Medical Device Market By Type (Electronics and Equipment, Diagnostic Imaging: X-Ray Equipment, Ultrasound Systems, Computed Tomography (CT) Scanners, Magnetic Resonance Imaging Systems, Others) (Patent Monitoring: Cardiac Monitoring Devices, Respiratory Monitoring Devices, Others) (Diabetic Care Devices: Insulin Pens, Blood Glucose Meters, Blood Glucose Strips, Continuous Glucose Monitoring Devices, Others) (Orthopedic Devices: Prosthetics, Braces, Fixation Devices, Others) (Dental Diagnostics and Operative Devices),(Others) (Disposables and Consumables: Wound Management Products, Drug Delivery Products, Diagnostic and Laboratory Disposables, Dialysis Disposables, Incontinence Products, Respiratory Supplies, Sterilization Supplies, Non-woven Disposables, Disposable Masks, Disposable Gloves, Hand Sanitizers- Gel Sanitizers, Foam Sanitizers, Liquid Sanitizers, Others)(Surgical Instruments: Surgical Sutures and Staplers, Handheld Surgical Devices- Forceps and Spatulas, Retractors, Dilators, Graspers, Auxiliary Instruments, Cutter Instruments, Others)(IVD Reagents: Antibodies- Monoclonal, Polyclonal)(Oligonucleotides, Nucleic Acid Probes, Purified Proteins or Peptides, Other Reagents) (Implants: Orthopedic Implants, ENT Implants, Breast Implants, Dental Implants, Cardiology related implants, others) By Region (U.S., Canada, Mexico, Rest of North America, France, UK, Germany, Spain, Italy, Nordic Countries, Benelux Union, Rest of Europe, China, Japan, India, New Zealand, Australia, South Korea, Southeast Asia, Rest of Asia Pacific, Saudi Arabia, UAE, Egypt, Kuwait, South Africa, Rest of MEA, Brazil, Argentina, Rest of Latin America) – Global Insights, Growth, Size, Comparative Analysis, Trends and Forecast, 2021 – 2029
Industry Trends
Global medical device market was valued at US$ 509.81 billion in 2020 and is expected to reach US$ 972.54 billion by 2029, growing at an estimated CAGR of 7.6% over the forecast period. Healthcare industry, one of those very few industries those have managed to reach the trillion dollar-marks in terms of revenue generation and major chunk of it comes from the medical devices market. The dreadful COVID-19 which has been wreaking havoc across the globe is only aiding the market to grow more. The demand for medical equipments like oxygen concentrators, ventilators and oximeters is going upwards each passing day because of the increasing number of hospitalizations.
Though the supply chain in this industry has been disrupted owing to the on-going situation, majority of the countries still have certain trade restrictions on. Along with this, extensive use of remote patient monitoring devices is likely to increase threats posed by data security issues.
While the healthcare infrastructure in most of the countries had almost collapsed in the recent pandemic, keeping this in mind, the hospitals and medical organisations will be more willing to be prepared for any upcoming epidemic or pandemic and in order to do so, they are expected to purchase all essential medical equipments. This clearly showcases that the medical devices market is more likely to witness growth in coming future.
Type
Sale of electronics equipments used by medical institutions dominates the market with highest share mainly because the demand for these products is constantly surging. Every type of health facility, irrespective of its size requires certain electronic medical devices to ensure right treatment to its patients.
Similarly, in order to know root cause of any disease, proper diagnosis is needed and this is only possible with the help of technically advanced medical equipments. Right from orthopaedic ailments to neurological issues, certain diagnostic imaging machines like X-RAY, ultrasound systems, CT scanners and magnetic resonance imaging systems are required for accurate diagnosis. This is the reason why these imaging machines have highest penetration in the medical devices market.
Just like diagnostic imaging equipment, machines used for monitoring the patients are sold in higher numbers. Increases cases of cardiovascular diseases over the period of last few years have resulted in surged demand for cardiac monitoring devices.
Today, chronical disease like diabetes kills more people as compared to any other disease. The number of people suffering from diabetes is increasing each passing day and this has resulted in a spike in the sales of insulin pens, blood glucose meters, and continuous glucose monitoring devices.
The sales of disposables and consumables have highest contribution to the medical devices market in terms of CAGR. Ever since the COVID-19 pandemic, the use for disposable masks, eye gear and hand sanitizers have gone up ultimately increasing demand for them.
Surgeries and medical procedure are integral part of any treatment and this requires instruments like surgical sutures, staplers to seal the wound or a cut. On the other hand, instruments like forceps and retractors are mainly used in oral treatments.
Similar to disposables, reagents like antibodies are witnessing a high demand. Antibodies of the patients who have successfully recovered from the COVID-19 are seen as a potential treatment.
On the other hand, replacement of any body part is a common type of treatment today. With the help of the science and technology, primarily with 3D printing technology, replica of most of the body parts including the heart can be created. While dental and orthopaedic implants are used extensively, breast implants are also used on a larger scale by medical experts in cases of breast cancer and fashion industry.
Region
The region of the North America continued to be biggest market for the medical devices mainly because of favorable government rules and regulations along with more advanced healthcare infrastructure, extensive use of new medical technologies and presence of leading companies in the sector. The United States is the world's largest medical device market and it shows no signs of slowing. The US medical device industry was valued at US$178.43 billion in 2020 and is projected to grow significantly by 2029 growing at a CAGR of 7.1%.
Medical Devices Market Revenue & Forecast, (US$ Million), 2021 – 2029
Competitive Landscape
The report provides both, qualitative and quantitative research of global medical devices market, as well as provides comprehensive insights and development methods adopted by the key contenders. The report also offers extensive research on the key players in this market and details on the competitiveness of these players. Key business strategies such as mergers and acquisitions (M&A), affiliations, collaborations, and contacts adopted by these major market participants are also recognized and analysed in the report. For each company, the report studies their global presence, competitors, service offerings and specification amongst others.
Some of the major players operating of the medical devices market are Johnson & Johnson, Medtronic, Stryker, Philips, Siemens Healthineers and 3M Company amongst others.
Medical Devices Market
By Type
- ? Electronics and Equipment
- ? Diagnostic Imaging
- o X-Ray Equipment
- o Ultrasound Systems
- o Computed Tomography (CT) Scanners
- o Magnetic Resonance Imaging Systems
- o Others
- ? Patient Monitoring
- ? Cardiac Monitoring Devices
- ? Respiratory Monitoring Devices
- ? Others
- ? Diabetic Care Devices
- ? Insulin Pens
- ? Blood Glucose Meters
- ? Blood Glucose Test Strips
- ? Continuous Glucose Monitoring Device
- ? Others
- ? Orthopedic Devices
- ? Prosthetics
- ? Braces
- ? Fixation Devices
- ? Others
- ? Diagnostic Imaging
- ? Dental Diagnostics and Operative Devices
- ? Others
- ? Disposables and Consumables
- ? Wound Management Products
- ? Drug Delivery Products
- ? Diagnostic and Laboratory Disposables
- ? Dialysis Disposables
- ? Incontinence Products
- ? Respiratory Supplies
- ? Sterilization Supplies
- ? Non-woven Disposables
- ? Disposable Masks
- ? Disposable Eye Gear
- ? Disposable Gloves
- ? Hand Sanitizers
- o Gel Sanitizers
- o Foam Sanitizers
- o Liquid Sanitizers
- o Other Sanitizers
- ? Others
- ? Surgical Instruments
- ? Surgical Sutures and Staplers
- ? Handheld Surgical Devices
- o Forceps and Spatulas
- o Retractors
- o Dilators
- o Graspers
- o Auxiliary Instruments
- o Cutter Instruments
- o Others
- ? Electrosurgical Devices
- ? Others
- ? IVD Reagents
- ? Antibodies
- ? Monoclonal
- ? Polyclonal
- ? Oligonucleotides
- ? Nucleic Acid Probes
- ? Purified Proteins or Peptides
- ? Other Reagents
- ? Antibodies
- ? Implants
- ? Orthopedic Implants
- ? ENT Implants
- ? Breast Implants
- ? Dental Implants
- ? Cardiology related implants
- ? Others
- ? Others
By Geography
- North America
- US
- Canada
- Mexico
- Rest of North America
- Europe
- France
- The UK
- Spain
- Germany
- Italy
- Nordic Countries
- Denmark
- Finland
- Iceland
- Sweden
- Norway
- Benelux Union
- Belgium
- The Netherlands
- Luxembourg
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- New Zealand
- Australia
- South Korea
- Southeast Asia
- Indonesia
- Thailand
- Malaysia
- Singapore
- Rest of Southeast Asia
- Rest of Asia Pacific
- Middle East and Africa
- Saudi Arabia
- UAE
- Egypt
- Kuwait
- South Africa
- Rest of Middle East & Africa
- Latin America
- Brazil
- Argentina
- Rest of Latin America
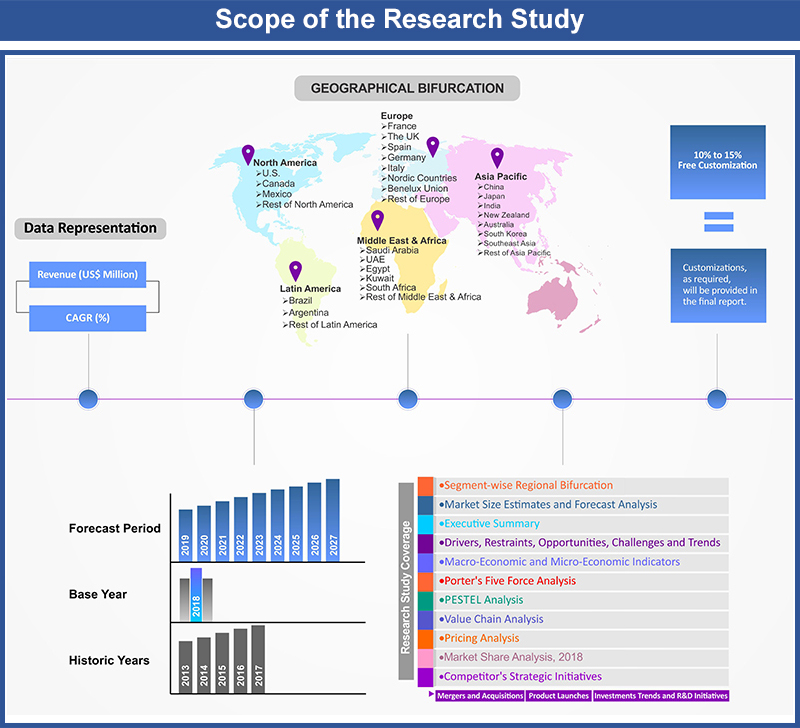
Table of Contents
![]()
1.
Market Scope
1.1. Market Segmentation
1.2. Years Considered
1.2.1. Historic Years: 2015 - 2018
1.2.2. Base Year: 2019
1.2.3. Forecast Years: 2020 – 2028
2. Key Target Audiences
3. Research Methodology
3.1. Primary Research
3.1.1. Research Questionnaire
3.1.2. Global Percentage Breakdown
3.1.3. Primary Interviews: Key Opinion Leaders (KOLs)
3.2. Secondary Research
3.2.1. Paid Databases
3.2.2. Secondary Sources
3.3. Market Size Estimates
3.3.1. Top-Down Approach
3.3.2. Bottom-Up Approach
3.4. Data Triangulation Methodology
3.5. Research Assumptions
4. Recommendations and Insights from AMI’s Perspective**
5. Holistic Overview of Medical
Device Market
6. Market Synopsis:
Medical Device Market
7. Medical Device Market
Analysis: Qualitative Perspective
7.1. Introduction
7.1.1. Product Definition
7.1.2. Industry Development
7.2. Market Dynamics
7.2.1. Drivers
7.2.2. Restraints
7.2.3. Opportunities
7.2.4. Challenges
7.3. Trends in Medical
Device Market
7.4. Market Determinants Radar Chart
7.5. Macro-Economic and Micro-Economic Indicators: Medical Device Market
7.6. Industry Gross Margin Overview
7.7. Porter’s Five Force Analysis
7.8. Impact of Covid-19 on this market
8. Global Medical Device
Market Analysis and Forecasts, 2020 – 2028
8.1. Overview
8.1.1. Global Medical Device
Market Revenue (US$ Mn)
8.2. Global Medical Device
Market Revenue (US$ Mn) and Forecasts, By Type
8.2.1. Electronics and
Equipment (Definition, Market Estimation and Penetration, 2015-2019, Market
Estimation (2015-2019), Market Forecast (2020 – 2028), Compound Annual Growth
Rate (CAGR), Regional Bifurcation (North America, Europe, Asia Pacific, Middle
East and Africa, Latin America) and Information on Diagnostic Imaging, Patient
Monitoring, Diabetic Care Devices, Orthopedic Devices, Dental Diagnostics and
Operative Devices, Others)
8.2.1.1. Diagnostic Imaging
8.2.1.1.1. X-Ray Equipment
8.2.1.1.2. Ultrasound Systems
8.2.1.1.3. Computed Tomography (CT) Scanners
8.2.1.1.4. Magnetic Resonance Imaging Systems
8.2.1.1.5. Others
8.2.1.2. Patient Monitoring
8.2.1.2.1. Cardiac Monitoring Devices
8.2.1.2.2. Respiratory Monitoring Devices
8.2.1.2.3. Others
8.2.1.3. Diabetic Care Devices
8.2.1.3.1. Insulin Pens
8.2.1.3.2. Blood Glucose Meters
8.2.1.3.3. Blood Glucose Test Strips
8.2.1.3.4. Continuous Glucose Monitoring Device
8.2.1.3.5. Others
8.2.1.4. Orthopedic Devices
8.2.1.4.1. Prosthetics
8.2.1.4.2. Braces
8.2.1.4.3. Fixation Devices
8.2.1.4.4. Others
8.2.1.5. Dental Diagnostics and Operative Devices
8.2.1.6. Others
8.2.2. Disposables and Consumables – Highest CAGR (Definition, Market
Estimation and Penetration, 2015-2019, Market Estimation (2015-2019), Market
Forecast (2020 – 2028), Compound Annual Growth Rate (CAGR), Regional
Bifurcation (North America, Europe, Asia Pacific, Middle East and Africa, Latin
America) and Information on Wound Management Products, Drug Delivery Products, Diagnostic and Laboratory Disposables, Dialysis Disposables, Incontinence Products,
Respiratory Supplies, Sterilization Supplies,
Non-woven Disposables, Disposable Masks, Disposable Eye Gear, Disposable
Gloves, Hand Sanitizers, Others)
8.2.2.1. Wound Management Products
8.2.2.2. Drug Delivery Products
8.2.2.3. Diagnostic and
Laboratory Disposables
8.2.2.4. Dialysis Disposables
8.2.2.5. Incontinence Products
8.2.2.6. Respiratory Supplies
8.2.2.7. Sterilization Supplies
8.2.2.8. Non-woven Disposables
8.2.2.9. Disposable Masks
8.2.2.10.
Disposable Eye Gear
8.2.2.11.
Disposable Gloves
8.2.2.12.
Hand Sanitizers
8.2.2.12.1. Gel Sanitizers
8.2.2.12.2. Foam Sanitizers
8.2.2.12.3. Liquid Sanitizers
8.2.2.12.4. Other Sanitizers
8.2.2.13.
Others
8.2.3. Surgical Instruments (Definition, Market Estimation and
Penetration, 2015-2019, Market Estimation (2015-2019), Market Forecast (2020 –
2028), Compound Annual Growth Rate (CAGR), Regional Bifurcation (North America,
Europe, Asia Pacific, Middle East and Africa, Latin America) and Information on
Surgical Sutures and Staplers, Handheld Surgical Devices, Electrosurgical
Devices, Others)
8.2.3.1. Surgical Sutures and Staplers
8.2.3.2. Handheld Surgical Devices
8.2.3.2.1. Forceps and Spatulas
8.2.3.2.2. Retractors
8.2.3.2.3. Dilators
8.2.3.2.4. Graspers
8.2.3.2.5. Auxiliary Instruments
8.2.3.2.6. Cutter Instruments
8.2.3.2.7. Others
8.2.3.3. Electrosurgical Devices
8.2.3.4. Others
8.2.4. IVD Reagents (Definition, Market Estimation and Penetration,
2015-2019, Market Estimation (2015-2019), Market Forecast (2020 – 2028),
Compound Annual Growth Rate (CAGR), Regional Bifurcation (North America,
Europe, Asia Pacific, Middle East and Africa, Latin America) and Information on
Antibodies, Oligonucleotides, Nucleic Acid Probes, Purified Proteins or
Peptides, Other Reagents)
8.2.4.1. Antibodies
8.2.4.1.1. Monoclonal
8.2.4.1.2. Polyclonal
8.2.4.2. Oligonucleotides
8.2.4.3. Nucleic Acid Probes
8.2.4.4. Purified Proteins or Peptides
8.2.4.5. Other Reagents
8.2.5. Implants (Definition, Market Estimation and Penetration,
2015-2019, Market Estimation (2015-2019), Market Forecast (2020 – 2028),
Compound Annual Growth Rate (CAGR), Regional Bifurcation (North America,
Europe, Asia Pacific, Middle East and Africa, Latin America) and Information
on Orthopedic Implants, ENT Implants,
Breast Implants, Dental Implants,
Cardiology related implants, Others)
8.2.5.1. Orthopedic Implants
8.2.5.2. ENT Implants
8.2.5.3. Breast Implants
8.2.5.4. Dental Implants
8.2.5.5. Cardiology related implants
8.2.5.6. Others
8.2.6. Others
8.2.6.1. Definition
8.2.6.2. Market Estimation and Penetration, 2015-2019
8.2.6.3. Market Forecast, 2020 – 2028
8.2.6.4. Compound Annual Growth Rate (CAGR)
8.2.6.5. Regional Bifurcation
8.2.6.5.1. North America
8.2.6.5.1.1. Market Estimation, 2015-2019
8.2.6.5.1.2. Market Forecast, 2020 – 2028
8.2.6.5.2. Europe
8.2.6.5.2.1. Market Estimation, 2015-2019
8.2.6.5.2.2. Market Forecast, 2020 – 2028
8.2.6.5.3. Asia Pacific
8.2.6.5.3.1. Market Estimation, 2015-2019
8.2.6.5.3.2. Market Forecast, 2020 – 2028
8.2.6.5.4. Middle East and Africa
8.2.6.5.4.1. Market Estimation, 2015-2019
8.2.6.5.4.2. Market Forecast, 2020 – 2028
8.2.6.5.5. Latin America
8.2.6.5.5.1. Market Estimation, 2015-2019
8.2.6.5.5.2. Market Forecast, 2020 – 2028
8.3. Key Segment for Channeling Investments
8.3.1. By Type
9. North America Medical
Device Market Analysis and Forecasts, 2020 - 2028
9.1. Overview
9.1.1. North America Medical Device
Market Revenue (US$ Mn)
9.2. North America Medical
Device Market Revenue (US$ Mn) and Forecasts, By Type
9.2.1. Electronics and
Equipment
9.2.1.1. Diagnostic Imaging
9.2.1.1.1. X-Ray Equipment
9.2.1.1.2. Ultrasound Systems
9.2.1.1.3. Computed Tomography (CT) Scanners
9.2.1.1.4. Magnetic Resonance Imaging Systems
9.2.1.1.5. Others
9.2.1.2. Patient Monitoring
9.2.1.2.1. Cardiac Monitoring Devices
9.2.1.2.2. Respiratory Monitoring Devices
9.2.1.2.3. Others
9.2.1.3. Diabetic Care Devices
9.2.1.3.1. Insulin Pens
9.2.1.3.2. Blood Glucose Meters
9.2.1.3.3. Blood Glucose Test Strips
9.2.1.3.4. Continuous Glucose Monitoring Device
9.2.1.3.5. Others
9.2.1.4. Orthopedic Devices
9.2.1.4.1. Prosthetics
9.2.1.4.2. Braces
9.2.1.4.3. Fixation Devices
9.2.1.4.4. Others
9.2.1.5. Dental Diagnostics and Operative Devices
9.2.1.6. Others
9.2.2. Disposables and Consumables – Highest CAGR
9.2.2.1. Wound Management Products
9.2.2.2. Drug Delivery Products
9.2.2.3. Diagnostic and
Laboratory Disposables
9.2.2.4. Dialysis Disposables
9.2.2.5. Incontinence Products
9.2.2.6. Respiratory Supplies
9.2.2.7. Sterilization Supplies
9.2.2.8. Non-woven Disposables
9.2.2.9. Disposable Masks
9.2.2.10.
