing
Market Revenue (US$ Mn) and Forecasts, By Services
10.3.8.1.1. Extractable and leachable
10.3.8.1.2. Material characterization
10.3.8.1.3. Physical testing
10.3.8.1.4. Bioburden testing
10.3.8.1.5. Sterility testing
10.4. Key
Segment for Channeling Investments
10.4.1. By
Country
10.4.2. By
Services
11.
Asia Pacific Medical Device
Analytical Testing Outsourcing Market Analysis and Forecasts, 2019 - 2027
11.1. Overview
11.1.1. Asia
Pacific Medical Device Analytical Testing Outsourcing Market Revenue (US$ Mn)
11.2. Asia
Pacific Medical Device Analytical Testing Outsourcing Market Revenue (US$ Mn)
and Forecasts, By Services
11.2.1. Extractable
and leachable
11.2.2. Material
characterization
11.2.3. Physical
testing
11.2.4. Bioburden
testing
11.2.5. Sterility
testing
11.3. Asia
Pacific Medical Device Analytical Testing Outsourcing Market Revenue (US$ Mn)
and Forecasts, By Country
11.3.1. China
11.3.1.1. China Medical Device Analytical Testing Outsourcing Market
Revenue (US$ Mn) and Forecasts, By Services
11.3.1.1.1. Extractable and leachable
11.3.1.1.2. Material characterization
11.3.1.1.3. Physical testing
11.3.1.1.4. Bioburden testing
11.3.1.1.5. Sterility testing
11.3.2. Japan
11.3.2.1. Japan Medical Device Analytical Testing Outsourcing Market
Revenue (US$ Mn) and Forecasts, By Services
11.3.2.1.1. Extractable and leachable
11.3.2.1.2. Material characterization
11.3.2.1.3. Physical testing
11.3.2.1.4. Bioburden testing
11.3.2.1.5. Sterility testing
11.3.3. India
11.3.3.1. India Medical Device Analytical Testing Outsourcing Market
Revenue (US$ Mn) and Forecasts, By Services
11.3.3.1.1. Extractable and leachable
11.3.3.1.2. Material characterization
11.3.3.1.3. Physical testing
11.3.3.1.4. Bioburden testing
11.3.3.1.5. Sterility testing
11.3.4. New
Zealand
11.3.4.1. New Zealand Medical Device Analytical Testing Outsourcing
Market Revenue (US$ Mn) and Forecasts, By Services
11.3.4.1.1. Extractable and leachable
11.3.4.1.2. Material characterization
11.3.4.1.3. Physical testing
11.3.4.1.4. Bioburden testing
11.3.4.1.5. Sterility testing
11.3.5. Australia
11.3.5.1. Australia Medical Device Analytical Testing Outsourcing Market
Revenue (US$ Mn) and Forecasts, By Services
11.3.5.1.1. Extractable and leachable
11.3.5.1.2. Material characterization
11.3.5.1.3. Physical testing
11.3.5.1.4. Bioburden testing
11.3.5.1.5. Sterility testing
11.3.6. South
Korea
11.3.6.1. South Korea Medical Device Analytical Testing Outsourcing
Market Revenue (US$ Mn) and Forecasts, By Services
11.3.6.1.1. Extractable and leachable
11.3.6.1.2. Material characterization
11.3.6.1.3. Physical testing
11.3.6.1.4. Bioburden testing
11.3.6.1.5. Sterility testing
11.3.7. Southeast
Asia
11.3.7.1. Southeast Asia Medical Device Analytical Testing Outsourcing
Market Revenue (US$ Mn) and Forecasts, By Services
11.3.7.1.1. Extractable and leachable
11.3.7.1.2. Material characterization
11.3.7.1.3. Physical testing
11.3.7.1.4. Bioburden testing
11.3.7.1.5. Sterility testing
11.3.7.2. Southeast Asia Medical Device Analytical Testing Outsourcing
Market Revenue (US$ Mn) and Forecasts, By Country
11.3.7.2.1. Indonesia
11.3.7.2.2. Thailand
11.3.7.2.3. Malaysia
11.3.7.2.4. Singapore
11.3.7.2.5. Rest of Southeast Asia
11.3.8. Rest of
Asia Pacific
11.3.8.1. Rest of Asia Pacific Medical Device Analytical Testing
Outsourcing Market Revenue (US$ Mn) and Forecasts, By Services
11.3.8.1.1. Extractable and leachable
11.3.8.1.2. Material characterization
11.3.8.1.3. Physical testing
11.3.8.1.4. Bioburden testing
11.3.8.1.5. Sterility testing
11.4. Key
Segment for Channeling Investments
11.4.1. By
Country
11.4.2. By
Services
12.
