Pharmaceutical Quality Control Market By Application (Allergens, antigens, vaccines & hormones, Cytokines, enzymes & human whole-blood, Plasma derivative, Immune sera, immunoglobulins & products of fermentation and Others (diagnostic agents for in vitro use, etc.)); By Offering (Consumables & Instruments, Solutions, & Services); By Techniques (GC/MS, LC/MS/ High Performance Liquid (HPLC), Ultra High Performance Liquid (UHPLC), Supercritical Fluid (SFC) chromatography, Elemental analysis techniques, & Others)); By Testing Categories (Elemental Impurities, Volatile Organic Impurities, Extractables & Leachables, Semi-Volatile Organic Impurities, Non-Volatile Organic Impurities, Counterion Analysis, & Others); By Analysis Type (Pharmaceutical Microbiology (Sterility Testing, Antimicrobial Efficacy Testing (AET), Microbial Limits Testing, Bioburden Determination, Endotoxin Testing, Environmental Monitoring & Identification, Packaging Testing, Pharmaceutical & Biopharmaceutical Release Testing, Pharmaceutical Gas Testing, Viral Safety Testing and Lot Release Testing and Others (Water Analysis etc.) and Pharmaceutical Chemistry (Elemental Impurities, Volatile Organic Impurities, Extractables & Leachables, Semi-Volatile Organic Impurities, Non-Volatile Organic Impurities, Counterion Analysis, & Others)); By Region (North America (U.S., Canada, Mexico, & Rest Of North America), Europe (France, The UK, Spain, Germany, Italy, Nordic Countries (Denmark, Finland, Iceland, Sweden & Norway), Benelux Union (Belgium, The Netherlands, Luxembourg), & Rest of Europe), Asia Pacific (China, Japan, India, New Zealand, Australia, South Korea, Southeast Asia (Indonesia, Thailand, Malaysia, Singapore & Rest of Southeast Asia), & Rest of Asia Pacific), Middle East & Africa (Saudi Arabia, UAE, Egypt, Kuwait, South Africa, & Rest of Middle East & Africa) Latin America (Brazil, Argentina, & Rest of Latin America)) - Global Insights, Growth, Size, Comparative Analysis, Trends and Forecast, 2021 – 2029
Industry Trends
In the global pharmaceutical quality control market, the major drivers contributing to the market growth are focus on the efficiency of pharmaceutical medicines, availability of better storage facilities, regulatory changes to simplify the process of obtaining permits related to medicine manufacturing and quality control for the same, and new therapeutic applications that aid the production of new medicines amongst others.
Concerns regarding supply chain is restraining the growth of global pharmaceutical quality control market. Disruptions in manufacturing processes tend to disrupt the market growth due to logistical issues and regulatory issues.
At every stage of drug development, manufacturers along with pharmaceutical experts explore workflow solutions and scientific choices in order to ensure quality of pharmaceutical drugs. Quality documentation assistance to facilitate accreditation and audits is also necessary for this purpose. From the early stage of drug development, complying with international specifications for quality control and pharmaceutical analysis is important. All these things are integral to pharmaceutical quality control.
Due to the China-originated coronavirus pandemic, global biopharma supply chain felt the need to speed up research to produce a cure. As the commonly used supplies in biopharma manufacturing such as surgical masks, testing swabs, and alcohol-based disinfectants faced increasing demand and depleted inventory due to reduction in production caused by lack of raw materials, many companies sought alternative suppliers so that at least standard operations could be maintained. Owing to the rapid spread of the infectious and chronic diseases, production of pharmaceuticals and biopharmaceuticals have been accelerated and the need for quality control has increased to ensure the effectiveness of medicines and vaccines. The pharmaceutical companies are striving to speed up their operations, supply chains, and manufacturing capabilities, along with investments in research to develop vaccines, which showcases significant growth opportunities for the global pharmaceutical quality control market over the future years.
Technological advancements in pharma quality control laboratories in the form of adoption of digital technology and automation is anticipated to contribute towards the growth of the global pharmaceutical quality control market over the forecast period. The emerging technologies range from internet connectivity to advanced analytics, robotics and automation. They are expected to boost productivity by 30% to 40% within already mature and efficient lab environments. With a new range of improvements, there can be possibility of over 50% reductions in overall quality control costs. By reducing manual errors and variability, digitization and automation can also ensure better quality and compliance, allowing faster as well as effective resolution of problems.
In terms of revenue, global pharmaceutical quality control market was valued at US$ 3481.94 Mn in 2020 growing at a CAGR of 13.85% over the forecast period (2021 – 2029). The study analyses the market in terms of revenue across all the major regions, which have been bifurcated into countries.
Global Pharmaceutical Quality Control Market Revenue & Forecast, (US$ Million), 2015 – 2029
Application Outlook:
As per the application, the global pharmaceutical quality control market is segmented into allergens, antigens, vaccines & hormones, cytokines, enzymes & human whole-blood, plasma derivative, immune sera, immunoglobulins & products of fermentation, and others. Allergens, antigens, vaccines and hormones segment accounted for the highest share in the global pharmaceutical quality control market in 2020.