Disposable Eye Gear
9.2.2.11.
Disposable Gloves
9.2.2.12.
Hand Sanitizers
9.2.2.12.1. Gel Sanitizers
9.2.2.12.2. Foam Sanitizers
9.2.2.12.3. Liquid Sanitizers
9.2.2.12.4. Other Sanitizers
9.2.2.13.
Others
9.2.3. Surgical Instruments
9.2.3.1. Surgical Sutures and Staplers
9.2.3.2. Handheld Surgical Devices
9.2.3.2.1. Forceps and Spatulas
9.2.3.2.2. Retractors
9.2.3.2.3. Dilators
9.2.3.2.4. Graspers
9.2.3.2.5. Auxiliary Instruments
9.2.3.2.6. Cutter Instruments
9.2.3.2.7. Others
9.2.3.3. Electrosurgical Devices
9.2.3.4. Others
9.2.4. IVD Reagents
9.2.4.1. Antibodies
9.2.4.1.1. Monoclonal
9.2.4.1.2. Polyclonal
9.2.4.2. Oligonucleotides
9.2.4.3. Nucleic Acid Probes
9.2.4.4. Purified Proteins or Peptides
9.2.4.5. Other Reagents
9.2.5. Implants
9.2.5.1. Orthopedic Implants
9.2.5.2. ENT Implants
9.2.5.3. Breast Implants
9.2.5.4. Dental Implants
9.2.5.5. Cardiology related implants
9.2.5.6. Others
9.2.6. Others
9.3. North America Medical
Device Market Revenue (US$ Mn) and Forecasts, By Country
9.3.1. U.S
9.3.1.1. U.S Medical Device
Market Revenue (US$ Mn) and Forecasts, By Type
9.3.1.1.1. Electronics and
Equipment
9.3.1.1.1.1. Diagnostic Imaging
9.3.1.1.1.1.1. X-Ray Equipment
9.3.1.1.1.1.2. Ultrasound Systems
9.3.1.1.1.1.3. Computed Tomography (CT) Scanners
9.3.1.1.1.1.4. Magnetic Resonance Imaging Systems
9.3.1.1.1.1.5. Others
9.3.1.1.1.2. Patient Monitoring
9.3.1.1.1.2.1. Cardiac Monitoring Devices
9.3.1.1.1.2.2. Respiratory Monitoring Devices
9.3.1.1.1.2.3. Others
9.3.1.1.1.3. Diabetic Care Devices
9.3.1.1.1.3.1. Insulin Pens
9.3.1.1.1.3.2. Blood Glucose Meters
9.3.1.1.1.3.3. Blood Glucose Test Strips
9.3.1.1.1.3.4. Continuous Glucose Monitoring Device
9.3.1.1.1.3.5. Others
9.3.1.1.1.4. Orthopedic Devices
9.3.1.1.1.4.1. Prosthetics
9.3.1.1.1.4.2. Braces
9.3.1.1.1.4.3. Fixation Devices
9.3.1.1.1.4.4. Others
9.3.1.1.1.5. Dental Diagnostics and Operative Devices
9.3.1.1.1.6. Others
9.3.1.1.2. Disposables and Consumables – Highest CAGR
9.3.1.1.2.1. Wound Management Products
9.3.1.1.2.2. Drug Delivery Products
9.3.1.1.2.3. Diagnostic and
Laboratory Disposables
9.3.1.1.2.4. Dialysis Disposables
9.3.1.1.2.5. Incontinence Products
9.3.1.1.2.6. Respiratory Supplies
9.3.1.1.2.7. Sterilization Supplies
9.3.1.1.2.8. Non-woven Disposables
9.3.1.1.2.9. Disposable Masks
9.3.1.1.2.10. Disposable Eye Gear
9.3.1.1.2.11. Disposable Gloves
9.3.1.1.2.12. Hand Sanitizers
9.3.1.1.2.12.1. Gel Sanitizers
9.3.1.1.2.12.2. Foam Sanitizers
9.3.1.1.2.12.3. Liquid Sanitizers
9.3.1.1.2.12.4. Other Sanitizers
9.3.1.1.2.13. Others
9.3.1.1.3. Surgical Instruments
9.3.1.1.3.1. Surgical Sutures and Staplers
9.3.1.1.3.2. Handheld Surgical Devices
9.3.1.1.3.2.1. Forceps and Spatulas
9.3.1.1.3.2.2. Retractors
9.3.1.1.3.2.3. Dilators
9.3.1.1.3.2.4. Graspers
9.3.1.1.3.2.5. Auxiliary Instruments
9.3.1.1.3.2.6. Cutter Instruments
9.3.1.1.3.2.7. Others
9.3.1.1.3.3. Electrosurgical Devices
9.3.1.1.3.4. Others
9.3.1.1.4. IVD Reagents
9.3.1.1.4.1. Antibodies
9.3.1.1.4.1.1. Monoclonal
9.3.1.1.4.1.2. Polyclonal
9.3.1.1.4.2. Oligonucleotides
9.3.1.1.4.3. Nucleic Acid Probes
9.3.1.1.4.4. Purified Proteins or Peptides
9.3.1.1.4.5. Other Reagents
9.3.1.1.5. Implants
9.3.1.1.5.1. Orthopedic Implants
9.3.1.1.5.2. ENT Implants
9.3.1.1.5.3. Breast Implants
9.3.1.1.5.4. Dental Implants
9.3.1.1.5.5. Cardiology related implants
9.3.1.1.5.6. Others
9.3.1.1.6. Others
9.3.2. Canada
9.3.2.1. Canada Medical Device
Market Revenue (US$ Mn) and Forecasts, By Type
9.3.2.1.1. Electronics and
Equipment
9.3.2.1.1.1. Diagnostic Imaging
9.3.2.1.1.1.1. X-Ray Equipment
9.3.2.1.1.1.2. Ultrasound Systems
9.3.2.1.1.1.3. Computed Tomography (CT) Scanners
9.3.2.1.1.1.4. Magnetic Resonance Imaging Systems
9.3.2.1.1.1.5. Others
9.3.2.1.1.2. Patient Monitoring
9.3.2.1.1.2.1. Cardiac Monitoring Devices
9.3.2.1.1.2.2. Respiratory Monitoring Devices
9.3.2.1.1.2.3. Others
9.3.2.1.1.3. Diabetic Care Devices
9.3.2.1.1.3.1. Insulin Pens
9.3.2.1.1.3.2. Blood Glucose Meters
9.3.2.1.1.3.3. Blood Glucose Test Strips
9.3.2.1.1.3.4. Continuous Glucose Monitoring Device
9.3.2.1.1.3.5. Others
9.3.2.1.1.4. Orthopedic Devices
9.3.2.1.1.4.1. Prosthetics
9.3.2.1.1.4.2. Braces
9.3.2.1.1.4.3. Fixation Devices
9.3.2.1.1.4.4. Others
9.3.2.1.1.5. Dental Diagnostics and Operative Devices
9.3.2.1.1.6. Others
9.3.2.1.2. Disposables and Consumables – Highest CAGR
9.3.2.1.2.1. Wound Management Products
9.3.2.1.2.2. Drug Delivery Products
9.3.2.1.2.3. Diagnostic and
Laboratory Disposables
9.3.2.1.2.4. Dialysis Disposables
9.3.2.1.2.5. Incontinence Products
9.3.2.1.2.6. Respiratory Supplies
9.3.2.1.2.7. Sterilization Supplies
9.3.2.1.2.8. Non-woven Disposables
9.3.2.1.2.9. Disposable Masks
9.3.2.1.2.10. Disposable Eye Gear
9.3.2.1.2.11. Disposable Gloves
9.3.2.1.2.12. Hand Sanitizers
9.3.2.1.2.12.1. Gel Sanitizers
9.3.2.1.2.12.2. Foam Sanitizers
9.3.2.1.2.12.3. Liquid Sanitizers
9.3.2.1.2.12.4. Other Sanitizers
9.3.2.1.2.13. Others
9.3.2.1.3. Surgical Instruments
9.3.2.1.3.1. Surgical Sutures and Staplers
9.3.2.1.3.2. Handheld Surgical Devices
9.3.2.1.3.2.1. Forceps and Spatulas
9.3.2.1.3.2.2. Retractors
9.3.2.1.3.2.3. Dilators
9.3.2.1.3.2.4. Graspers
9.3.2.1.3.2.5. Auxiliary Instruments
9.3.2.1.3.2.6. Cutter Instruments
9.3.2.1.3.2.7. Others
9.3.2.1.3.3. Electrosurgical Devices
9.3.2.1.3.4. Others
9.3.2.1.4. IVD Reagents
9.3.2.1.4.1. Antibodies
9.3.2.1.4.1.1. Monoclonal
9.3.2.1.4.1.2. Polyclonal
9.3.2.1.4.2. Oligonucleotides
9.3.2.1.4.3. Nucleic Acid Probes
9.3.2.1.4.4. Purified Proteins or Peptides
9.3.2.1.4.5. Other Reagents
9.3.2.1.5. Implants
9.3.2.1.5.1. Orthopedic Implants
9.3.2.1.5.2. ENT Implants
9.3.2.1.5.3. Breast Implants
9.3.2.1.5.4. Dental Implants
9.3.2.1.5.5. Cardiology related implants
9.3.2.1.5.6. Others
9.3.2.1.6. Others
9.3.3. Mexico
9.3.3.1. Mexico Medical Device
Market Revenue (US$ Mn) and Forecasts, By Type
9.3.3.1.1. Electronics and
Equipment
9.3.3.1.1.1. Diagnostic Imaging
9.3.3.1.1.1.1. X-Ray Equipment
9.3.3.1.1.1.2. Ultrasound Systems
9.3.3.1.1.1.3. Computed Tomography (CT) Scanners
9.3.3.1.1.1.4. Magnetic Resonance Imaging Systems
9.3.3.1.1.1.5. Others
9.3.3.1.1.2. Patient Monitoring
9.3.3.1.1.2.1. Cardiac Monitoring Devices
9.3.3.1.1.2.2. Respiratory Monitoring Devices
9.3.3.1.1.2.3. Others
9.3.3.1.1.3. Diabetic Care Devices
9.3.3.1.1.3.1. Insulin Pens
9.3.3.1.1.3.2. Blood Glucose Meters
9.3.3.1.1.3.3. Blood Glucose Test Strips
9.3.3.1.1.3.4. Continuous Glucose Monitoring Device
9.3.3.1.1.3.5. Others
9.3.3.1.1.4. Orthopedic Devices
9.3.3.1.1.4.1. Prosthetics
9.3.3.1.1.4.2. Braces
9.3.3.1.1.4.3. Fixation Devices
9.3.3.1.1.4.4. Others
9.3.3.1.1.5. Dental Diagnostics and Operative Devices
9.3.3.1.1.6. Others
9.3.3.1.2. Disposables and Consumables – Highest CAGR
9.3.3.1.2.1. Wound Management Products
9.3.3.1.2.2. Drug Delivery Products
9.3.3.1.2.3. Diagnostic and
Laboratory Disposables
9.3.3.1.2.4. Dialysis Disposables
9.3.3.1.2.5. Incontinence Products
9.3.3.1.2.6. Respiratory Supplies
9.3.3.1.2.7. Sterilization Supplies
9.3.3.1.2.8. Non-woven Disposables
9.3.3.1.2.9. Disposable Masks
9.3.3.1.2.10. Disposable Eye Gear
9.3.3.1.2.11. Disposable Gloves
9.3.3.1.2.12. Hand Sanitizers
9.3.3.1.2.12.1. Gel Sanitizers
9.3.3.1.2.12.2. Foam Sanitizers
9.3.3.1.2.12.3. Liquid Sanitizers
9.3.3.1.2.12.4. Other Sanitizers
9.3.3.1.2.13. Others
9.3.3.1.3. Surgical Instruments
9.3.3.1.3.1. Surgical Sutures and Staplers
9.3.3.1.3.2. Handheld Surgical Devices
9.3.3.1.3.2.1. Forceps and Spatulas
9.3.3.1.3.2.2. Retractors
9.3.3.1.3.2.3. Dilators
9.3.3.1.3.2.4. Graspers
9.3.3.1.3.2.5. Auxiliary Instruments
9.3.3.1.3.2.6. Cutter Instruments
9.3.3.1.3.2.7. Others
9.3.3.1.3.3. Electrosurgical Devices
9.3.3.1.3.4. Others
9.3.3.1.4. IVD Reagents
9.3.3.1.4.1. Antibodies
9.3.3.1.4.1.1. Monoclonal
9.3.3.1.4.1.2. Polyclonal
9.3.3.1.4.2. Oligonucleotides
9.3.3.1.4.3. Nucleic Acid Probes
9.3.3.1.4.4. Purified Proteins or Peptides
9.3.3.1.4.5. Other Reagents
9.3.3.1.5. Implants
9.3.3.1.5.1. Orthopedic Implants
9.3.3.1.5.2. ENT Implants
9.3.3.1.5.3. Breast Implants
9.3.3.1.5.4. Dental Implants
9.3.3.1.5.5. Cardiology related implants
9.3.3.1.5.6. Others
9.3.3.1.6. Others
9.3.4. Rest of North America
9.3.4.1. Rest of North America Medical
Device Market Revenue (US$ Mn) and Forecasts, By Type
9.3.4.1.1. Electronics and
Equipment
9.3.4.1.1.1. Diagnostic Imaging
9.3.4.1.1.1.1. X-Ray Equipment
9.3.4.1.1.1.2. Ultrasound Systems
9.3.4.1.1.1.3. Computed Tomography (CT) Scanners
9.3.4.1.1.1.4. Magnetic Resonance Imaging Systems
9.3.4.1.1.1.5. Others
9.3.4.1.1.2. Patient Monitoring
9.3.4.1.1.2.1. Cardiac Monitoring Devices
9.3.4.1.1.2.2. Respiratory Monitoring Devices
9.3.4.1.1.2.3. Others
9.3.4.1.1.3. Diabetic Care Devices
9.3.4.1.1.3.1. Insulin Pens
9.3.4.1.1.3.2. Blood Glucose Meters
9.3.4.1.1.3.3. Blood Glucose Test Strips
9.3.4.1.1.3.4. Continuous Glucose Monitoring Device
9.3.4.1.1.3.5. Others
9.3.4.1.1.4. Orthopedic Devices
9.3.4.1.1.4.1. Prosthetics
9.3.4.1.1.4.2. Braces
9.3.4.1.1.4.3. Fixation Devices
9.3.4.1.1.4.4. Others
9.3.4.1.1.5. Dental Diagnostics and Operative Devices
9.3.4.1.1.6. Others
9.3.4.1.2. Disposables and Consumables – Highest CAGR
9.3.4.1.2.1. Wound Management Products
9.3.4.1.2.2. Drug Delivery Products
9.3.4.1.2.3. Diagnostic and
Laboratory Disposables
9.3.4.1.2.4. Dialysis Disposables
9.3.4.1.2.5. Incontinence Products
9.3.4.1.2.6. Respiratory Supplies
9.3.4.1.2.7. Sterilization Supplies
9.3.4.1.2.8. Non-woven Disposables
9.3.4.1.2.9. Disposable Masks
9.3.4.1.2.10. Disposable Eye Gear
9.3.4.1.2.11. Disposable Gloves
9.3.4.1.2.12. Hand Sanitizers
9.3.4.1.2.12.1. Gel Sanitizers
9.3.4.1.2.12.2. Foam Sanitizers
9.3.4.1.2.12.3. Liquid Sanitizers
9.3.4.1.2.12.4. Other Sanitizers
9.3.4.1.2.13. Others
9.3.4.1.3. Surgical Instruments
9.3.4.1.3.1. Surgical Sutures and Staplers
9.3.4.1.3.2. Handheld Surgical Devices
9.3.4.1.3.2.1. Forceps and Spatulas
9.3.4.1.3.2.2. Retractors
9.3.4.1.3.2.3. Dilators
9.3.4.1.3.2.4. Graspers
9.3.4.1.3.2.5. Auxiliary Instruments
9.3.4.1.3.2.6. Cutter Instruments
9.3.4.1.3.2.7. Others
9.3.4.1.3.3. Electrosurgical Devices
9.3.4.1.3.4. Others
9.3.4.1.4. IVD Reagents
9.3.4.1.4.1. Antibodies
9.3.4.1.4.1.1. Monoclonal
9.3.4.1.4.1.2. Polyclonal
9.3.4.1.4.2. Oligonucleotides
9.3.4.1.4.3. Nucleic Acid Probes
9.3.4.1.4.4. Purified Proteins or Peptides
9.3.4.1.4.5. Other Reagents
9.3.4.1.5. Implants
9.3.4.1.5.1. Orthopedic Implants
9.3.4.1.5.2. ENT Implants
9.3.4.1.5.3. Breast Implants
9.3.4.1.5.4. Dental Implants
9.3.4.1.5.5. Cardiology related implants
9.3.4.1.5.6. Others
9.3.4.1.6. Others
9.4. Key Segment for Channeling Investments
9.4.1. By Country
9.4.2. By Type
10. Europe Medical Device
Market Analysis and Forecasts, 2020 - 2028
10.1.
Overview
10.1.1. Europe Medical Device
Market Revenue (US$ Mn)
10.2.
Europe Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
10.2.1. Electronics and
Equipment
10.2.1.1.
Diagnostic Imaging
10.2.1.1.1. X-Ray Equipment
10.2.1.1.2. Ultrasound Systems
10.2.1.1.3. Computed Tomography (CT) Scanners
10.2.1.1.4. Magnetic Resonance Imaging Systems
10.2.1.1.5. Others
10.2.1.2.
Patient Monitoring
10.2.1.2.1. Cardiac Monitoring Devices
10.2.1.2.2. Respiratory Monitoring Devices
10.2.1.2.3. Others
10.2.1.3.
Diabetic Care Devices
10.2.1.3.1. Insulin Pens
10.2.1.3.2. Blood Glucose Meters
10.2.1.3.3. Blood Glucose Test Strips
10.2.1.3.4. Continuous Glucose Monitoring Device
10.2.1.3.5. Others
10.2.1.4.
Orthopedic Devices
10.2.1.4.1. Prosthetics
10.2.1.4.2. Braces
10.2.1.4.3. Fixation Devices
10.2.1.4.4. Others
10.2.1.5.
Dental Diagnostics and Operative
Devices
10.2.1.6.
Others
10.2.2. Disposables and Consumables – Highest CAGR
10.2.2.1.