Middle East and Africa
Medical Device Analytical Testing Outsourcing Market Analysis and Forecasts, 2019
- 2027
12.1. Overview
12.1.1. Middle
East and Africa Medical Device Analytical Testing Outsourcing Market Revenue
(US$ Mn)
12.2. Middle
East and Africa Medical Device Analytical Testing Outsourcing Market Revenue
(US$ Mn) and Forecasts, By Services
12.2.1. Extractable
and leachable
12.2.2. Material
characterization
12.2.3. Physical
testing
12.2.4. Bioburden
testing
12.2.5. Sterility
testing
12.3. Middle
East and Africa Medical Device Analytical Testing Outsourcing Market Revenue
(US$ Mn) and Forecasts, By Country
12.3.1. Saudi
Arabia
12.3.1.1. Saudi Arabia Medical Device Analytical Testing Outsourcing
Market Revenue (US$ Mn) and Forecasts, By Services
12.3.1.1.1. Extractable and leachable
12.3.1.1.2. Material characterization
12.3.1.1.3. Physical testing
12.3.1.1.4. Bioburden testing
12.3.1.1.5. Sterility testing
12.3.2. UAE
12.3.2.1. UAE Medical Device Analytical Testing Outsourcing Market
Revenue (US$ Mn) and Forecasts, By Services
12.3.2.1.1. Extractable and leachable
12.3.2.1.2. Material characterization
12.3.2.1.3. Physical testing
12.3.2.1.4. Bioburden testing
12.3.2.1.5. Sterility testing
12.3.3. Egypt
12.3.3.1. Egypt Medical Device Analytical Testing Outsourcing Market
Revenue (US$ Mn) and Forecasts, By Services
12.3.3.1.1. Extractable and leachable
12.3.3.1.2. Material characterization
12.3.3.1.3. Physical testing
12.3.3.1.4. Bioburden testing
12.3.3.1.5. Sterility testing
12.3.4. Kuwait
12.3.4.1. Kuwait Medical Device Analytical Testing Outsourcing Market
Revenue (US$ Mn) and Forecasts, By Services
12.3.4.1.1. Extractable and leachable
12.3.4.1.2. Material characterization
12.3.4.1.3. Physical testing
12.3.4.1.4. Bioburden testing
12.3.4.1.5. Sterility testing
12.3.5. South
Africa
12.3.5.1. South Africa Medical Device Analytical Testing Outsourcing
Market Revenue (US$ Mn) and Forecasts, By Services
12.3.5.1.1. Extractable and leachable
12.3.5.1.2. Material characterization
12.3.5.1.3. Physical testing
12.3.5.1.4. Bioburden testing
12.3.5.1.5. Sterility testing
12.3.6. Rest of
Middle East & Africa
12.3.6.1. Rest of Middle East & Africa Medical Device Analytical
Testing Outsourcing Market Revenue (US$ Mn) and Forecasts, By Services
12.3.6.1.1. Extractable and leachable
12.3.6.1.2. Material characterization
12.3.6.1.3. Physical testing
12.3.6.1.4. Bioburden testing
12.3.6.1.5. Sterility testing
12.4. Key
Segment for Channeling Investments
12.4.1. By
Country
12.4.2. By
Services
13.
Latin America Medical
Device Analytical Testing Outsourcing Market Analysis and Forecasts, 2019 -
2027
13.1. Overview
13.1.1. Latin
America Medical Device Analytical Testing Outsourcing Market Revenue (US$ Mn)
13.2. Latin
America Medical Device Analytical Testing Outsourcing Market Revenue (US$ Mn)
and Forecasts, By Services
13.2.1. Extractable
and leachable
13.2.2. Material
characterization
13.2.3. Physical
testing
13.2.4. Bioburden
testing
13.2.5. Sterility
testing
13.3. Latin America
Medical Device Analytical Testing Outsourcing Market Revenue (US$ Mn) and
Forecasts, By Country
13.3.1. Brazil
13.3.1.1. Brazil Medical Device Analytical Testing Outsourcing Market
Revenue (US$ Mn) and Forecasts, By Services
13.3.1.1.1. Extractable and leachable
13.3.1.1.2. Material characterization
13.3.1.1.3. Physical testing
13.3.1.1.4. Bioburden testing
13.3.1.1.5. Sterility testing
13.3.2. Argentina
13.3.2.1. Argentina Medical Device Analytical Testing Outsourcing Market
Revenue (US$ Mn) and Forecasts, By Services
13.3.2.1.1. Extractable and leachable
13.3.2.1.2. Material characterization
13.3.2.1.3. Physical testing
13.3.2.1.4. Bioburden testing
13.3.2.1.5. Sterility testing
13.3.3. Rest of
Latin America
13.3.3.1. Rest of Latin America Medical Device Analytical Testing
Outsourcing Market Revenue (US$ Mn) and Forecasts, By Services
13.3.3.1.1. Extractable and leachable
13.3.3.1.2. Material characterization
13.3.3.1.3. Physical testing
13.3.3.1.4. Bioburden testing
13.3.3.1.5. Sterility testing
13.4. Key
Segment for Channeling Investments
13.4.1. By
Country
13.4.2. By
Services
14.
Competitive Benchmarking
14.1. Market
Share Analysis, 2018
14.2. Global
Presence and Growth Strategies
14.2.1. Mergers
and Acquisitions
14.2.2. Product
Launches
14.2.3. Investments
Trends
14.2.4. R&D
Initiatives
15.
Player Profiles
15.1. Charles
River Laboratories International Inc.
15.1.1. Company
Details
15.1.2. Company
Overview
15.1.3. Product
Offerings
15.1.4. Key
Developments
15.1.5. Financial
Analysis
15.1.6. SWOT
Analysis
15.1.7. Business
Strategies
15.2. Envigo
15.2.1. Company
Details
15.2.2. Company
Overview
15.2.3. Product
Offerings
15.2.4. Key
Developments
15.2.5. Financial
Analysis
15.2.6. SWOT
Analysis
15.2.7. Business
Strategies
15.3. Eurofins
Scientific
15.3.1. Company
Details
15.3.2. Company
Overview
15.3.3. Product
Offerings
15.3.4. Key
Developments
15.3.5. Financial
Analysis
15.3.6. SWOT
Analysis
15.3.7. Business
Strategies
15.4. Intertek
Group plc
15.4.1. Company
Details
15.4.2. Company
Overview
15.4.3. Product
Offerings
15.4.4. Key
Developments
15.4.5. Financial
Analysis
15.4.6. SWOT
Analysis
15.4.7. Business
Strategies
15.5. Medical
Device Testing Services
15.5.1. Company
Details
15.5.2. Company
Overview
15.5.3. Product
Offerings
15.5.4. Key
Developments
15.5.5. Financial
Analysis
15.5.6. SWOT
Analysis
15.5.7. Business
Strategies
15.6. NORTH
AMERICAN SCIENCE ASSOCIATES INC.
15.6.1. Company
Details
15.6.2. Company
Overview
15.6.3. Product
Offerings
15.6.4. Key
Developments
15.6.5. Financial
Analysis
15.6.6. SWOT
Analysis
15.6.7. Business
Strategies
15.7. Pace
Analytical Services, LLC
15.7.1. Company
Details
15.7.2. Company
Overview
15.7.3. Product
Offerings
15.7.4. Key
Developments
15.7.5. Financial
Analysis
15.7.6. SWOT
Analysis
15.7.7. Business
Strategies
15.8. SGS SA
15.8.1. Company
Details
15.8.2. Company
Overview
15.8.3. Product
Offerings
15.8.4. Key
Developments
15.8.5. Financial
Analysis
15.8.6. SWOT
Analysis
15.8.7. Business
Strategies
15.9. Toxikon,
Inc.
15.9.1. Company
Details
15.9.2. Company
Overview
15.9.3. Product
Offerings
15.9.4. Key
Developments
15.9.5. Financial
Analysis
15.9.6. SWOT
Analysis
15.9.7. Business
Strategies
15.10. WuXi
AppTec
15.10.1. Company
Details
15.10.2. Company
Overview
15.10.3. Product
Offerings
15.10.4. Key
Developments
15.10.5. Financial
Analysis
15.10.6. SWOT
Analysis
15.10.7. Business
Strategies
15.11. Other
Market Participants
16.
Key Findings
Note: This ToC is tentative
and can be changed according to the research study conducted during the course
of report completion.
**Exclusive for Multi-User and
Enterprise User.
At Absolute Markets Insights, we are engaged in building both global as well as country specific reports. As a result, the approach taken for deriving the estimation and forecast for a specific country is a bit unique and different in comparison to the global research studies. In this case, we not only study the concerned market factors & trends prevailing in a particular country (from secondary research) but we also tend to calculate the actual market size & forecast from the revenue generated from the market participants involved in manufacturing or distributing the any concerned product. These companies can also be service providers. For analyzing any country specifically, we do consider the growth factors prevailing under the states/cities/county for the same. For instance, if we are analyzing an industry specific to United States, we primarily need to study about the states present under the same(where the product/service has the highest growth). Similar analysis will be followed by other countries. Our scope of the report changes with different markets.
Our research study is mainly implement through a mix of both secondary and primary research. Various sources such as industry magazines, trade journals, and government websites and trade associations are reviewed for gathering precise data. Primary interviews are conducted to validate the market size derived from secondary research. Industry experts, major manufacturers and distributors are contacted for further validation purpose on the current market penetration and growth trends.
Prominent participants in our primary research process include:
- Key Opinion Leaders namely the CEOs, CSOs, VPs, purchasing managers, amongst others
- Research and development participants, distributors/suppliers and subject matter experts
Secondary Research includes data extracted from paid data sources:
- Reuters
- Factiva
- Bloomberg
- One Source
- Hoovers
Research Methodology
Key Inclusions

Reach to us
Call us on
+91-74002-42424
Drop us an email at
sales@absolutemarketsinsights.com
Why Absolute Markets Insights?
An effective strategy is the entity that influences a business to stand out of the crowd. An organization with a phenomenal strategy for success dependably has the edge over the rivals in the market. It offers the organizations a head start in planning their strategy. Absolute Market Insights is the new initiation in the industry that will furnish you with the lead your business needs. Absolute Market Insights is the best destination for your business intelligence and analytical solutions; essentially because our qualitative and quantitative sources of information are competent to give one-stop solutions. We inventively combine qualitative and quantitative research in accurate proportions to have the best report, which not only gives the most recent insights but also assists you to grow.