Wound Management Products
10.2.2.2.
Drug Delivery Products
10.2.2.3.
Diagnostic and Laboratory Disposables
10.2.2.4.
Dialysis Disposables
10.2.2.5.
Incontinence Products
10.2.2.6.
Respiratory Supplies
10.2.2.7.
Sterilization Supplies
10.2.2.8.
Non-woven Disposables
10.2.2.9.
Disposable Masks
10.2.2.10. Disposable Eye Gear
10.2.2.11. Disposable Gloves
10.2.2.12. Hand Sanitizers
10.2.2.12.1. Gel Sanitizers
10.2.2.12.2. Foam Sanitizers
10.2.2.12.3. Liquid Sanitizers
10.2.2.12.4. Other Sanitizers
10.2.2.13. Others
10.2.3. Surgical Instruments
10.2.3.1.
Surgical Sutures and Staplers
10.2.3.2.
Handheld Surgical Devices
10.2.3.2.1. Forceps and Spatulas
10.2.3.2.2. Retractors
10.2.3.2.3. Dilators
10.2.3.2.4. Graspers
10.2.3.2.5. Auxiliary Instruments
10.2.3.2.6. Cutter Instruments
10.2.3.2.7. Others
10.2.3.3.
Electrosurgical Devices
10.2.3.4.
Others
10.2.4. IVD Reagents
10.2.4.1.
Antibodies
10.2.4.1.1. Monoclonal
10.2.4.1.2. Polyclonal
10.2.4.2.
Oligonucleotides
10.2.4.3.
Nucleic Acid Probes
10.2.4.4.
Purified Proteins or Peptides
10.2.4.5.
Other Reagents
10.2.5. Implants
10.2.5.1.
Orthopedic Implants
10.2.5.2.
ENT Implants
10.2.5.3.
Breast Implants
10.2.5.4.
Dental Implants
10.2.5.5.
Cardiology related implants
10.2.5.6.
Others
10.2.6. Others
10.3.
Europe Medical Device Market Revenue (US$ Mn) and Forecasts,
By Country
10.3.1. France
10.3.1.1.
France Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
10.3.1.1.1. Electronics and
Equipment
10.3.1.1.1.1. Diagnostic Imaging
10.3.1.1.1.1.1. X-Ray Equipment
10.3.1.1.1.1.2. Ultrasound Systems
10.3.1.1.1.1.3. Computed Tomography (CT) Scanners
10.3.1.1.1.1.4. Magnetic Resonance Imaging Systems
10.3.1.1.1.1.5. Others
10.3.1.1.1.2. Patient Monitoring
10.3.1.1.1.2.1. Cardiac Monitoring Devices
10.3.1.1.1.2.2. Respiratory Monitoring Devices
10.3.1.1.1.2.3. Others
10.3.1.1.1.3. Diabetic Care Devices
10.3.1.1.1.3.1. Insulin Pens
10.3.1.1.1.3.2. Blood Glucose Meters
10.3.1.1.1.3.3. Blood Glucose Test Strips
10.3.1.1.1.3.4. Continuous Glucose Monitoring Device
10.3.1.1.1.3.5. Others
10.3.1.1.1.4. Orthopedic Devices
10.3.1.1.1.4.1. Prosthetics
10.3.1.1.1.4.2. Braces
10.3.1.1.1.4.3. Fixation Devices
10.3.1.1.1.4.4. Others
10.3.1.1.1.5. Dental Diagnostics and Operative Devices
10.3.1.1.1.6. Others
10.3.1.1.2. Disposables and Consumables – Highest CAGR
10.3.1.1.2.1. Wound Management Products
10.3.1.1.2.2. Drug Delivery Products
10.3.1.1.2.3. Diagnostic and
Laboratory Disposables
10.3.1.1.2.4. Dialysis Disposables
10.3.1.1.2.5. Incontinence Products
10.3.1.1.2.6. Respiratory Supplies
10.3.1.1.2.7. Sterilization Supplies
10.3.1.1.2.8. Non-woven Disposables
10.3.1.1.2.9. Disposable Masks
10.3.1.1.2.10. Disposable Eye Gear
10.3.1.1.2.11. Disposable Gloves
10.3.1.1.2.12. Hand Sanitizers
10.3.1.1.2.12.1. Gel Sanitizers
10.3.1.1.2.12.2. Foam Sanitizers
10.3.1.1.2.12.3. Liquid Sanitizers
10.3.1.1.2.12.4. Other Sanitizers
10.3.1.1.2.13. Others
10.3.1.1.3. Surgical Instruments
10.3.1.1.3.1. Surgical Sutures and Staplers
10.3.1.1.3.2. Handheld Surgical Devices
10.3.1.1.3.2.1. Forceps and Spatulas
10.3.1.1.3.2.2. Retractors
10.3.1.1.3.2.3. Dilators
10.3.1.1.3.2.4. Graspers
10.3.1.1.3.2.5. Auxiliary Instruments
10.3.1.1.3.2.6. Cutter Instruments
10.3.1.1.3.2.7. Others
10.3.1.1.3.3. Electrosurgical Devices
10.3.1.1.3.4. Others
10.3.1.1.4. IVD Reagents
10.3.1.1.4.1. Antibodies
10.3.1.1.4.1.1. Monoclonal
10.3.1.1.4.1.2. Polyclonal
10.3.1.1.4.2. Oligonucleotides
10.3.1.1.4.3. Nucleic Acid Probes
10.3.1.1.4.4. Purified Proteins or Peptides
10.3.1.1.4.5. Other Reagents
10.3.1.1.5. Implants
10.3.1.1.5.1. Orthopedic Implants
10.3.1.1.5.2. ENT Implants
10.3.1.1.5.3. Breast Implants
10.3.1.1.5.4. Dental Implants
10.3.1.1.5.5. Cardiology related implants
10.3.1.1.5.6. Others
10.3.1.1.6. Others
10.3.2. The UK
10.3.2.1.
The UK Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
10.3.2.1.1. Electronics and
Equipment
10.3.2.1.1.1. Diagnostic Imaging
10.3.2.1.1.1.1. X-Ray Equipment
10.3.2.1.1.1.2. Ultrasound Systems
10.3.2.1.1.1.3. Computed Tomography (CT) Scanners
10.3.2.1.1.1.4. Magnetic Resonance Imaging Systems
10.3.2.1.1.1.5. Others
10.3.2.1.1.2. Patient Monitoring
10.3.2.1.1.2.1. Cardiac Monitoring Devices
10.3.2.1.1.2.2. Respiratory Monitoring Devices
10.3.2.1.1.2.3. Others
10.3.2.1.1.3. Diabetic Care Devices
10.3.2.1.1.3.1. Insulin Pens
10.3.2.1.1.3.2. Blood Glucose Meters
10.3.2.1.1.3.3. Blood Glucose Test Strips
10.3.2.1.1.3.4. Continuous Glucose Monitoring Device
10.3.2.1.1.3.5. Others
10.3.2.1.1.4. Orthopedic Devices
10.3.2.1.1.4.1. Prosthetics
10.3.2.1.1.4.2. Braces
10.3.2.1.1.4.3. Fixation Devices
10.3.2.1.1.4.4. Others
10.3.2.1.1.5. Dental Diagnostics and Operative Devices
10.3.2.1.1.6. Others
10.3.2.1.2. Disposables and Consumables – Highest CAGR
10.3.2.1.2.1. Wound Management Products
10.3.2.1.2.2. Drug Delivery Products
10.3.2.1.2.3. Diagnostic and
Laboratory Disposables
10.3.2.1.2.4. Dialysis Disposables
10.3.2.1.2.5. Incontinence Products
10.3.2.1.2.6. Respiratory Supplies
10.3.2.1.2.7. Sterilization Supplies
10.3.2.1.2.8. Non-woven Disposables
10.3.2.1.2.9. Disposable Masks
10.3.2.1.2.10. Disposable Eye Gear
10.3.2.1.2.11. Disposable Gloves
10.3.2.1.2.12. Hand Sanitizers
10.3.2.1.2.12.1. Gel Sanitizers
10.3.2.1.2.12.2. Foam Sanitizers
10.3.2.1.2.12.3. Liquid Sanitizers
10.3.2.1.2.12.4. Other Sanitizers
10.3.2.1.2.13. Others
10.3.2.1.3. Surgical Instruments
10.3.2.1.3.1. Surgical Sutures and Staplers
10.3.2.1.3.2. Handheld Surgical Devices
10.3.2.1.3.2.1. Forceps and Spatulas
10.3.2.1.3.2.2. Retractors
10.3.2.1.3.2.3. Dilators
10.3.2.1.3.2.4. Graspers
10.3.2.1.3.2.5. Auxiliary Instruments
10.3.2.1.3.2.6. Cutter Instruments
10.3.2.1.3.2.7. Others
10.3.2.1.3.3. Electrosurgical Devices
10.3.2.1.3.4. Others
10.3.2.1.4. IVD Reagents
10.3.2.1.4.1. Antibodies
10.3.2.1.4.1.1. Monoclonal
10.3.2.1.4.1.2. Polyclonal
10.3.2.1.4.2. Oligonucleotides
10.3.2.1.4.3. Nucleic Acid Probes
10.3.2.1.4.4. Purified Proteins or Peptides
10.3.2.1.4.5. Other Reagents
10.3.2.1.5. Implants
10.3.2.1.5.1. Orthopedic Implants
10.3.2.1.5.2. ENT Implants
10.3.2.1.5.3. Breast Implants
10.3.2.1.5.4. Dental Implants
10.3.2.1.5.5. Cardiology related implants
10.3.2.1.5.6. Others
10.3.2.1.6. Others
10.3.3. Spain
10.3.3.1.
Spain Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
10.3.3.1.1. Electronics and
Equipment
10.3.3.1.1.1. Diagnostic Imaging
10.3.3.1.1.1.1. X-Ray Equipment
10.3.3.1.1.1.2. Ultrasound Systems
10.3.3.1.1.1.3. Computed Tomography (CT) Scanners
10.3.3.1.1.1.4. Magnetic Resonance Imaging Systems
10.3.3.1.1.1.5. Others
10.3.3.1.1.2. Patient Monitoring
10.3.3.1.1.2.1. Cardiac Monitoring Devices
10.3.3.1.1.2.2. Respiratory Monitoring Devices
10.3.3.1.1.2.3. Others
10.3.3.1.1.3. Diabetic Care Devices
10.3.3.1.1.3.1. Insulin Pens
10.3.3.1.1.3.2. Blood Glucose Meters
10.3.3.1.1.3.3. Blood Glucose Test Strips
10.3.3.1.1.3.4. Continuous Glucose Monitoring Device
10.3.3.1.1.3.5. Others
10.3.3.1.1.4. Orthopedic Devices
10.3.3.1.1.4.1. Prosthetics
10.3.3.1.1.4.2. Braces
10.3.3.1.1.4.3. Fixation Devices
10.3.3.1.1.4.4. Others
10.3.3.1.1.5. Dental Diagnostics and Operative Devices
10.3.3.1.1.6. Others
10.3.3.1.2. Disposables and Consumables – Highest CAGR
10.3.3.1.2.1. Wound Management Products
10.3.3.1.2.2. Drug Delivery Products
10.3.3.1.2.3. Diagnostic and
Laboratory Disposables
10.3.3.1.2.4. Dialysis Disposables
10.3.3.1.2.5. Incontinence Products
10.3.3.1.2.6. Respiratory Supplies
10.3.3.1.2.7. Sterilization Supplies
10.3.3.1.2.8. Non-woven Disposables
10.3.3.1.2.9. Disposable Masks
10.3.3.1.2.10. Disposable Eye Gear
10.3.3.1.2.11. Disposable Gloves
10.3.3.1.2.12. Hand Sanitizers
10.3.3.1.2.12.1. Gel Sanitizers
10.3.3.1.2.12.2. Foam Sanitizers
10.3.3.1.2.12.3. Liquid Sanitizers
10.3.3.1.2.12.4. Other Sanitizers
10.3.3.1.2.13. Others
10.3.3.1.3. Surgical Instruments
10.3.3.1.3.1. Surgical Sutures and Staplers
10.3.3.1.3.2. Handheld Surgical Devices
10.3.3.1.3.2.1. Forceps and Spatulas
10.3.3.1.3.2.2. Retractors
10.3.3.1.3.2.3. Dilators
10.3.3.1.3.2.4. Graspers
10.3.3.1.3.2.5. Auxiliary Instruments
10.3.3.1.3.2.6. Cutter Instruments
10.3.3.1.3.2.7. Others
10.3.3.1.3.3. Electrosurgical Devices
10.3.3.1.3.4. Others
10.3.3.1.4. IVD Reagents
10.3.3.1.4.1. Antibodies
10.3.3.1.4.1.1. Monoclonal
10.3.3.1.4.1.2. Polyclonal
10.3.3.1.4.2. Oligonucleotides
10.3.3.1.4.3. Nucleic Acid Probes
10.3.3.1.4.4. Purified Proteins or Peptides
10.3.3.1.4.5. Other Reagents
10.3.3.1.5. Implants
10.3.3.1.5.1. Orthopedic Implants
10.3.3.1.5.2. ENT Implants
10.3.3.1.5.3. Breast Implants
10.3.3.1.5.4. Dental Implants
10.3.3.1.5.5. Cardiology related implants
10.3.3.1.5.6. Others
10.3.3.1.6. Others
10.3.4. Germany
10.3.4.1.
Germany Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
10.3.4.1.1. Electronics and
Equipment
10.3.4.1.1.1. Diagnostic Imaging
10.3.4.1.1.1.1. X-Ray Equipment
10.3.4.1.1.1.2. Ultrasound Systems
10.3.4.1.1.1.3. Computed Tomography (CT) Scanners
10.3.4.1.1.1.4. Magnetic Resonance Imaging Systems
10.3.4.1.1.1.5. Others
10.3.4.1.1.2. Patient Monitoring
10.3.4.1.1.2.1. Cardiac Monitoring Devices
10.3.4.1.1.2.2. Respiratory Monitoring Devices
10.3.4.1.1.2.3. Others
10.3.4.1.1.3. Diabetic Care Devices
10.3.4.1.1.3.1. Insulin Pens
10.3.4.1.1.3.2. Blood Glucose Meters
10.3.4.1.1.3.3. Blood Glucose Test Strips
10.3.4.1.1.3.4. Continuous Glucose Monitoring Device
10.3.4.1.1.3.5. Others
10.3.4.1.1.4. Orthopedic Devices
10.3.4.1.1.4.1. Prosthetics
10.3.4.1.1.4.2. Braces
10.3.4.1.1.4.3. Fixation Devices
10.3.4.1.1.4.4. Others
10.3.4.1.1.5. Dental Diagnostics and Operative Devices
10.3.4.1.1.6. Others
10.3.4.1.2. Disposables and Consumables – Highest CAGR
10.3.4.1.2.1. Wound Management Products
10.3.4.1.2.2. Drug Delivery Products
10.3.4.1.2.3. Diagnostic and
Laboratory Disposables
10.3.4.1.2.4. Dialysis Disposables
10.3.4.1.2.5. Incontinence Products
10.3.4.1.2.6. Respiratory Supplies
10.3.4.1.2.7. Sterilization Supplies
10.3.4.1.2.8. Non-woven Disposables
10.3.4.1.2.9. Disposable Masks
10.3.4.1.2.10. Disposable Eye Gear
10.3.4.1.2.11. Disposable Gloves
10.3.4.1.2.12. Hand Sanitizers
10.3.4.1.2.12.1. Gel Sanitizers
10.3.4.1.2.12.2. Foam Sanitizers
10.3.4.1.2.12.3. Liquid Sanitizers
10.3.4.1.2.12.4. Other Sanitizers
10.3.4.1.2.13. Others
10.3.4.1.3. Surgical Instruments
10.3.4.1.3.1. Surgical Sutures and Staplers
10.3.4.1.3.2. Handheld Surgical Devices
10.3.4.1.3.2.1. Forceps and Spatulas
10.3.4.1.3.2.2. Retractors
10.3.4.1.3.2.3. Dilators
10.3.4.1.3.2.4. Graspers
10.3.4.1.3.2.5. Auxiliary Instruments
10.3.4.1.3.2.6. Cutter Instruments
10.3.4.1.3.2.7. Others
10.3.4.1.3.3. Electrosurgical Devices
10.3.4.1.3.4. Others
10.3.4.1.4. IVD Reagents
10.3.4.1.4.1. Antibodies
10.3.4.1.4.1.1. Monoclonal
10.3.4.1.4.1.2. Polyclonal
10.3.4.1.4.2. Oligonucleotides
10.3.4.1.4.3. Nucleic Acid Probes
10.3.4.1.4.4. Purified Proteins or Peptides
10.3.4.1.4.5. Other Reagents
10.3.4.1.5. Implants
10.3.4.1.5.1. Orthopedic Implants
10.3.4.1.5.2. ENT Implants
10.3.4.1.5.3. Breast Implants
10.3.4.1.5.4. Dental Implants
10.3.4.1.5.5. Cardiology related implants
10.3.4.1.5.6. Others
10.3.4.1.6. Others
10.3.5. Italy
10.3.5.1.
Italy Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
10.3.5.1.1. Electronics and
Equipment
10.3.5.1.1.1. Diagnostic Imaging
10.3.5.1.1.1.1. X-Ray Equipment
10.3.5.1.1.1.2. Ultrasound Systems
10.3.5.1.1.1.3. Computed Tomography (CT) Scanners
10.3.5.1.1.1.4. Magnetic Resonance Imaging Systems
10.3.5.1.1.1.5. Others
10.3.5.1.1.2. Patient Monitoring
10.3.5.1.1.2.1. Cardiac Monitoring Devices
10.3.5.1.1.2.2. Respiratory Monitoring Devices
10.3.5.1.1.2.3. Others
10.3.5.1.1.3. Diabetic Care Devices
10.3.5.1.1.3.1. Insulin Pens
10.3.5.1.1.3.2. Blood Glucose Meters
10.3.5.1.1.3.3. Blood Glucose Test Strips
10.3.5.1.1.3.4. Continuous Glucose Monitoring Device
10.3.5.1.1.3.5. Others
10.3.5.1.1.4. Orthopedic Devices
10.3.5.1.1.4.1. Prosthetics
10.3.5.1.1.4.2. Braces
10.3.5.1.1.4.3. Fixation Devices
10.3.5.1.1.4.4. Others
10.3.5.1.1.5. Dental Diagnostics and Operative Devices
10.3.5.1.1.6. Others
10.3.5.1.2. Disposables and Consumables – Highest CAGR
10.3.5.1.2.1. Wound Management Products
10.3.5.1.2.2. Drug Delivery Products
10.3.5.1.2.3. Diagnostic and
Laboratory Disposables
10.3.5.1.2.4. Dialysis Disposables
10.3.5.1.2.5. Incontinence Products
10.3.5.1.2.6. Respiratory Supplies
10.3.5.1.2.7. Sterilization Supplies
10.3.5.1.2.8. Non-woven Disposables
10.3.5.1.2.9. Disposable Masks
10.3.5.1.2.10. Disposable Eye Gear
10.3.5.1.2.11. Disposable Gloves
10.3.5.1.2.12. Hand Sanitizers
10.3.5.1.2.12.1. Gel Sanitizers
10.3.5.1.2.12.2. Foam Sanitizers
10.3.5.1.2.12.3. Liquid Sanitizers
10.3.5.1.2.12.4. Other Sanitizers
10.3.5.1.2.13. Others
10.3.5.1.3. Surgical Instruments
10.3.5.1.3.1. Surgical Sutures and Staplers
10.3.5.1.3.2. Handheld Surgical Devices
10.3.5.1.3.2.1. Forceps and Spatulas
10.3.5.1.3.2.2. Retractors
10.3.5.1.3.2.3. Dilators
10.3.5.1.3.2.4. Graspers
10.3.5.1.3.2.5. Auxiliary Instruments
10.3.5.1.3.2.6. Cutter Instruments
10.3.5.1.3.2.7. Others
10.3.5.1.3.3. Electrosurgical Devices
10.3.5.1.3.4. Others
10.3.5.1.4. IVD Reagents
10.3.5.1.4.1. Antibodies
10.3.5.1.4.1.1. Monoclonal
10.3.5.1.4.1.2. Polyclonal
10.3.5.1.4.2. Oligonucleotides
10.3.5.1.4.3. Nucleic Acid Probes
10.3.5.1.4.4. Purified Proteins or Peptides
10.3.5.1.4.5. Other Reagents
10.3.5.1.5. Implants
10.3.5.1.5.1. Orthopedic Implants
10.3.5.1.5.2. ENT Implants
10.3.5.1.5.3. Breast Implants
10.3.5.1.5.4. Dental Implants
10.3.5.1.5.5. Cardiology related implants
10.3.5.1.5.6. Others
10.3.5.1.6. Others
10.3.6. Nordic Countries
10.3.6.1.
Nordic Countries Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
10.3.6.1.1. Electronics and
Equipment
10.3.6.1.1.1. Diagnostic Imaging
10.3.6.1.1.1.1. X-Ray Equipment
10.3.6.1.1.1.2. Ultrasound Systems
10.3.6.1.1.1.3. Computed Tomography (CT) Scanners
10.3.6.1.1.1.4. Magnetic Resonance Imaging Systems
10.3.6.1.1.1.5. Others
10.3.6.1.1.2. Patient Monitoring
10.3.6.1.1.2.1. Cardiac Monitoring Devices
10.3.6.1.1.2.2. Respiratory Monitoring Devices
10.3.6.1.1.2.3. Others
10.3.6.1.1.3. Diabetic Care Devices
10.3.6.1.1.3.1. Insulin Pens
10.3.6.1.1.3.2. Blood Glucose Meters
10.3.6.1.1.3.3. Blood Glucose Test Strips
10.3.6.1.1.3.4. Continuous Glucose Monitoring Device
10.3.6.1.1.3.5. Others
10.3.6.1.1.4. Orthopedic Devices
10.3.6.1.1.4.1. Prosthetics
10.3.6.1.1.4.2. Braces
10.3.6.1.1.4.3. Fixation Devices
10.3.6.1.1.4.4. Others
10.3.6.1.1.5. Dental Diagnostics and Operative Devices
10.3.6.1.1.6. Others
10.3.6.1.2. Disposables and Consumables – Highest CAGR
10.3.6.1.2.1. Wound Management Products
10.3.6.1.2.2. Drug Delivery Products
10.3.6.1.2.3. Diagnostic and
Laboratory Disposables
10.3.6.1.2.4. Dialysis Disposables
10.3.6.1.2.5. Incontinence Products
10.3.6.1.2.6. Respiratory Supplies
10.3.6.1.2.7. Sterilization Supplies
10.3.6.1.2.8. Non-woven Disposables
10.3.6.1.2.9. Disposable Masks
10.3.6.1.2.10. Disposable Eye Gear
10.3.6.1.2.11. Disposable Gloves
10.3.6.1.2.12. Hand Sanitizers
10.3.6.1.2.12.1. Gel Sanitizers
10.3.6.1.2.12.2. Foam Sanitizers
10.3.6.1.2.12.3. Liquid Sanitizers
10.3.6.1.2.12.4. Other Sanitizers
10.3.6.1.2.13. Others
10.3.6.1.3. Surgical Instruments
10.3.6.1.3.1. Surgical Sutures and Staplers
10.3.6.1.3.2. Handheld Surgical Devices
10.3.6.1.3.2.1. Forceps and Spatulas
10.3.6.1.3.2.2. Retractors
10.3.6.1.3.2.3. Dilators
10.3.6.1.3.2.4. Graspers
10.3.6.1.3.2.5. Auxiliary Instruments
10.3.6.1.3.2.6. Cutter Instruments
10.3.6.1.3.2.7. Others
10.3.6.1.3.3. Electrosurgical Devices
10.3.6.1.3.4. Others
10.3.6.1.4. IVD Reagents
10.3.6.1.4.1. Antibodies
10.3.6.1.4.1.1. Monoclonal
10.3.6.1.4.1.2. Polyclonal
10.3.6.1.4.2. Oligonucleotides
10.3.6.1.4.3. Nucleic Acid Probes
10.3.6.1.4.4. Purified Proteins or Peptides
10.3.6.1.4.5. Other Reagents
10.3.6.1.5. Implants
10.3.6.1.5.1. Orthopedic Implants
10.3.6.1.5.2. ENT Implants
10.3.6.1.5.3. Breast Implants
10.3.6.1.5.4. Dental Implants
10.3.6.1.5.5. Cardiology related implants
10.3.6.1.5.6. Others
10.3.6.1.6. Others
10.3.6.2.
Nordic Countries Medical Device Market Revenue (US$ Mn) and Forecasts,
By Country
10.3.6.2.1. Denmark
10.3.6.2.2. Finland
10.3.6.2.3. Iceland
10.3.6.2.4. Sweden
10.3.6.2.5. Norway
10.3.7. Benelux Union
10.3.7.1.
Benelux Union Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
10.3.7.1.1. Electronics and
Equipment
10.3.7.1.1.1. Diagnostic Imaging
10.3.7.1.1.1.1. X-Ray Equipment
10.3.7.1.1.1.2. Ultrasound Systems
10.3.7.1.1.1.3. Computed Tomography (CT) Scanners
10.3.7.1.1.1.4. Magnetic Resonance Imaging Systems
10.3.7.1.1.1.5. Others
10.3.7.1.1.2. Patient Monitoring
10.3.7.1.1.2.1. Cardiac Monitoring Devices
10.3.7.1.1.2.2. Respiratory Monitoring Devices
10.3.7.1.1.2.3. Others
10.3.7.1.1.3. Diabetic Care Devices
10.3.7.1.1.3.1. Insulin Pens
10.3.7.1.1.3.2. Blood Glucose Meters
10.3.7.1.1.3.3. Blood Glucose Test Strips
10.3.7.1.1.3.4. Continuous Glucose Monitoring Device
10.3.7.1.1.3.5. Others
10.3.7.1.1.4. Orthopedic Devices
10.3.7.1.1.4.1. Prosthetics
10.3.7.1.1.4.2. Braces
10.3.7.1.1.4.3. Fixation Devices
10.3.7.1.1.4.4. Others
10.3.7.1.1.5. Dental Diagnostics and Operative Devices
10.3.7.1.1.6. Others
10.3.7.1.2. Disposables and Consumables – Highest CAGR
10.3.7.1.2.1. Wound Management Products
10.3.7.1.2.2. Drug Delivery Products
10.3.7.1.2.3. Diagnostic and
Laboratory Disposables
10.3.7.1.2.4. Dialysis Disposables
10.3.7.1.2.5. Incontinence Products
10.3.7.1.2.6. Respiratory Supplies
10.3.7.1.2.7. Sterilization Supplies
10.3.7.1.2.8. Non-woven Disposables
10.3.7.1.2.9. Disposable Masks
10.3.7.1.2.10. Disposable Eye Gear
10.3.7.1.2.11. Disposable Gloves
10.3.7.1.2.12. Hand Sanitizers
10.3.7.1.2.12.1. Gel Sanitizers
10.3.7.1.2.12.2. Foam Sanitizers
10.3.7.1.2.12.3. Liquid Sanitizers
10.3.7.1.2.12.4. Other Sanitizers
10.3.7.1.2.13. Others
10.3.7.1.3. Surgical Instruments
10.3.7.1.3.1. Surgical Sutures and Staplers
10.3.7.1.3.2. Handheld Surgical Devices
10.3.7.1.3.2.1. Forceps and Spatulas
10.3.7.1.3.2.2. Retractors
10.3.7.1.3.2.3. Dilators
10.3.7.1.3.2.4. Graspers
10.3.7.1.3.2.5. Auxiliary Instruments
10.3.7.1.3.2.6. Cutter Instruments
10.3.7.1.3.2.7. Others
10.3.7.1.3.3. Electrosurgical Devices
10.3.7.1.3.4. Others
10.3.7.1.4. IVD Reagents
10.3.7.1.4.1. Antibodies
10.3.7.1.4.1.1. Monoclonal
10.3.7.1.4.1.2. Polyclonal
10.3.7.1.4.2. Oligonucleotides
10.3.7.1.4.3. Nucleic Acid Probes
10.3.7.1.4.4. Purified Proteins or Peptides
10.3.7.1.4.5. Other Reagents
10.3.7.1.5. Implants
10.3.7.1.5.1. Orthopedic Implants
10.3.7.1.5.2. ENT Implants
10.3.7.1.5.3. Breast Implants
10.3.7.1.5.4. Dental Implants
10.3.7.1.5.5. Cardiology related implants
10.3.7.1.5.6. Others
10.3.7.1.6. Others
10.3.7.2.
Benelux Union Medical Device Market Revenue (US$ Mn) and Forecasts,
By Country
10.3.7.2.1. Belgium
10.3.7.2.2. The Netherlands
10.3.7.2.3. Luxembourg
10.3.8. Rest of Europe
10.3.8.1.
Rest of Europe Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
10.3.8.1.1. Electronics and
Equipment
10.3.8.1.1.1. Diagnostic Imaging
10.3.8.1.1.1.1. X-Ray Equipment
10.3.8.1.1.1.2. Ultrasound Systems
10.3.8.1.1.1.3. Computed Tomography (CT) Scanners
10.3.8.1.1.1.4. Magnetic Resonance Imaging Systems
10.3.8.1.1.1.5. Others
10.3.8.1.1.2. Patient Monitoring
10.3.8.1.1.2.1. Cardiac Monitoring Devices
10.3.8.1.1.2.2. Respiratory Monitoring Devices
10.3.8.1.1.2.3. Others
10.3.8.1.1.3. Diabetic Care Devices
10.3.8.1.1.3.1. Insulin Pens
10.3.8.1.1.3.2. Blood Glucose Meters
10.3.8.1.1.3.3. Blood Glucose Test Strips
10.3.8.1.1.3.4. Continuous Glucose Monitoring Device
10.3.8.1.1.3.5. Others
10.3.8.1.1.4. Orthopedic Devices
10.3.8.1.1.4.1. Prosthetics
10.3.8.1.1.4.2. Braces
10.3.8.1.1.4.3. Fixation Devices
10.3.8.1.1.4.4. Others
10.3.8.1.1.5. Dental Diagnostics and Operative Devices
10.3.8.1.1.6. Others
10.3.8.1.2. Disposables and Consumables – Highest CAGR
10.3.8.1.2.1. Wound Management Products
10.3.8.1.2.2. Drug Delivery Products
10.3.8.1.2.3. Diagnostic and
Laboratory Disposables
10.3.8.1.2.4. Dialysis Disposables
10.3.8.1.2.5. Incontinence Products
10.3.8.1.2.6. Respiratory Supplies
10.3.8.1.2.7. Sterilization Supplies
10.3.8.1.2.8. Non-woven Disposables
10.3.8.1.2.9. Disposable Masks
10.3.8.1.2.10. Disposable Eye Gear
10.3.8.1.2.11. Disposable Gloves
10.3.8.1.2.12. Hand Sanitizers
10.3.8.1.2.12.1. Gel Sanitizers
10.3.8.1.2.12.2. Foam Sanitizers
10.3.8.1.2.12.3. Liquid Sanitizers
10.3.8.1.2.12.4. Other Sanitizers
10.3.8.1.2.13. Others
10.3.8.1.3. Surgical Instruments
10.3.8.1.3.1. Surgical Sutures and Staplers
10.3.8.1.3.2. Handheld Surgical Devices
10.3.8.1.3.2.1. Forceps and Spatulas
10.3.8.1.3.2.2. Retractors
10.3.8.1.3.2.3. Dilators
10.3.8.1.3.2.4. Graspers
10.3.8.1.3.2.5. Auxiliary Instruments
10.3.8.1.3.2.6. Cutter Instruments
10.3.8.1.3.2.7. Others
10.3.8.1.3.3. Electrosurgical Devices
10.3.8.1.3.4. Others
10.3.8.1.4. IVD Reagents
10.3.8.1.4.1. Antibodies
10.3.8.1.4.1.1. Monoclonal
10.3.8.1.4.1.2. Polyclonal
10.3.8.1.4.2. Oligonucleotides
10.3.8.1.4.3. Nucleic Acid Probes
10.3.8.1.4.4. Purified Proteins or Peptides
10.3.8.1.4.5. Other Reagents
10.3.8.1.5. Implants
10.3.8.1.5.1. Orthopedic Implants
10.3.8.1.5.2. ENT Implants
10.3.8.1.5.3. Breast Implants
10.3.8.1.5.4. Dental Implants
10.3.8.1.5.5. Cardiology related implants
10.3.8.1.5.6. Others
10.3.8.1.6. Others
10.4.
Key Segment for Channeling
Investments
10.4.1. By Country
10.4.2. By Type
11. Asia Pacific Medical
Device Market Analysis and Forecasts, 2020 - 2028
11.1.
Overview
11.1.1. Asia Pacific Medical
Device Market Revenue (US$ Mn)
11.2.
Asia Pacific Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
11.2.1. Electronics and
Equipment
11.2.1.1.
Diagnostic Imaging
11.2.1.1.1. X-Ray Equipment
11.2.1.1.2. Ultrasound Systems
11.2.1.1.3. Computed Tomography (CT) Scanners
11.2.1.1.4. Magnetic Resonance Imaging Systems
11.2.1.1.5. Others
11.2.1.2.
Patient Monitoring
11.2.1.2.1. Cardiac Monitoring Devices
11.2.1.2.2. Respiratory Monitoring Devices
11.2.1.2.3. Others
11.2.1.3.
Diabetic Care Devices
11.2.1.3.1. Insulin Pens
11.2.1.3.2. Blood Glucose Meters
11.2.1.3.3. Blood Glucose Test Strips
11.2.1.3.4. Continuous Glucose Monitoring Device
11.2.1.3.5. Others
11.2.1.4.
Orthopedic Devices
11.2.1.4.1. Prosthetics
11.2.1.4.2. Braces
11.2.1.4.3. Fixation Devices
11.2.1.4.4. Others
11.2.1.5.
Dental Diagnostics and Operative
Devices
11.2.1.6.
Others
11.2.2. Disposables and Consumables – Highest CAGR
11.2.2.1.
Wound Management Products
11.2.2.2.
Drug Delivery Products
11.2.2.3.
Diagnostic and Laboratory Disposables
11.2.2.4.
Dialysis Disposables
11.2.2.5.
Incontinence Products
11.2.2.6.
Respiratory Supplies
11.2.2.7.
Sterilization Supplies
11.2.2.8.
Non-woven Disposables
11.2.2.9.
Disposable Masks
11.2.2.10. Disposable Eye Gear
11.2.2.11. Disposable Gloves
11.2.2.12. Hand Sanitizers
11.2.2.12.1. Gel Sanitizers
11.2.2.12.2. Foam Sanitizers
11.2.2.12.3. Liquid Sanitizers
11.2.2.12.4. Other Sanitizers
11.2.2.13. Others
11.2.3. Surgical Instruments
11.2.3.1.
Surgical Sutures and Staplers
11.2.3.2.
Handheld Surgical Devices
11.2.3.2.1. Forceps and Spatulas
11.2.3.2.2. Retractors
11.2.3.2.3. Dilators
11.2.3.2.4. Graspers
11.2.3.2.5. Auxiliary Instruments
11.2.3.2.6. Cutter Instruments
11.2.3.2.7. Others
11.2.3.3.
Electrosurgical Devices
11.2.3.4.
Others
11.2.4. IVD Reagents
11.2.4.1.
Antibodies
11.2.4.1.1. Monoclonal
11.2.4.1.2. Polyclonal
11.2.4.2.
Oligonucleotides
11.2.4.3.
Nucleic Acid Probes
11.2.4.4.
Purified Proteins or Peptides
11.2.4.5.
Other Reagents
11.2.5. Implants
11.2.5.1.
Orthopedic Implants
11.2.5.2.
ENT Implants
11.2.5.3.
Breast Implants
11.2.5.4.
Dental Implants
11.2.5.5.
Cardiology related implants
11.2.5.6.
Others
11.2.6. Others
11.3.
Asia Pacific Medical Device Market Revenue (US$ Mn) and Forecasts,
By Country
11.3.1. China
11.3.1.1.
China Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
11.3.1.1.1. Electronics and
Equipment
11.3.1.1.1.1. Diagnostic Imaging
11.3.1.1.1.1.1. X-Ray Equipment
11.3.1.1.1.1.2. Ultrasound Systems
11.3.1.1.1.1.3. Computed Tomography (CT) Scanners
11.3.1.1.1.1.4. Magnetic Resonance Imaging Systems
11.3.1.1.1.1.5. Others
11.3.1.1.1.2. Patient Monitoring
11.3.1.1.1.2.1. Cardiac Monitoring Devices
11.3.1.1.1.2.2. Respiratory Monitoring Devices
11.3.1.1.1.2.3. Others
11.3.1.1.1.3. Diabetic Care Devices
11.3.1.1.1.3.1. Insulin Pens
11.3.1.1.1.3.2. Blood Glucose Meters
11.3.1.1.1.3.3. Blood Glucose Test Strips
11.3.1.1.1.3.4. Continuous Glucose Monitoring Device
11.3.1.1.1.3.5. Others
11.3.1.1.1.4. Orthopedic Devices
11.3.1.1.1.4.1. Prosthetics
11.3.1.1.1.4.2. Braces
11.3.1.1.1.4.3. Fixation Devices
11.3.1.1.1.4.4. Others
11.3.1.1.1.5. Dental Diagnostics and Operative Devices
11.3.1.1.1.6. Others
11.3.1.1.2. Disposables and Consumables – Highest CAGR
11.3.1.1.2.1. Wound Management Products
11.3.1.1.2.2. Drug Delivery Products
11.3.1.1.2.3. Diagnostic and
Laboratory Disposables
11.3.1.1.2.4. Dialysis Disposables
11.3.1.1.2.5. Incontinence Products
11.3.1.1.2.6. Respiratory Supplies
11.3.1.1.2.7. Sterilization Supplies
11.3.1.1.2.8. Non-woven Disposables
11.3.1.1.2.9. Disposable Masks
11.3.1.1.2.10. Disposable Eye Gear
11.3.1.1.2.11. Disposable Gloves
11.3.1.1.2.12. Hand Sanitizers
11.3.1.1.2.12.1. Gel Sanitizers
11.3.1.1.2.12.2. Foam Sanitizers
11.3.1.1.2.12.3. Liquid Sanitizers
11.3.1.1.2.12.4. Other Sanitizers
11.3.1.1.2.13. Others
11.3.1.1.3. Surgical Instruments
11.3.1.1.3.1. Surgical Sutures and Staplers
11.3.1.1.3.2. Handheld Surgical Devices
11.3.1.1.3.2.1. Forceps and Spatulas
11.3.1.1.3.2.2. Retractors
11.3.1.1.3.2.3. Dilators
11.3.1.1.3.2.4. Graspers
11.3.1.1.3.2.5. Auxiliary Instruments
11.3.1.1.3.2.6. Cutter Instruments
11.3.1.1.3.2.7. Others
11.3.1.1.3.3. Electrosurgical Devices
11.3.1.1.3.4. Others
11.3.1.1.4. IVD Reagents
11.3.1.1.4.1. Antibodies
11.3.1.1.4.1.1. Monoclonal
11.3.1.1.4.1.2. Polyclonal
11.3.1.1.4.2. Oligonucleotides
11.3.1.1.4.3. Nucleic Acid Probes
11.3.1.1.4.4. Purified Proteins or Peptides
11.3.1.1.4.5. Other Reagents
11.3.1.1.5. Implants
11.3.1.1.5.1. Orthopedic Implants
11.3.1.1.5.2. ENT Implants
11.3.1.1.5.3. Breast Implants
11.3.1.1.5.4. Dental Implants
11.3.1.1.5.5. Cardiology related implants
11.3.1.1.5.6. Others
11.3.1.1.6. Others
11.3.2. Japan
11.3.2.1.
Japan Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
11.3.2.1.1. Electronics and
Equipment
11.3.2.1.1.1. Diagnostic Imaging
11.3.2.1.1.1.1. X-Ray Equipment
11.3.2.1.1.1.2. Ultrasound Systems
11.3.2.1.1.1.3. Computed Tomography (CT) Scanners
11.3.2.1.1.1.4. Magnetic Resonance Imaging Systems
11.3.2.1.1.1.5. Others
11.3.2.1.1.2. Patient Monitoring
11.3.2.1.1.2.1. Cardiac Monitoring Devices
11.3.2.1.1.2.2. Respiratory Monitoring Devices
11.3.2.1.1.2.3. Others
11.3.2.1.1.3. Diabetic Care Devices
11.3.2.1.1.3.1. Insulin Pens
11.3.2.1.1.3.2. Blood Glucose Meters
11.3.2.1.1.3.3. Blood Glucose Test Strips
11.3.2.1.1.3.4. Continuous Glucose Monitoring Device
11.3.2.1.1.3.5. Others
11.3.2.1.1.4. Orthopedic Devices
11.3.2.1.1.4.1. Prosthetics
11.3.2.1.1.4.2. Braces
11.3.2.1.1.4.3. Fixation Devices
11.3.2.1.1.4.4. Others
11.3.2.1.1.5. Dental Diagnostics and Operative Devices
11.3.2.1.1.6. Others
11.3.2.1.2. Disposables and Consumables – Highest CAGR
11.3.2.1.2.1. Wound Management Products
11.3.2.1.2.2. Drug Delivery Products
11.3.2.1.2.3. Diagnostic and
Laboratory Disposables
11.3.2.1.2.4. Dialysis Disposables
11.3.2.1.2.5. Incontinence Products
11.3.2.1.2.6. Respiratory Supplies
11.3.2.1.2.7. Sterilization Supplies
11.3.2.1.2.8. Non-woven Disposables
11.3.2.1.2.9. Disposable Masks
11.3.2.1.2.10. Disposable Eye Gear
11.3.2.1.2.11. Disposable Gloves
11.3.2.1.2.12. Hand Sanitizers
11.3.2.1.2.12.1. Gel Sanitizers
11.3.2.1.2.12.2. Foam Sanitizers
11.3.2.1.2.12.3. Liquid Sanitizers
11.3.2.1.2.12.4. Other Sanitizers
11.3.2.1.2.13. Others
11.3.2.1.3. Surgical Instruments
11.3.2.1.3.1. Surgical Sutures and Staplers
11.3.2.1.3.2. Handheld Surgical Devices
11.3.2.1.3.2.1. Forceps and Spatulas
11.3.2.1.3.2.2. Retractors
11.3.2.1.3.2.3. Dilators
11.3.2.1.3.2.4. Graspers
11.3.2.1.3.2.5. Auxiliary Instruments
11.3.2.1.3.2.6. Cutter Instruments
11.3.2.1.3.2.7. Others
11.3.2.1.3.3. Electrosurgical Devices
11.3.2.1.3.4. Others
11.3.2.1.4. IVD Reagents
11.3.2.1.4.1. Antibodies
11.3.2.1.4.1.1. Monoclonal
11.3.2.1.4.1.2. Polyclonal
11.3.2.1.4.2. Oligonucleotides
11.3.2.1.4.3. Nucleic Acid Probes
11.3.2.1.4.4. Purified Proteins or Peptides
11.3.2.1.4.5. Other Reagents
11.3.2.1.5. Implants
11.3.2.1.5.1. Orthopedic Implants
11.3.2.1.5.2. ENT Implants
11.3.2.1.5.3. Breast Implants
11.3.2.1.5.4. Dental Implants
11.3.2.1.5.5. Cardiology related implants
11.3.2.1.5.6. Others
11.3.2.1.6. Others
11.3.3. India
11.3.3.1.
India Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
11.3.3.1.1. Electronics and
Equipment
11.3.3.1.1.1. Diagnostic Imaging
11.3.3.1.1.1.1. X-Ray Equipment
11.3.3.1.1.1.2. Ultrasound Systems
11.3.3.1.1.1.3. Computed Tomography (CT) Scanners
11.3.3.1.1.1.4. Magnetic Resonance Imaging Systems
11.3.3.1.1.1.5. Others
11.3.3.1.1.2. Patient Monitoring
11.3.3.1.1.2.1. Cardiac Monitoring Devices
11.3.3.1.1.2.2. Respiratory Monitoring Devices
11.3.3.1.1.2.3. Others
11.3.3.1.1.3. Diabetic Care Devices
11.3.3.1.1.3.1. Insulin Pens
11.3.3.1.1.3.2. Blood Glucose Meters
11.3.3.1.1.3.3. Blood Glucose Test Strips
11.3.3.1.1.3.4. Continuous Glucose Monitoring Device
11.3.3.1.1.3.5. Others
11.3.3.1.1.4. Orthopedic Devices
11.3.3.1.1.4.1. Prosthetics
11.3.3.1.1.4.2. Braces
11.3.3.1.1.4.3. Fixation Devices
11.3.3.1.1.4.4. Others
11.3.3.1.1.5. Dental Diagnostics and Operative Devices
11.3.3.1.1.6. Others
11.3.3.1.2. Disposables and Consumables – Highest CAGR
11.3.3.1.2.1. Wound Management Products
11.3.3.1.2.2. Drug Delivery Products
11.3.3.1.2.3. Diagnostic and
Laboratory Disposables
11.3.3.1.2.4. Dialysis Disposables
11.3.3.1.2.5. Incontinence Products
11.3.3.1.2.6. Respiratory Supplies
11.3.3.1.2.7. Sterilization Supplies
11.3.3.1.2.8. Non-woven Disposables
11.3.3.1.2.9. Disposable Masks
11.3.3.1.2.10. Disposable Eye Gear
11.3.3.1.2.11. Disposable Gloves
11.3.3.1.2.12. Hand Sanitizers
11.3.3.1.2.12.1. Gel Sanitizers
11.3.3.1.2.12.2. Foam Sanitizers
11.3.3.1.2.12.3. Liquid Sanitizers
11.3.3.1.2.12.4. Other Sanitizers
11.3.3.1.2.13. Others
11.3.3.1.3. Surgical Instruments
11.3.3.1.3.1. Surgical Sutures and Staplers
11.3.3.1.3.2. Handheld Surgical Devices
11.3.3.1.3.2.1. Forceps and Spatulas
11.3.3.1.3.2.2. Retractors
11.3.3.1.3.2.3. Dilators
11.3.3.1.3.2.4. Graspers
11.3.3.1.3.2.5. Auxiliary Instruments
11.3.3.1.3.2.6. Cutter Instruments
11.3.3.1.3.2.7. Others
11.3.3.1.3.3. Electrosurgical Devices
11.3.3.1.3.4. Others
11.3.3.1.4. IVD Reagents
11.3.3.1.4.1. Antibodies
11.3.3.1.4.1.1. Monoclonal
11.3.3.1.4.1.2. Polyclonal
11.3.3.1.4.2. Oligonucleotides
11.3.3.1.4.3. Nucleic Acid Probes
11.3.3.1.4.4. Purified Proteins or Peptides
11.3.3.1.4.5. Other Reagents
11.3.3.1.5. Implants
11.3.3.1.5.1. Orthopedic Implants
11.3.3.1.5.2. ENT Implants
11.3.3.1.5.3. Breast Implants
11.3.3.1.5.4. Dental Implants
11.3.3.1.5.5. Cardiology related implants
11.3.3.1.5.6. Others
11.3.3.1.6. Others
11.3.4. New Zealand
11.3.4.1.
New Zealand Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
11.3.4.1.1. Electronics and
Equipment
11.3.4.1.1.1. Diagnostic Imaging
11.3.4.1.1.1.1. X-Ray Equipment
11.3.4.1.1.1.2. Ultrasound Systems
11.3.4.1.1.1.3. Computed Tomography (CT) Scanners
11.3.4.1.1.1.4. Magnetic Resonance Imaging Systems
11.3.4.1.1.1.5. Others
11.3.4.1.1.2. Patient Monitoring
11.3.4.1.1.2.1. Cardiac Monitoring Devices
11.3.4.1.1.2.2. Respiratory Monitoring Devices
11.3.4.1.1.2.3. Others
11.3.4.1.1.3. Diabetic Care Devices
11.3.4.1.1.3.1. Insulin Pens
11.3.4.1.1.3.2. Blood Glucose Meters
11.3.4.1.1.3.3. Blood Glucose Test Strips
11.3.4.1.1.3.4. Continuous Glucose Monitoring Device
11.3.4.1.1.3.5. Others
11.3.4.1.1.4. Orthopedic Devices
11.3.4.1.1.4.1. Prosthetics
11.3.4.1.1.4.2. Braces
11.3.4.1.1.4.3. Fixation Devices
11.3.4.1.1.4.4. Others
11.3.4.1.1.5. Dental Diagnostics and Operative Devices
11.3.4.1.1.6. Others
11.3.4.1.2. Disposables and Consumables – Highest CAGR
11.3.4.1.2.1. Wound Management Products
11.3.4.1.2.2. Drug Delivery Products
11.3.4.1.2.3. Diagnostic and
Laboratory Disposables
11.3.4.1.2.4. Dialysis Disposables
11.3.4.1.2.5. Incontinence Products
11.3.4.1.2.6. Respiratory Supplies
11.3.4.1.2.7. Sterilization Supplies
11.3.4.1.2.8. Non-woven Disposables
11.3.4.1.2.9. Disposable Masks
11.3.4.1.2.10. Disposable Eye Gear
11.3.4.1.2.11. Disposable Gloves
11.3.4.1.2.12. Hand Sanitizers
11.3.4.1.2.12.1. Gel Sanitizers
11.3.4.1.2.12.2. Foam Sanitizers
11.3.4.1.2.12.3. Liquid Sanitizers
11.3.4.1.2.12.4. Other Sanitizers
11.3.4.1.2.13. Others
11.3.4.1.3. Surgical Instruments
11.3.4.1.3.1. Surgical Sutures and Staplers
11.3.4.1.3.2. Handheld Surgical Devices
11.3.4.1.3.2.1. Forceps and Spatulas
11.3.4.1.3.2.2. Retractors
11.3.4.1.3.2.3. Dilators
11.3.4.1.3.2.4. Graspers
11.3.4.1.3.2.5. Auxiliary Instruments
11.3.4.1.3.2.6. Cutter Instruments
11.3.4.1.3.2.7. Others
11.3.4.1.3.3. Electrosurgical Devices
11.3.4.1.3.4. Others
11.3.4.1.4. IVD Reagents
11.3.4.1.4.1. Antibodies
11.3.4.1.4.1.1. Monoclonal
11.3.4.1.4.1.2. Polyclonal
11.3.4.1.4.2. Oligonucleotides
11.3.4.1.4.3. Nucleic Acid Probes
11.3.4.1.4.4. Purified Proteins or Peptides
11.3.4.1.4.5. Other Reagents
11.3.4.1.5. Implants
11.3.4.1.5.1. Orthopedic Implants
11.3.4.1.5.2. ENT Implants
11.3.4.1.5.3. Breast Implants
11.3.4.1.5.4. Dental Implants
11.3.4.1.5.5. Cardiology related implants
11.3.4.1.5.6. Others
11.3.4.1.6. Others
11.3.5. Australia
11.3.5.1.
Australia Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
11.3.5.1.1. Electronics and
Equipment
11.3.5.1.1.1. Diagnostic Imaging
11.3.5.1.1.1.1. X-Ray Equipment
11.3.5.1.1.1.2. Ultrasound Systems
11.3.5.1.1.1.3. Computed Tomography (CT) Scanners
11.3.5.1.1.1.4. Magnetic Resonance Imaging Systems
11.3.5.1.1.1.5. Others
11.3.5.1.1.2. Patient Monitoring
11.3.5.1.1.2.1. Cardiac Monitoring Devices
11.3.5.1.1.2.2. Respiratory Monitoring Devices
11.3.5.1.1.2.3. Others
11.3.5.1.1.3. Diabetic Care Devices
11.3.5.1.1.3.1. Insulin Pens
11.3.5.1.1.3.2. Blood Glucose Meters
11.3.5.1.1.3.3. Blood Glucose Test Strips
11.3.5.1.1.3.4. Continuous Glucose Monitoring Device
11.3.5.1.1.3.5. Others
11.3.5.1.1.4. Orthopedic Devices
11.3.5.1.1.4.1. Prosthetics
11.3.5.1.1.4.2. Braces
11.3.5.1.1.4.3. Fixation Devices
11.3.5.1.1.4.4. Others
11.3.5.1.1.5. Dental Diagnostics and Operative Devices
11.3.5.1.1.6. Others
11.3.5.1.2. Disposables and Consumables – Highest CAGR
11.3.5.1.2.1. Wound Management Products
11.3.5.1.2.2. Drug Delivery Products
11.3.5.1.2.3. Diagnostic and
Laboratory Disposables
11.3.5.1.2.4. Dialysis Disposables
11.3.5.1.2.5. Incontinence Products
11.3.5.1.2.6. Respiratory Supplies
11.3.5.1.2.7. Sterilization Supplies
11.3.5.1.2.8. Non-woven Disposables
11.3.5.1.2.9. Disposable Masks
11.3.5.1.2.10. Disposable Eye Gear
11.3.5.1.2.11. Disposable Gloves
11.3.5.1.2.12. Hand Sanitizers
11.3.5.1.2.12.1. Gel Sanitizers
11.3.5.1.2.12.2. Foam Sanitizers
11.3.5.1.2.12.3. Liquid Sanitizers
11.3.5.1.2.12.4. Other Sanitizers
11.3.5.1.2.13. Others
11.3.5.1.3. Surgical Instruments
11.3.5.1.3.1. Surgical Sutures and Staplers
11.3.5.1.3.2. Handheld Surgical Devices
11.3.5.1.3.2.1. Forceps and Spatulas
11.3.5.1.3.2.2. Retractors
11.3.5.1.3.2.3. Dilators
11.3.5.1.3.2.4. Graspers
11.3.5.1.3.2.5. Auxiliary Instruments
11.3.5.1.3.2.6. Cutter Instruments
11.3.5.1.3.2.7. Others
11.3.5.1.3.3. Electrosurgical Devices
11.3.5.1.3.4. Others
11.3.5.1.4. IVD Reagents
11.3.5.1.4.1. Antibodies
11.3.5.1.4.1.1. Monoclonal
11.3.5.1.4.1.2. Polyclonal
11.3.5.1.4.2. Oligonucleotides
11.3.5.1.4.3. Nucleic Acid Probes
11.3.5.1.4.4. Purified Proteins or Peptides
11.3.5.1.4.5. Other Reagents
11.3.5.1.5. Implants
11.3.5.1.5.1. Orthopedic Implants
11.3.5.1.5.2. ENT Implants
11.3.5.1.5.3. Breast Implants
11.3.5.1.5.4. Dental Implants
11.3.5.1.5.5. Cardiology related implants
11.3.5.1.5.6. Others
11.3.5.1.6. Others
11.3.6. South Korea
11.3.6.1.
South Korea Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
11.3.6.1.1. Electronics and
Equipment
11.3.6.1.1.1. Diagnostic Imaging
11.3.6.1.1.1.1. X-Ray Equipment
11.3.6.1.1.1.2. Ultrasound Systems
11.3.6.1.1.1.3. Computed Tomography (CT) Scanners
11.3.6.1.1.1.4. Magnetic Resonance Imaging Systems
11.3.6.1.1.1.5. Others
11.3.6.1.1.2. Patient Monitoring
11.3.6.1.1.2.1. Cardiac Monitoring Devices
11.3.6.1.1.2.2. Respiratory Monitoring Devices
11.3.6.1.1.2.3. Others
11.3.6.1.1.3. Diabetic Care Devices
11.3.6.1.1.3.1. Insulin Pens
11.3.6.1.1.3.2. Blood Glucose Meters
11.3.6.1.1.3.3. Blood Glucose Test Strips
11.3.6.1.1.3.4. Continuous Glucose Monitoring Device
11.3.6.1.1.3.5. Others
11.3.6.1.1.4. Orthopedic Devices
11.3.6.1.1.4.1. Prosthetics
11.3.6.1.1.4.2. Braces
11.3.6.1.1.4.3. Fixation Devices
11.3.6.1.1.4.4. Others
11.3.6.1.1.5. Dental Diagnostics and Operative Devices
11.3.6.1.1.6. Others
11.3.6.1.2. Disposables and Consumables – Highest CAGR
11.3.6.1.2.1. Wound Management Products
11.3.6.1.2.2. Drug Delivery Products
11.3.6.1.2.3. Diagnostic and
Laboratory Disposables
11.3.6.1.2.4. Dialysis Disposables
11.3.6.1.2.5. Incontinence Products
11.3.6.1.2.6. Respiratory Supplies
11.3.6.1.2.7. Sterilization Supplies
11.3.6.1.2.8. Non-woven Disposables
11.3.6.1.2.9. Disposable Masks
11.3.6.1.2.10. Disposable Eye Gear
11.3.6.1.2.11. Disposable Gloves
11.3.6.1.2.12. Hand Sanitizers
11.3.6.1.2.12.1. Gel Sanitizers
11.3.6.1.2.12.2. Foam Sanitizers
11.3.6.1.2.12.3. Liquid Sanitizers
11.3.6.1.2.12.4. Other Sanitizers
11.3.6.1.2.13. Others
11.3.6.1.3. Surgical Instruments
11.3.6.1.3.1. Surgical Sutures and Staplers
11.3.6.1.3.2. Handheld Surgical Devices
11.3.6.1.3.2.1. Forceps and Spatulas
11.3.6.1.3.2.2. Retractors
11.3.6.1.3.2.3. Dilators
11.3.6.1.3.2.4. Graspers
11.3.6.1.3.2.5. Auxiliary Instruments
11.3.6.1.3.2.6. Cutter Instruments
11.3.6.1.3.2.7. Others
11.3.6.1.3.3. Electrosurgical Devices
11.3.6.1.3.4. Others
11.3.6.1.4. IVD Reagents
11.3.6.1.4.1. Antibodies
11.3.6.1.4.1.1. Monoclonal
11.3.6.1.4.1.2. Polyclonal
11.3.6.1.4.2. Oligonucleotides
11.3.6.1.4.3. Nucleic Acid Probes
11.3.6.1.4.4. Purified Proteins or Peptides
11.3.6.1.4.5. Other Reagents
11.3.6.1.5. Implants
11.3.6.1.5.1. Orthopedic Implants
11.3.6.1.5.2. ENT Implants
11.3.6.1.5.3. Breast Implants
11.3.6.1.5.4. Dental Implants
11.3.6.1.5.5. Cardiology related implants
11.3.6.1.5.6. Others
11.3.6.1.6. Others
11.3.7. Southeast Asia
11.3.7.1.
Southeast Asia Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
11.3.7.1.1. Electronics and
Equipment
11.3.7.1.1.1. Diagnostic Imaging
11.3.7.1.1.1.1. X-Ray Equipment
11.3.7.1.1.1.2. Ultrasound Systems
11.3.7.1.1.1.3. Computed Tomography (CT) Scanners
11.3.7.1.1.1.4. Magnetic Resonance Imaging Systems
11.3.7.1.1.1.5. Others
11.3.7.1.1.2. Patient Monitoring
11.3.7.1.1.2.1. Cardiac Monitoring Devices
11.3.7.1.1.2.2. Respiratory Monitoring Devices
11.3.7.1.1.2.3. Others
11.3.7.1.1.3. Diabetic Care Devices
11.3.7.1.1.3.1. Insulin Pens
11.3.7.1.1.3.2. Blood Glucose Meters
11.3.7.1.1.3.3. Blood Glucose Test Strips
11.3.7.1.1.3.4. Continuous Glucose Monitoring Device
11.3.7.1.1.3.5. Others
11.3.7.1.1.4. Orthopedic Devices
11.3.7.1.1.4.1. Prosthetics
11.3.7.1.1.4.2. Braces
11.3.7.1.1.4.3. Fixation Devices
11.3.7.1.1.4.4. Others
11.3.7.1.1.5. Dental Diagnostics and Operative Devices
11.3.7.1.1.6. Others
11.3.7.1.2. Disposables and Consumables – Highest CAGR
11.3.7.1.2.1. Wound Management Products
11.3.7.1.2.2. Drug Delivery Products
11.3.7.1.2.3. Diagnostic and
Laboratory Disposables
11.3.7.1.2.4. Dialysis Disposables
11.3.7.1.2.5. Incontinence Products
11.3.7.1.2.6. Respiratory Supplies
11.3.7.1.2.7. Sterilization Supplies
11.3.7.1.2.8. Non-woven Disposables
11.3.7.1.2.9. Disposable Masks
11.3.7.1.2.10. Disposable Eye Gear
11.3.7.1.2.11. Disposable Gloves
11.3.7.1.2.12. Hand Sanitizers
11.3.7.1.2.12.1. Gel Sanitizers
11.3.7.1.2.12.2. Foam Sanitizers
11.3.7.1.2.12.3. Liquid Sanitizers
11.3.7.1.2.12.4. Other Sanitizers
11.3.7.1.2.13. Others
11.3.7.1.3. Surgical Instruments
11.3.7.1.3.1. Surgical Sutures and Staplers
11.3.7.1.3.2. Handheld Surgical Devices
11.3.7.1.3.2.1. Forceps and Spatulas
11.3.7.1.3.2.2. Retractors
11.3.7.1.3.2.3. Dilators
11.3.7.1.3.2.4. Graspers
11.3.7.1.3.2.5. Auxiliary Instruments
11.3.7.1.3.2.6. Cutter Instruments
11.3.7.1.3.2.7. Others
11.3.7.1.3.3. Electrosurgical Devices
11.3.7.1.3.4. Others
11.3.7.1.4. IVD Reagents
11.3.7.1.4.1. Antibodies
11.3.7.1.4.1.1. Monoclonal
11.3.7.1.4.1.2. Polyclonal
11.3.7.1.4.2. Oligonucleotides
11.3.7.1.4.3. Nucleic Acid Probes
11.3.7.1.4.4. Purified Proteins or Peptides
11.3.7.1.4.5. Other Reagents
11.3.7.1.5. Implants
11.3.7.1.5.1. Orthopedic Implants
11.3.7.1.5.2. ENT Implants
11.3.7.1.5.3. Breast Implants
11.3.7.1.5.4. Dental Implants
11.3.7.1.5.5. Cardiology related implants
11.3.7.1.5.6. Others
11.3.7.1.6. Others
11.3.7.2.
Southeast Asia Medical Device Market Revenue (US$ Mn) and Forecasts,
By Country
11.3.7.2.1. Indonesia
11.3.7.2.2. Thailand
11.3.7.2.3. Malaysia
11.3.7.2.4. Singapore
11.3.7.2.5. Rest of Southeast Asia
11.3.8. Rest of Asia Pacific
11.3.8.1.
Rest of Asia Pacific
Medical Device Market Revenue (US$ Mn)
and Forecasts, By Type
11.3.8.1.1. Electronics and
Equipment
11.3.8.1.1.1. Diagnostic Imaging
11.3.8.1.1.1.1. X-Ray Equipment
11.3.8.1.1.1.2. Ultrasound Systems
11.3.8.1.1.1.3. Computed Tomography (CT) Scanners
11.3.8.1.1.1.4. Magnetic Resonance Imaging Systems
11.3.8.1.1.1.5. Others
11.3.8.1.1.2. Patient Monitoring
11.3.8.1.1.2.1. Cardiac Monitoring Devices
11.3.8.1.1.2.2. Respiratory Monitoring Devices
11.3.8.1.1.2.3. Others
11.3.8.1.1.3. Diabetic Care Devices
11.3.8.1.1.3.1. Insulin Pens
11.3.8.1.1.3.2. Blood Glucose Meters
11.3.8.1.1.3.3. Blood Glucose Test Strips
11.3.8.1.1.3.4. Continuous Glucose Monitoring Device
11.3.8.1.1.3.5. Others
11.3.8.1.1.4. Orthopedic Devices
11.3.8.1.1.4.1. Prosthetics
11.3.8.1.1.4.2. Braces
11.3.8.1.1.4.3. Fixation Devices
11.3.8.1.1.4.4. Others
11.3.8.1.1.5. Dental Diagnostics and Operative Devices
11.3.8.1.1.6. Others
11.3.8.1.2. Disposables and Consumables – Highest CAGR
11.3.8.1.2.1. Wound Management Products
11.3.8.1.2.2. Drug Delivery Products
11.3.8.1.2.3. Diagnostic and
Laboratory Disposables
11.3.8.1.2.4. Dialysis Disposables
11.3.8.1.2.5. Incontinence Products
11.3.8.1.2.6. Respiratory Supplies
11.3.8.1.2.7. Sterilization Supplies
11.3.8.1.2.8. Non-woven Disposables
11.3.8.1.2.9. Disposable Masks
11.3.8.1.2.10. Disposable Eye Gear
11.3.8.1.2.11. Disposable Gloves
11.3.8.1.2.12. Hand Sanitizers
11.3.8.1.2.12.1. Gel Sanitizers
11.3.8.1.2.12.2. Foam Sanitizers
11.3.8.1.2.12.3. Liquid Sanitizers
11.3.8.1.2.12.4. Other Sanitizers
11.3.8.1.2.13. Others
11.3.8.1.3. Surgical Instruments
11.3.8.1.3.1. Surgical Sutures and Staplers
11.3.8.1.3.2. Handheld Surgical Devices
11.3.8.1.3.2.1. Forceps and Spatulas
11.3.8.1.3.2.2. Retractors
11.3.8.1.3.2.3. Dilators
11.3.8.1.3.2.4. Graspers
11.3.8.1.3.2.5. Auxiliary Instruments
11.3.8.1.3.2.6. Cutter Instruments
11.3.8.1.3.2.7. Others
11.3.8.1.3.3. Electrosurgical Devices
11.3.8.1.3.4. Others
11.3.8.1.4. IVD Reagents
11.3.8.1.4.1. Antibodies
11.3.8.1.4.1.1. Monoclonal
11.3.8.1.4.1.2. Polyclonal
11.3.8.1.4.2. Oligonucleotides
11.3.8.1.4.3. Nucleic Acid Probes
11.3.8.1.4.4. Purified Proteins or Peptides
11.3.8.1.4.5. Other Reagents
11.3.8.1.5. Implants
11.3.8.1.5.1. Orthopedic Implants
11.3.8.1.5.2. ENT Implants
11.3.8.1.5.3. Breast Implants
11.3.8.1.5.4. Dental Implants
11.3.8.1.5.5. Cardiology related implants
11.3.8.1.5.6. Others
11.3.8.1.6. Others
11.4.
Key Segment for Channeling
Investments
11.4.1. By Country
11.4.2. By Type
12. Middle East and Africa Medical Device Market Analysis and Forecasts, 2020 -
2028
12.1.
Overview
12.1.1. Middle East and Africa Medical
Device Market Revenue (US$ Mn)
12.2.
Middle East and Africa
Medical Device Market Revenue (US$ Mn)
and Forecasts, By Type
12.2.1. Electronics and Equipment
12.2.1.1.
Diagnostic Imaging
12.2.1.1.1. X-Ray Equipment
12.2.1.1.2. Ultrasound Systems
12.2.1.1.3. Computed Tomography (CT) Scanners
12.2.1.1.4. Magnetic Resonance Imaging Systems
12.2.1.1.5. Others
12.2.1.2.
Patient Monitoring
12.2.1.2.1. Cardiac Monitoring Devices
12.2.1.2.2. Respiratory Monitoring Devices
12.2.1.2.3. Others
12.2.1.3.
Diabetic Care Devices
12.2.1.3.1. Insulin Pens
12.2.1.3.2. Blood Glucose Meters
12.2.1.3.3. Blood Glucose Test Strips
12.2.1.3.4. Continuous Glucose Monitoring Device
12.2.1.3.5. Others
12.2.1.4.
Orthopedic Devices
12.2.1.4.1. Prosthetics
12.2.1.4.2. Braces
12.2.1.4.3. Fixation Devices
12.2.1.4.4. Others
12.2.1.5.
Dental Diagnostics and Operative
Devices
12.2.1.6.
Others
12.2.2. Disposables and Consumables – Highest CAGR
12.2.2.1.
Wound Management Products
12.2.2.2.
Drug Delivery Products
12.2.2.3.
Diagnostic and Laboratory Disposables
12.2.2.4.
Dialysis Disposables
12.2.2.5.
Incontinence Products
12.2.2.6.
Respiratory Supplies
12.2.2.7.
Sterilization Supplies
12.2.2.8.
Non-woven Disposables
12.2.2.9.
Disposable Masks
12.2.2.10. Disposable Eye Gear
12.2.2.11. Disposable Gloves
12.2.2.12. Hand Sanitizers
12.2.2.12.1. Gel Sanitizers
12.2.2.12.2. Foam Sanitizers
12.2.2.12.3. Liquid Sanitizers
12.2.2.12.4. Other Sanitizers
12.2.2.13. Others
12.2.3. Surgical Instruments
12.2.3.1.
Surgical Sutures and Staplers
12.2.3.2.
Handheld Surgical Devices
12.2.3.2.1. Forceps and Spatulas
12.2.3.2.2. Retractors
12.2.3.2.3. Dilators
12.2.3.2.4. Graspers
12.2.3.2.5. Auxiliary Instruments
12.2.3.2.6. Cutter Instruments
12.2.3.2.7. Others
12.2.3.3.
Electrosurgical Devices
12.2.3.4.
Others
12.2.4. IVD Reagents
12.2.4.1.
Antibodies
12.2.4.1.1. Monoclonal
12.2.4.1.2. Polyclonal
12.2.4.2.
Oligonucleotides
12.2.4.3.
Nucleic Acid Probes
12.2.4.4.
Purified Proteins or Peptides
12.2.4.5.
Other Reagents
12.2.5. Implants
12.2.5.1.
Orthopedic Implants
12.2.5.2.
ENT Implants
12.2.5.3.
Breast Implants
12.2.5.4.
Dental Implants
12.2.5.5.
Cardiology related implants
12.2.5.6.
Others
12.2.6. Others
12.3.
Middle East and Africa
Medical Device Market Revenue (US$ Mn)
and Forecasts, By Country
12.3.1. Saudi Arabia
12.3.1.1.
Saudi Arabia Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
12.3.1.1.1. Electronics and
Equipment
12.3.1.1.1.1. Diagnostic Imaging
12.3.1.1.1.1.1. X-Ray Equipment
12.3.1.1.1.1.2. Ultrasound Systems
12.3.1.1.1.1.3. Computed Tomography (CT) Scanners
12.3.1.1.1.1.4. Magnetic Resonance Imaging Systems
12.3.1.1.1.1.5. Others
12.3.1.1.1.2. Patient Monitoring
12.3.1.1.1.2.1. Cardiac Monitoring Devices
12.3.1.1.1.2.2. Respiratory Monitoring Devices
12.3.1.1.1.2.3. Others
12.3.1.1.1.3. Diabetic Care Devices
12.3.1.1.1.3.1. Insulin Pens
12.3.1.1.1.3.2. Blood Glucose Meters
12.3.1.1.1.3.3. Blood Glucose Test Strips
12.3.1.1.1.3.4. Continuous Glucose Monitoring Device
12.3.1.1.1.3.5. Others
12.3.1.1.1.4. Orthopedic Devices
12.3.1.1.1.4.1. Prosthetics
12.3.1.1.1.4.2. Braces
12.3.1.1.1.4.3. Fixation Devices
12.3.1.1.1.4.4. Others
12.3.1.1.1.5. Dental Diagnostics and Operative Devices
12.3.1.1.1.6. Others
12.3.1.1.2. Disposables and Consumables – Highest CAGR
12.3.1.1.2.1. Wound Management Products
12.3.1.1.2.2. Drug Delivery Products
12.3.1.1.2.3. Diagnostic and
Laboratory Disposables
12.3.1.1.2.4. Dialysis Disposables
12.3.1.1.2.5. Incontinence Products
12.3.1.1.2.6. Respiratory Supplies
12.3.1.1.2.7. Sterilization Supplies
12.3.1.1.2.8. Non-woven Disposables
12.3.1.1.2.9. Disposable Masks
12.3.1.1.2.10. Disposable Eye Gear
12.3.1.1.2.11. Disposable Gloves
12.3.1.1.2.12. Hand Sanitizers
12.3.1.1.2.12.1. Gel Sanitizers
12.3.1.1.2.12.2. Foam Sanitizers
12.3.1.1.2.12.3. Liquid Sanitizers
12.3.1.1.2.12.4. Other Sanitizers
12.3.1.1.2.13. Others
12.3.1.1.3. Surgical Instruments
12.3.1.1.3.1. Surgical Sutures and Staplers
12.3.1.1.3.2. Handheld Surgical Devices
12.3.1.1.3.2.1. Forceps and Spatulas
12.3.1.1.3.2.2. Retractors
12.3.1.1.3.2.3. Dilators
12.3.1.1.3.2.4. Graspers
12.3.1.1.3.2.5. Auxiliary Instruments
12.3.1.1.3.2.6. Cutter Instruments
12.3.1.1.3.2.7. Others
12.3.1.1.3.3. Electrosurgical Devices
12.3.1.1.3.4. Others
12.3.1.1.4. IVD Reagents
12.3.1.1.4.1. Antibodies
12.3.1.1.4.1.1. Monoclonal
12.3.1.1.4.1.2. Polyclonal
12.3.1.1.4.2. Oligonucleotides
12.3.1.1.4.3. Nucleic Acid Probes
12.3.1.1.4.4. Purified Proteins or Peptides
12.3.1.1.4.5. Other Reagents
12.3.1.1.5. Implants
12.3.1.1.5.1. Orthopedic Implants
12.3.1.1.5.2. ENT Implants
12.3.1.1.5.3. Breast Implants
12.3.1.1.5.4. Dental Implants
12.3.1.1.5.5. Cardiology related implants
12.3.1.1.5.6. Others
12.3.1.1.6. Others
12.3.2. UAE
12.3.2.1.
UAE Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
12.3.2.1.1. Electronics and
Equipment
12.3.2.1.1.1. Diagnostic Imaging
12.3.2.1.1.1.1. X-Ray Equipment
12.3.2.1.1.1.2. Ultrasound Systems
12.3.2.1.1.1.3. Computed Tomography (CT) Scanners
12.3.2.1.1.1.4. Magnetic Resonance Imaging Systems
12.3.2.1.1.1.5. Others
12.3.2.1.1.2. Patient Monitoring
12.3.2.1.1.2.1. Cardiac Monitoring Devices
12.3.2.1.1.2.2. Respiratory Monitoring Devices
12.3.2.1.1.2.3. Others
12.3.2.1.1.3. Diabetic Care Devices
12.3.2.1.1.3.1. Insulin Pens
12.3.2.1.1.3.2. Blood Glucose Meters
12.3.2.1.1.3.3. Blood Glucose Test Strips
12.3.2.1.1.3.4. Continuous Glucose Monitoring Device
12.3.2.1.1.3.5. Others
12.3.2.1.1.4. Orthopedic Devices
12.3.2.1.1.4.1. Prosthetics
12.3.2.1.1.4.2. Braces
12.3.2.1.1.4.3. Fixation Devices
12.3.2.1.1.4.4. Others
12.3.2.1.1.5. Dental Diagnostics and Operative Devices
12.3.2.1.1.6. Others
12.3.2.1.2. Disposables and Consumables – Highest CAGR
12.3.2.1.2.1. Wound Management Products
12.3.2.1.2.2. Drug Delivery Products
12.3.2.1.2.3. Diagnostic and
Laboratory Disposables
12.3.2.1.2.4. Dialysis Disposables
12.3.2.1.2.5. Incontinence Products
12.3.2.1.2.6. Respiratory Supplies
12.3.2.1.2.7. Sterilization Supplies
12.3.2.1.2.8. Non-woven Disposables
12.3.2.1.2.9. Disposable Masks
12.3.2.1.2.10. Disposable Eye Gear
12.3.2.1.2.11. Disposable Gloves
12.3.2.1.2.12. Hand Sanitizers
12.3.2.1.2.12.1. Gel Sanitizers
12.3.2.1.2.12.2. Foam Sanitizers
12.3.2.1.2.12.3. Liquid Sanitizers
12.3.2.1.2.12.4. Other Sanitizers
12.3.2.1.2.13. Others
12.3.2.1.3. Surgical Instruments
12.3.2.1.3.1. Surgical Sutures and Staplers
12.3.2.1.3.2. Handheld Surgical Devices
12.3.2.1.3.2.1. Forceps and Spatulas
12.3.2.1.3.2.2. Retractors
12.3.2.1.3.2.3. Dilators
12.3.2.1.3.2.4. Graspers
12.3.2.1.3.2.5. Auxiliary Instruments
12.3.2.1.3.2.6. Cutter Instruments
12.3.2.1.3.2.7. Others
12.3.2.1.3.3. Electrosurgical Devices
12.3.2.1.3.4. Others
12.3.2.1.4. IVD Reagents
12.3.2.1.4.1. Antibodies
12.3.2.1.4.1.1. Monoclonal
12.3.2.1.4.1.2. Polyclonal
12.3.2.1.4.2. Oligonucleotides
12.3.2.1.4.3. Nucleic Acid Probes
12.3.2.1.4.4. Purified Proteins or Peptides
12.3.2.1.4.5. Other Reagents
12.3.2.1.5. Implants
12.3.2.1.5.1. Orthopedic Implants
12.3.2.1.5.2. ENT Implants
12.3.2.1.5.3. Breast Implants
12.3.2.1.5.4. Dental Implants
12.3.2.1.5.5. Cardiology related implants
12.3.2.1.5.6. Others
12.3.2.1.6. Others
12.3.3. Egypt
12.3.3.1.
Egypt Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
12.3.3.1.1. Electronics and
Equipment
12.3.3.1.1.1. Diagnostic Imaging
12.3.3.1.1.1.1. X-Ray Equipment
12.3.3.1.1.1.2. Ultrasound Systems
12.3.3.1.1.1.3. Computed Tomography (CT) Scanners
12.3.3.1.1.1.4. Magnetic Resonance Imaging Systems
12.3.3.1.1.1.5. Others
12.3.3.1.1.2. Patient Monitoring
12.3.3.1.1.2.1. Cardiac Monitoring Devices
12.3.3.1.1.2.2. Respiratory Monitoring Devices
12.3.3.1.1.2.3. Others
12.3.3.1.1.3. Diabetic Care Devices
12.3.3.1.1.3.1. Insulin Pens
12.3.3.1.1.3.2. Blood Glucose Meters
12.3.3.1.1.3.3. Blood Glucose Test Strips
12.3.3.1.1.3.4. Continuous Glucose Monitoring Device
12.3.3.1.1.3.5. Others
12.3.3.1.1.4. Orthopedic Devices
12.3.3.1.1.4.1. Prosthetics
12.3.3.1.1.4.2. Braces
12.3.3.1.1.4.3. Fixation Devices
12.3.3.1.1.4.4. Others
12.3.3.1.1.5. Dental Diagnostics and Operative Devices
12.3.3.1.1.6. Others
12.3.3.1.2. Disposables and Consumables – Highest CAGR
12.3.3.1.2.1. Wound Management Products
12.3.3.1.2.2. Drug Delivery Products
12.3.3.1.2.3. Diagnostic and
Laboratory Disposables
12.3.3.1.2.4. Dialysis Disposables
12.3.3.1.2.5. Incontinence Products
12.3.3.1.2.6. Respiratory Supplies
12.3.3.1.2.7. Sterilization Supplies
12.3.3.1.2.8. Non-woven Disposables
12.3.3.1.2.9. Disposable Masks
12.3.3.1.2.10. Disposable Eye Gear
12.3.3.1.2.11. Disposable Gloves
12.3.3.1.2.12. Hand Sanitizers
12.3.3.1.2.12.1. Gel Sanitizers
12.3.3.1.2.12.2. Foam Sanitizers
12.3.3.1.2.12.3. Liquid Sanitizers
12.3.3.1.2.12.4. Other Sanitizers
12.3.3.1.2.13. Others
12.3.3.1.3. Surgical Instruments
12.3.3.1.3.1. Surgical Sutures and Staplers
12.3.3.1.3.2. Handheld Surgical Devices
12.3.3.1.3.2.1. Forceps and Spatulas
12.3.3.1.3.2.2. Retractors
12.3.3.1.3.2.3. Dilators
12.3.3.1.3.2.4. Graspers
12.3.3.1.3.2.5. Auxiliary Instruments
12.3.3.1.3.2.6. Cutter Instruments
12.3.3.1.3.2.7. Others
12.3.3.1.3.3. Electrosurgical Devices
12.3.3.1.3.4. Others
12.3.3.1.4. IVD Reagents
12.3.3.1.4.1. Antibodies
12.3.3.1.4.1.1. Monoclonal
12.3.3.1.4.1.2. Polyclonal
12.3.3.1.4.2. Oligonucleotides
12.3.3.1.4.3. Nucleic Acid Probes
12.3.3.1.4.4. Purified Proteins or Peptides
12.3.3.1.4.5. Other Reagents
12.3.3.1.5. Implants
12.3.3.1.5.1. Orthopedic Implants
12.3.3.1.5.2. ENT Implants
12.3.3.1.5.3. Breast Implants
12.3.3.1.5.4. Dental Implants
12.3.3.1.5.5. Cardiology related implants
12.3.3.1.5.6. Others
12.3.3.1.6. Others
12.3.4. Kuwait
12.3.4.1.
Kuwait Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
12.3.4.1.1. Electronics and
Equipment
12.3.4.1.1.1. Diagnostic Imaging
12.3.4.1.1.1.1. X-Ray Equipment
12.3.4.1.1.1.2. Ultrasound Systems
12.3.4.1.1.1.3. Computed Tomography (CT) Scanners
12.3.4.1.1.1.4. Magnetic Resonance Imaging Systems
12.3.4.1.1.1.5. Others
12.3.4.1.1.2. Patient Monitoring
12.3.4.1.1.2.1. Cardiac Monitoring Devices
12.3.4.1.1.2.2. Respiratory Monitoring Devices
12.3.4.1.1.2.3. Others
12.3.4.1.1.3. Diabetic Care Devices
12.3.4.1.1.3.1. Insulin Pens
12.3.4.1.1.3.2. Blood Glucose Meters
12.3.4.1.1.3.3. Blood Glucose Test Strips
12.3.4.1.1.3.4. Continuous Glucose Monitoring Device
12.3.4.1.1.3.5. Others
12.3.4.1.1.4. Orthopedic Devices
12.3.4.1.1.4.1. Prosthetics
12.3.4.1.1.4.2. Braces
12.3.4.1.1.4.3. Fixation Devices
12.3.4.1.1.4.4. Others
12.3.4.1.1.5. Dental Diagnostics and Operative Devices
12.3.4.1.1.6. Others
12.3.4.1.2. Disposables and Consumables – Highest CAGR
12.3.4.1.2.1. Wound Management Products
12.3.4.1.2.2. Drug Delivery Products
12.3.4.1.2.3. Diagnostic and
Laboratory Disposables
12.3.4.1.2.4. Dialysis Disposables
12.3.4.1.2.5. Incontinence Products
12.3.4.1.2.6. Respiratory Supplies
12.3.4.1.2.7. Sterilization Supplies
12.3.4.1.2.8. Non-woven Disposables
12.3.4.1.2.9. Disposable Masks
12.3.4.1.2.10. Disposable Eye Gear
12.3.4.1.2.11. Disposable Gloves
12.3.4.1.2.12. Hand Sanitizers
12.3.4.1.2.12.1. Gel Sanitizers
12.3.4.1.2.12.2. Foam Sanitizers
12.3.4.1.2.12.3. Liquid Sanitizers
12.3.4.1.2.12.4. Other Sanitizers
12.3.4.1.2.13. Others
12.3.4.1.3. Surgical Instruments
12.3.4.1.3.1. Surgical Sutures and Staplers
12.3.4.1.3.2. Handheld Surgical Devices
12.3.4.1.3.2.1. Forceps and Spatulas
12.3.4.1.3.2.2. Retractors
12.3.4.1.3.2.3. Dilators
12.3.4.1.3.2.4. Graspers
12.3.4.1.3.2.5. Auxiliary Instruments
12.3.4.1.3.2.6. Cutter Instruments
12.3.4.1.3.2.7. Others
12.3.4.1.3.3. Electrosurgical Devices
12.3.4.1.3.4. Others
12.3.4.1.4. IVD Reagents
12.3.4.1.4.1. Antibodies
12.3.4.1.4.1.1. Monoclonal
12.3.4.1.4.1.2. Polyclonal
12.3.4.1.4.2. Oligonucleotides
12.3.4.1.4.3. Nucleic Acid Probes
12.3.4.1.4.4. Purified Proteins or Peptides
12.3.4.1.4.5. Other Reagents
12.3.4.1.5. Implants
12.3.4.1.5.1. Orthopedic Implants
12.3.4.1.5.2. ENT Implants
12.3.4.1.5.3. Breast Implants
12.3.4.1.5.4. Dental Implants
12.3.4.1.5.5. Cardiology related implants
12.3.4.1.5.6. Others
12.3.4.1.6. Others
12.3.5. South Africa
12.3.5.1.
South Africa Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
12.3.5.1.1. Electronics and
Equipment
12.3.5.1.1.1. Diagnostic Imaging
12.3.5.1.1.1.1. X-Ray Equipment
12.3.5.1.1.1.2. Ultrasound Systems
12.3.5.1.1.1.3. Computed Tomography (CT) Scanners
12.3.5.1.1.1.4. Magnetic Resonance Imaging Systems
12.3.5.1.1.1.5. Others
12.3.5.1.1.2. Patient Monitoring
12.3.5.1.1.2.1. Cardiac Monitoring Devices
12.3.5.1.1.2.2. Respiratory Monitoring Devices
12.3.5.1.1.2.3. Others
12.3.5.1.1.3. Diabetic Care Devices
12.3.5.1.1.3.1. Insulin Pens
12.3.5.1.1.3.2. Blood Glucose Meters
12.3.5.1.1.3.3. Blood Glucose Test Strips
12.3.5.1.1.3.4. Continuous Glucose Monitoring Device
12.3.5.1.1.3.5. Others
12.3.5.1.1.4. Orthopedic Devices
12.3.5.1.1.4.1. Prosthetics
12.3.5.1.1.4.2. Braces
12.3.5.1.1.4.3. Fixation Devices
12.3.5.1.1.4.4. Others
12.3.5.1.1.5. Dental Diagnostics and Operative Devices
12.3.5.1.1.6. Others
12.3.5.1.2. Disposables and Consumables – Highest CAGR
12.3.5.1.2.1. Wound Management Products
12.3.5.1.2.2. Drug Delivery Products
12.3.5.1.2.3. Diagnostic and
Laboratory Disposables
12.3.5.1.2.4. Dialysis Disposables
12.3.5.1.2.5. Incontinence Products
12.3.5.1.2.6. Respiratory Supplies
12.3.5.1.2.7. Sterilization Supplies
12.3.5.1.2.8. Non-woven Disposables
12.3.5.1.2.9. Disposable Masks
12.3.5.1.2.10. Disposable Eye Gear
12.3.5.1.2.11. Disposable Gloves
12.3.5.1.2.12. Hand Sanitizers
12.3.5.1.2.12.1. Gel Sanitizers
12.3.5.1.2.12.2. Foam Sanitizers
12.3.5.1.2.12.3. Liquid Sanitizers
12.3.5.1.2.12.4. Other Sanitizers
12.3.5.1.2.13. Others
12.3.5.1.3. Surgical Instruments
12.3.5.1.3.1. Surgical Sutures and Staplers
12.3.5.1.3.2. Handheld Surgical Devices
12.3.5.1.3.2.1. Forceps and Spatulas
12.3.5.1.3.2.2. Retractors
12.3.5.1.3.2.3. Dilators
12.3.5.1.3.2.4. Graspers
12.3.5.1.3.2.5. Auxiliary Instruments
12.3.5.1.3.2.6. Cutter Instruments
12.3.5.1.3.2.7. Others
12.3.5.1.3.3. Electrosurgical Devices
12.3.5.1.3.4. Others
12.3.5.1.4. IVD Reagents
12.3.5.1.4.1. Antibodies
12.3.5.1.4.1.1. Monoclonal
12.3.5.1.4.1.2. Polyclonal
12.3.5.1.4.2. Oligonucleotides
12.3.5.1.4.3. Nucleic Acid Probes
12.3.5.1.4.4. Purified Proteins or Peptides
12.3.5.1.4.5. Other Reagents
12.3.5.1.5. Implants
12.3.5.1.5.1. Orthopedic Implants
12.3.5.1.5.2. ENT Implants
12.3.5.1.5.3. Breast Implants
12.3.5.1.5.4. Dental Implants
12.3.5.1.5.5. Cardiology related implants
12.3.5.1.5.6. Others
12.3.5.1.6. Others
12.3.6. Rest of Middle East & Africa
12.3.6.1.
Rest of Middle East & Africa
Medical Device Market Revenue (US$ Mn)
and Forecasts, By Type
12.3.6.1.1. Electronics and
Equipment
12.3.6.1.1.1. Diagnostic Imaging
12.3.6.1.1.1.1. X-Ray Equipment
12.3.6.1.1.1.2. Ultrasound Systems
12.3.6.1.1.1.3. Computed Tomography (CT) Scanners
12.3.6.1.1.1.4. Magnetic Resonance Imaging Systems
12.3.6.1.1.1.5. Others
12.3.6.1.1.2. Patient Monitoring
12.3.6.1.1.2.1. Cardiac Monitoring Devices
12.3.6.1.1.2.2. Respiratory Monitoring Devices
12.3.6.1.1.2.3. Others
12.3.6.1.1.3. Diabetic Care Devices
12.3.6.1.1.3.1. Insulin Pens
12.3.6.1.1.3.2. Blood Glucose Meters
12.3.6.1.1.3.3. Blood Glucose Test Strips
12.3.6.1.1.3.4. Continuous Glucose Monitoring Device
12.3.6.1.1.3.5. Others
12.3.6.1.1.4. Orthopedic Devices
12.3.6.1.1.4.1. Prosthetics
12.3.6.1.1.4.2. Braces
12.3.6.1.1.4.3. Fixation Devices
12.3.6.1.1.4.4. Others
12.3.6.1.1.5. Dental Diagnostics and Operative Devices
12.3.6.1.1.6. Others
12.3.6.1.2. Disposables and Consumables – Highest CAGR
12.3.6.1.2.1. Wound Management Products
12.3.6.1.2.2. Drug Delivery Products
12.3.6.1.2.3. Diagnostic and
Laboratory Disposables
12.3.6.1.2.4. Dialysis Disposables
12.3.6.1.2.5. Incontinence Products
12.3.6.1.2.6. Respiratory Supplies
12.3.6.1.2.7. Sterilization Supplies
12.3.6.1.2.8. Non-woven Disposables
12.3.6.1.2.9. Disposable Masks
12.3.6.1.2.10. Disposable Eye Gear
12.3.6.1.2.11. Disposable Gloves
12.3.6.1.2.12. Hand Sanitizers
12.3.6.1.2.12.1. Gel Sanitizers
12.3.6.1.2.12.2. Foam Sanitizers
12.3.6.1.2.12.3. Liquid Sanitizers
12.3.6.1.2.12.4. Other Sanitizers
12.3.6.1.2.13. Others
12.3.6.1.3. Surgical Instruments
12.3.6.1.3.1. Surgical Sutures and Staplers
12.3.6.1.3.2. Handheld Surgical Devices
12.3.6.1.3.2.1. Forceps and Spatulas
12.3.6.1.3.2.2. Retractors
12.3.6.1.3.2.3. Dilators
12.3.6.1.3.2.4. Graspers
12.3.6.1.3.2.5. Auxiliary Instruments
12.3.6.1.3.2.6. Cutter Instruments
12.3.6.1.3.2.7. Others
12.3.6.1.3.3. Electrosurgical Devices
12.3.6.1.3.4. Others
12.3.6.1.4. IVD Reagents
12.3.6.1.4.1. Antibodies
12.3.6.1.4.1.1. Monoclonal
12.3.6.1.4.1.2. Polyclonal
12.3.6.1.4.2. Oligonucleotides
12.3.6.1.4.3. Nucleic Acid Probes
12.3.6.1.4.4. Purified Proteins or Peptides
12.3.6.1.4.5. Other Reagents
12.3.6.1.5. Implants
12.3.6.1.5.1. Orthopedic Implants
12.3.6.1.5.2. ENT Implants
12.3.6.1.5.3. Breast Implants
12.3.6.1.5.4. Dental Implants
12.3.6.1.5.5. Cardiology related implants
12.3.6.1.5.6. Others
12.3.6.1.6. Others
12.4.
Key Segment for Channeling
Investments
12.4.1. By Country
12.4.2. By Type
13. Latin America Medical
Device Market Analysis and Forecasts, 2020 - 2028
13.1.
Overview
13.1.1. Latin America Medical
Device Market Revenue (US$ Mn)
13.2.
Latin America Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
13.2.1. Electronics and
Equipment
13.2.1.1.
Diagnostic Imaging
13.2.1.1.1. X-Ray Equipment
13.2.1.1.2. Ultrasound Systems
13.2.1.1.3. Computed Tomography (CT) Scanners
13.2.1.1.4. Magnetic Resonance Imaging Systems
13.2.1.1.5. Others
13.2.1.2.
Patient Monitoring
13.2.1.2.1. Cardiac Monitoring Devices
13.2.1.2.2. Respiratory Monitoring Devices
13.2.1.2.3. Others
13.2.1.3.
Diabetic Care Devices
13.2.1.3.1. Insulin Pens
13.2.1.3.2. Blood Glucose Meters
13.2.1.3.3. Blood Glucose Test Strips
13.2.1.3.4. Continuous Glucose Monitoring Device
13.2.1.3.5. Others
13.2.1.4.
Orthopedic Devices
13.2.1.4.1. Prosthetics
13.2.1.4.2. Braces
13.2.1.4.3. Fixation Devices
13.2.1.4.4. Others
13.2.1.5.
Dental Diagnostics and Operative
Devices
13.2.1.6.
Others
13.2.2. Disposables and Consumables – Highest CAGR
13.2.2.1.
Wound Management Products
13.2.2.2.
Drug Delivery Products
13.2.2.3.
Diagnostic and Laboratory Disposables
13.2.2.4.
Dialysis Disposables
13.2.2.5.
Incontinence Products
13.2.2.6.
Respiratory Supplies
13.2.2.7.
Sterilization Supplies
13.2.2.8.
Non-woven Disposables
13.2.2.9.
Disposable Masks
13.2.2.10. Disposable Eye Gear
13.2.2.11. Disposable Gloves
13.2.2.12. Hand Sanitizers
13.2.2.12.1. Gel Sanitizers
13.2.2.12.2. Foam Sanitizers
13.2.2.12.3. Liquid Sanitizers
13.2.2.12.4. Other Sanitizers
13.2.2.13. Others
13.2.3. Surgical Instruments
13.2.3.1.
Surgical Sutures and Staplers
13.2.3.2.
Handheld Surgical Devices
13.2.3.2.1. Forceps and Spatulas
13.2.3.2.2. Retractors
13.2.3.2.3. Dilators
13.2.3.2.4. Graspers
13.2.3.2.5. Auxiliary Instruments
13.2.3.2.6. Cutter Instruments
13.2.3.2.7. Others
13.2.3.3.
Electrosurgical Devices
13.2.3.4.
Others
13.2.4. IVD Reagents
13.2.4.1.
Antibodies
13.2.4.1.1. Monoclonal
13.2.4.1.2. Polyclonal
13.2.4.2.
Oligonucleotides
13.2.4.3.
Nucleic Acid Probes
13.2.4.4.
Purified Proteins or Peptides
13.2.4.5.
Other Reagents
13.2.5. Implants
13.2.5.1.
Orthopedic Implants
13.2.5.2.
ENT Implants
13.2.5.3.
Breast Implants
13.2.5.4.
Dental Implants
13.2.5.5.
Cardiology related implants
13.2.5.6.
Others
13.2.6. Others
13.3.
Latin America Medical Device Market Revenue (US$ Mn) and Forecasts,
By Country
13.3.1. Brazil
13.3.1.1.
Brazil Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
13.3.1.1.1. Electronics and
Equipment
13.3.1.1.1.1. Diagnostic Imaging
13.3.1.1.1.1.1. X-Ray Equipment
13.3.1.1.1.1.2. Ultrasound Systems
13.3.1.1.1.1.3. Computed Tomography (CT) Scanners
13.3.1.1.1.1.4. Magnetic Resonance Imaging Systems
13.3.1.1.1.1.5. Others
13.3.1.1.1.2. Patient Monitoring
13.3.1.1.1.2.1. Cardiac Monitoring Devices
13.3.1.1.1.2.2. Respiratory Monitoring Devices
13.3.1.1.1.2.3. Others
13.3.1.1.1.3. Diabetic Care Devices
13.3.1.1.1.3.1. Insulin Pens
13.3.1.1.1.3.2. Blood Glucose Meters
13.3.1.1.1.3.3. Blood Glucose Test Strips
13.3.1.1.1.3.4. Continuous Glucose Monitoring Device
13.3.1.1.1.3.5. Others
13.3.1.1.1.4. Orthopedic Devices
13.3.1.1.1.4.1. Prosthetics
13.3.1.1.1.4.2. Braces
13.3.1.1.1.4.3. Fixation Devices
13.3.1.1.1.4.4. Others
13.3.1.1.1.5. Dental Diagnostics and Operative Devices
13.3.1.1.1.6. Others
13.3.1.1.2. Disposables and Consumables – Highest CAGR
13.3.1.1.2.1. Wound Management Products
13.3.1.1.2.2. Drug Delivery Products
13.3.1.1.2.3. Diagnostic and
Laboratory Disposables
13.3.1.1.2.4. Dialysis Disposables
13.3.1.1.2.5. Incontinence Products
13.3.1.1.2.6. Respiratory Supplies
13.3.1.1.2.7. Sterilization Supplies
13.3.1.1.2.8. Non-woven Disposables
13.3.1.1.2.9. Disposable Masks
13.3.1.1.2.10. Disposable Eye Gear
13.3.1.1.2.11. Disposable Gloves
13.3.1.1.2.12. Hand Sanitizers
13.3.1.1.2.12.1. Gel Sanitizers
13.3.1.1.2.12.2. Foam Sanitizers
13.3.1.1.2.12.3. Liquid Sanitizers
13.3.1.1.2.12.4. Other Sanitizers
13.3.1.1.2.13. Others
13.3.1.1.3. Surgical Instruments
13.3.1.1.3.1. Surgical Sutures and Staplers
13.3.1.1.3.2. Handheld Surgical Devices
13.3.1.1.3.2.1. Forceps and Spatulas
13.3.1.1.3.2.2. Retractors
13.3.1.1.3.2.3. Dilators
13.3.1.1.3.2.4. Graspers
13.3.1.1.3.2.5. Auxiliary Instruments
13.3.1.1.3.2.6. Cutter Instruments
13.3.1.1.3.2.7. Others
13.3.1.1.3.3. Electrosurgical Devices
13.3.1.1.3.4. Others
13.3.1.1.4. IVD Reagents
13.3.1.1.4.1. Antibodies
13.3.1.1.4.1.1. Monoclonal
13.3.1.1.4.1.2. Polyclonal
13.3.1.1.4.2. Oligonucleotides
13.3.1.1.4.3. Nucleic Acid Probes
13.3.1.1.4.4. Purified Proteins or Peptides
13.3.1.1.4.5. Other Reagents
13.3.1.1.5. Implants
13.3.1.1.5.1. Orthopedic Implants
13.3.1.1.5.2. ENT Implants
13.3.1.1.5.3. Breast Implants
13.3.1.1.5.4. Dental Implants
13.3.1.1.5.5. Cardiology related implants
13.3.1.1.5.6. Others
13.3.1.1.6. Others
13.3.2. Argentina
13.3.2.1.
Argentina Medical Device Market Revenue (US$ Mn) and Forecasts,
By Type
13.3.2.1.1. Electronics and
Equipment
13.3.2.1.1.1. Diagnostic Imaging
13.3.2.1.1.1.1. X-Ray Equipment
13.3.2.1.1.1.2. Ultrasound Systems
13.3.2.1.1.1.3. Computed Tomography (CT) Scanners
13.3.2.1.1.1.4. Magnetic Resonance Imaging Systems
13.3.2.1.1.1.5. Others
13.3.2.1.1.2. Patient Monitoring
13.3.2.1.1.2.1. Cardiac Monitoring Devices
13.3.2.1.1.2.2. Respiratory Monitoring Devices
13.3.2.1.1.2.3. Others
13.3.2.1.1.3. Diabetic Care Devices
13.3.2.1.1.3.1. Insulin Pens
13.3.2.1.1.3.2. Blood Glucose Meters
13.3.2.1.1.3.3. Blood Glucose Test Strips
13.3.2.1.1.3.4. Continuous Glucose Monitoring Device
13.3.2.1.1.3.5. Others
13.3.2.1.1.4. Orthopedic Devices
13.3.2.1.1.4.1. Prosthetics
13.3.2.1.1.4.2. Braces
13.3.2.1.1.4.3. Fixation Devices
13.3.2.1.1.4.4. Others
13.3.2.1.1.5. Dental Diagnostics and Operative Devices
13.3.2.1.1.6. Others
13.3.2.1.2. Disposables and Consumables – Highest CAGR
13.3.2.1.2.1. Wound Management Products
13.3.2.1.2.2. Drug Delivery Products
13.3.2.1.2.3. Diagnostic and
Laboratory Disposables
13.3.2.1.2.4. Dialysis Disposables
13.3.2.1.2.5. Incontinence Products
13.3.2.1.2.6. Respiratory Supplies
13.3.2.1.2.7. Sterilization Supplies
13.3.2.1.2.8. Non-woven Disposables
13.3.2.1.2.9. Disposable Masks
13.3.2.1.2.10. Disposable Eye Gear
13.3.2.1.2.11. Disposable Gloves
13.3.2.1.2.12. Hand Sanitizers
13.3.2.1.2.12.1. Gel Sanitizers
13.3.2.1.2.12.2. Foam Sanitizers
13.3.2.1.2.12.3. Liquid Sanitizers
13.3.2.1.2.12.4. Other Sanitizers
13.3.2.1.2.13. Others
13.3.2.1.3. Surgical Instruments
13.3.2.1.3.1. Surgical Sutures and Staplers
13.3.2.1.3.2. Handheld Surgical Devices
13.3.2.1.3.2.1. Forceps and Spatulas
13.3.2.1.3.2.2. Retractors
13.3.2.1.3.2.3. Dilators
13.3.2.1.3.2.4. Graspers
13.3.2.1.3.2.5. Auxiliary Instruments
13.3.2.1.3.2.6. Cutter Instruments
13.3.2.1.3.2.7. Others
13.3.2.1.3.3. Electrosurgical Devices
13.3.2.1.3.4. Others
13.3.2.1.4. IVD Reagents
13.3.2.1.4.1. Antibodies
13.3.2.1.4.1.1. Monoclonal
13.3.2.1.4.1.2. Polyclonal
13.3.2.1.4.2. Oligonucleotides
13.3.2.1.4.3. Nucleic Acid Probes
13.3.2.1.4.4. Purified Proteins or Peptides
13.3.2.1.4.5. Other Reagents
13.3.2.1.5. Implants
13.3.2.1.5.1. Orthopedic Implants
13.3.2.1.5.2. ENT Implants
13.3.2.1.5.3. Breast Implants
13.3.2.1.5.4. Dental Implants
13.3.2.1.5.5. Cardiology related implants
13.3.2.1.5.6. Others
13.3.2.1.6. Others
13.3.3. Rest of Latin America
13.3.3.1.
Rest of Latin America
Medical Device Market Revenue (US$ Mn)
and Forecasts, By Type
13.3.3.1.1. Electronics and
Equipment
13.3.3.1.1.1. Diagnostic Imaging
13.3.3.1.1.1.1. X-Ray Equipment
13.3.3.1.1.1.2. Ultrasound Systems
13.3.3.1.1.1.3. Computed Tomography (CT) Scanners
13.3.3.1.1.1.4. Magnetic Resonance Imaging Systems
13.3.3.1.1.1.5. Others
13.3.3.1.1.2. Patient Monitoring
13.3.3.1.1.2.1. Cardiac Monitoring Devices
13.3.3.1.1.2.2. Respiratory Monitoring Devices
13.3.3.1.1.2.3. Others
13.3.3.1.1.3. Diabetic Care Devices
13.3.3.1.1.3.1. Insulin Pens
13.3.3.1.1.3.2. Blood Glucose Meters
13.3.3.1.1.3.3. Blood Glucose Test Strips
13.3.3.1.1.3.4. Continuous Glucose Monitoring Device
13.3.3.1.1.3.5. Others
13.3.3.1.1.4. Orthopedic Devices
13.3.3.1.1.4.1. Prosthetics
13.3.3.1.1.4.2. Braces
13.3.3.1.1.4.3. Fixation Devices
13.3.3.1.1.4.4. Others
13.3.3.1.1.5. Dental Diagnostics and Operative Devices
13.3.3.1.1.6. Others
13.3.3.1.2. Disposables and Consumables – Highest CAGR
13.3.3.1.2.1. Wound Management Products
13.3.3.1.2.2. Drug Delivery Products
13.3.3.1.2.3. Diagnostic and Laboratory
Disposables
13.3.3.1.2.4. Dialysis Disposables
13.3.3.1.2.5. Incontinence Products
13.3.3.1.2.6. Respiratory Supplies
13.3.3.1.2.7. Sterilization Supplies
13.3.3.1.2.8. Non-woven Disposables
13.3.3.1.2.9. Disposable Masks
13.3.3.1.2.10. Disposable Eye Gear
13.3.3.1.2.11. Disposable Gloves
13.3.3.1.2.12. Hand Sanitizers
13.3.3.1.2.12.1. Gel Sanitizers
13.3.3.1.2.12.2. Foam Sanitizers
13.3.3.1.2.12.3. Liquid Sanitizers
13.3.3.1.2.12.4. Other Sanitizers
13.3.3.1.2.13. Others
13.3.3.1.3. Surgical Instruments
13.3.3.1.3.1. Surgical Sutures and Staplers
13.3.3.1.3.2. Handheld Surgical Devices
13.3.3.1.3.2.1. Forceps and Spatulas
13.3.3.1.3.2.2. Retractors
13.3.3.1.3.2.3. Dilators
13.3.3.1.3.2.4. Graspers
13.3.3.1.3.2.5. Auxiliary Instruments
13.3.3.1.3.2.6. Cutter Instruments
13.3.3.1.3.2.7. Others
13.3.3.1.3.3. Electrosurgical Devices
13.3.3.1.3.4. Others
13.3.3.1.4. IVD Reagents
13.3.3.1.4.1. Antibodies
13.3.3.1.4.1.1. Monoclonal
13.3.3.1.4.1.2. Polyclonal
13.3.3.1.4.2. Oligonucleotides
13.3.3.1.4.3. Nucleic Acid Probes
13.3.3.1.4.4. Purified Proteins or Peptides
13.3.3.1.4.5. Other Reagents
13.3.3.1.5. Implants
13.3.3.1.5.1. Orthopedic Implants
13.3.3.1.5.2. ENT Implants
13.3.3.1.5.3. Breast Implants
13.3.3.1.5.4. Dental Implants
13.3.3.1.5.5. Cardiology related implants
13.3.3.1.5.6. Others
13.3.3.1.6. Others
13.4.
Key Segment for Channeling Investments
13.4.1. By Country
13.4.2. By Type
14. Competitive Benchmarking
14.1.
Market Share Analysis, 2019
14.2.
Global Presence and Growth Strategies
14.2.1. Mergers and Acquisitions
14.2.2. Product Launches
14.2.3. Investments Trends
14.2.4. R&D Initiatives
15. Player Profiles
15.1. Beckton
Dickinson & Company
15.1.1. Company Details
15.1.2. Company Overview
15.1.3. Product Offerings
15.1.4. Key Developments
15.1.5. Financial Analysis
15.1.6. SWOT Analysis
15.1.7.
Business Strategies
15.2.
Siemens
Healthineers
15.2.1. Company Details
15.2.2. Company Overview
15.2.3. Product Offerings
15.2.4. Key Developments
15.2.5. Financial Analysis
15.2.6. SWOT Analysis
15.2.7.
Business Strategies
15.3. Fresenius
Medical Care
15.3.1. Company Details
15.3.2. Company Overview
15.3.3. Product Offerings
15.3.4. Key Developments
15.3.5. Financial Analysis
15.3.6. SWOT Analysis
15.3.7.
Business Strategies
15.4. Philips
15.4.1. Company Details
15.4.2. Company Overview
15.4.3. Product Offerings
15.4.4. Key Developments
15.4.5. Financial Analysis
15.4.6. SWOT Analysis
15.4.7.
Business Strategies
15.5.
Abbott
15.5.1. Company Details
15.5.2. Company Overview
15.5.3. Product Offerings
15.5.4. Key Developments
15.5.5. Financial Analysis
15.5.6. SWOT Analysis
15.5.7.
Business Strategies
15.6.
Thermo Fisher
Scientific
15.6.1. Company Details
15.6.2. Company Overview
15.6.3. Product Offerings
15.6.4. Key Developments
15.6.5. Financial Analysis
15.6.6. SWOT Analysis
15.6.7.
Business Strategies
15.7.
Johnson &
Johnson
15.7.1. Company Details
15.7.2. Company Overview
15.7.3. Product Offerings
15.7.4. Key Developments
15.7.5. Financial Analysis
15.7.6. SWOT Analysis
15.7.7.
Business Strategies
15.8. Medtronic
15.8.1. Company Details
15.8.2. Company Overview
15.8.3. Product Offerings
15.8.4. Key Developments
15.8.5. Financial Analysis
15.8.6. SWOT Analysis
15.8.7.
Business Strategies
15.9. General
Electric Company
15.9.1. Company Details
15.9.2. Company Overview
15.9.3. Product Offerings
15.9.4. Key Developments
15.9.5. Financial Analysis
15.9.6. SWOT Analysis
15.9.7.
Business Strategies
15.10. Fresenius
Medical Care AG & Co. KGaA
15.10.1. Company Details
15.10.2. Company Overview
15.10.3. Product Offerings
15.10.4. Key Developments
15.10.5. Financial Analysis
15.10.6. SWOT Analysis
15.10.7. Business Strategies
15.11.
Other Market Participants
16. Key Findings
Note: This ToC is tentative and can
be changed according to the research study conducted during the course of report
completion.
**Exclusive for Multi-User and
Enterprise User.
At Absolute Markets Insights, we are engaged in building both global as well as country specific reports. As a result, the approach taken for deriving the estimation and forecast for a specific country is a bit unique and different in comparison to the global research studies. In this case, we not only study the concerned market factors & trends prevailing in a particular country (from secondary research) but we also tend to calculate the actual market size & forecast from the revenue generated from the market participants involved in manufacturing or distributing the any concerned product. These companies can also be service providers. For analyzing any country specifically, we do consider the growth factors prevailing under the states/cities/county for the same. For instance, if we are analyzing an industry specific to United States, we primarily need to study about the states present under the same(where the product/service has the highest growth). Similar analysis will be followed by other countries. Our scope of the report changes with different markets.
Our research study is mainly implement through a mix of both secondary and primary research. Various sources such as industry magazines, trade journals, and government websites and trade associations are reviewed for gathering precise data. Primary interviews are conducted to validate the market size derived from secondary research. Industry experts, major manufacturers and distributors are contacted for further validation purpose on the current market penetration and growth trends.
Prominent participants in our primary research process include:
- Key Opinion Leaders namely the CEOs, CSOs, VPs, purchasing managers, amongst others
- Research and development participants, distributors/suppliers and subject matter experts
Secondary Research includes data extracted from paid data sources:
- Reuters
- Factiva
- Bloomberg
- One Source
- Hoovers
Research Methodology
Key Inclusions

Reach to us
Call us on
+91-74002-42424
Drop us an email at
sales@absolutemarketsinsights.com
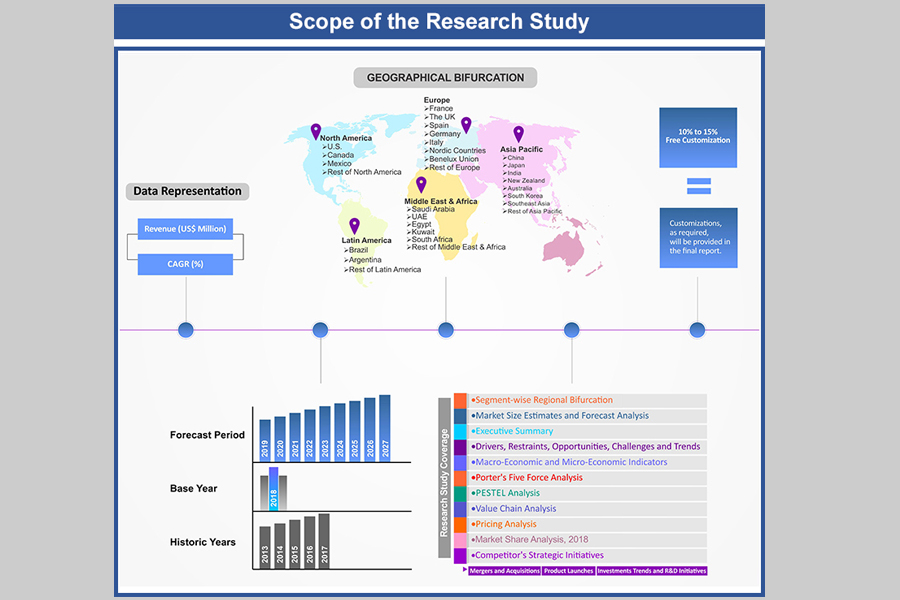
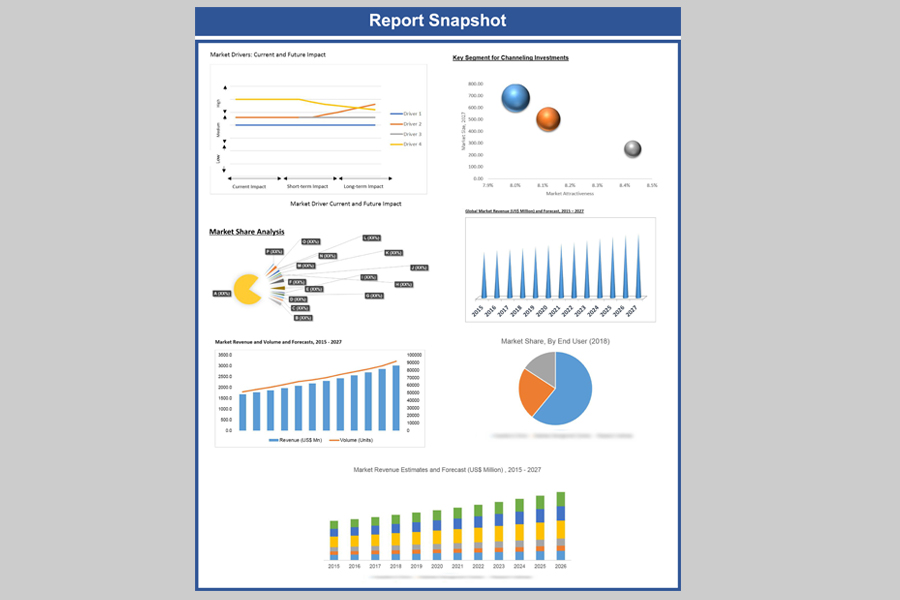
Why Absolute Markets Insights?
An effective strategy is the entity that influences a business to stand out of the crowd. An organization with a phenomenal strategy for success dependably has the edge over the rivals in the market. It offers the organizations a head start in planning their strategy. Absolute Market Insights is the new initiation in the industry that will furnish you with the lead your business needs. Absolute Market Insights is the best destination for your business intelligence and analytical solutions; essentially because our qualitative and quantitative sources of information are competent to give one-stop solutions. We inventively combine qualitative and quantitative research in accurate proportions to have the best report, which not only gives the most recent insights but also assists you to grow.