During the forecast period, the plasma derivative segment is anticipated to witness highest growth rate segment. Plasma derivatives are defined as concentrates of specific plasma proteins that are prepared from pools (many donor units) of plasma. Due to high prevalence of life-threatening ailments such as immunodeficiency diseases and hemophilia, pharmaceutical companies are increasingly investing in the production, quality check, and supply of plasma derivative. For instance, in May 2021 Liminal BioSciences entered into an agreement with Kedrion, to divest its plasma collection and plasma-derived therapeutics business.
Offering Outlook:
In the context of offering, the global pharmaceutical quality control market has been primarily segmented into consumables & instruments, solutions, and services. Among these, consumables & instruments accounted for the highest market share in 2020. Consumables are defined as the key materials that are used in support of manufacturing. The term "consumables" can also be used for materials that come in contact with the product during manufacturing that may not become part of the final product. The three categories of consumables are known as direct quality impacting consumables, indirect quality impacting consumables, and non-quality impacting consumables. Instruments are defined as a wide variety of equipment, machinery, and products. Each instrument is important in the unique step in the manufacturing process. In 2021, the use of Lyophilized beads (Lyobeads) technology increased in the pharma and diagnostics sectors. Lyobeads are defined as spheres of customizable lyophilized material. They contain a defined volume of material per unit. Services segment is expected to achieve tremendous growth in the global pharmaceutical quality control market during the forecast period.
Techniques Outlook:
Based on techniques, the global pharmaceutical quality control market has been segmented into GC/MS, LC/MS/ high performance liquid (HPLC), ultra high performance liquid (UHPLC), supercritical fluid (SFC) chromatography, elemental analysis techniques, and others. GC/MS accounted for the highest market share in the global pharmaceutical quality control market in 2020. During the forecast period (2021 – 2029), supercritical fluid (SFC) chromatography segment is expected to register highest CAGR. The less expensive procedure of SFC over gas chromatography is expected to lead the market growth for SFC chromatography during the forecast period as the former is cost effective. In March 2021, PerkinElmer Inc. acquired ES Industries, based in New Jersey, U.S., adding state-of-the-art columns and chemistries of the latter to its LC consumables portfolio.
Testing Categories Outlook:
Considering the testing categories, the global pharmaceutical quality control market has been segmented into elemental impurities, volatile organic impurities, extractables & leachables, semi-volatile organic impurities, non-volatile organic impurities, counterion analysis, and others.
Extractable & leachable testing segment was the largest market segment in 2020. Extractable analysis identifies the substances that can potentially migrate from metallic, polymeric, or glass material into the patient. Such substances include catalysts, dyes, and plasticizers. Extractables are defined as organic chemical species and inorganic chemical species that can be released from the surfaces of components used in the manufacture and storage of drug products under laboratory conditions, such as accelerated temperature or exaggerated temperature, solvents or surface exposure. The leachable analysis identifies substances that migrate from metallic, polymeric, or glass material into the patient and are typically a subset of those identified in the extractable analysis. This type of analysis is required when there is a risk that harmful substances may have leached into a liquid product, such as eye drops, from its container or packaging.
Analysis Type Outlook:
Based on analysis type, the global pharmaceutical quality control market has been segmented into pharmaceutical microbiology and pharmaceutical chemistry. Pharmaceutical microbiology segment has been further segmented into sterility testing, antimicrobial efficacy testing (AET), microbial limits testing, bioburden determination, endotoxin testing, environmental monitoring & identification, packaging testing, pharmaceutical & biopharmaceutical release testing, pharmaceutical gas testing, viral safety testing & lot release testing and others (water analysis etc.). Pharmaceutical chemistry segment has been further segmented into elemental impurities, volatile organic impurities, extractables & leachables, semi-volatile organic impurities, non-volatile organic impurities, counterion analysis, and others.
Pharmaceutical microbiology segment accounted for the highest market share in global pharmaceutical quality control market in 2020 and during the forecast period, this segment is expected to witness fastest growth rate. Sterility testing is a good manufacturing practice (GMP) microbiology testing requirement for sterile pharmaceuticals, medical devices, and materials and is essential to ensure that they are safe for use. The most common ways of sterility testing include membrane filtration and direct inoculation. Membrane filtration process is defined as a physical separation method that is characterized by the ability to separate molecules of different sizes and characteristics. This process involves the use of membrane technology to separate biomolecules and particles for the concentration of process fluids. In direct inoculation method, test articles are inoculated directly into tubes or bottles containing an appropriate medium before the articles are incubated for 14 days. With sterility testing being one of their specialties, Precision NanoSystems announced their plans to expand global operations and headquarters in August 2021.
Region Outlook:
Based on the regions, North America accounted for a significant share in the global pharmaceutical quality control market in 2020. However, during the forecast period (2021 – 2029), the Asia Pacific region is expected to witness highest growth rate because a large variation in pharmaceutical spending has been observed in this region in past few years. Governments in Asian countries like China, India, Singapore, South Korea, and Japan are supporting the growth of medical sector and thus, investment is increasing in the pharmaceutical sector with emphasis on quality control. In countries like Singapore, rules about the quality of pharmaceutical medicines are strict. Growing medical sector in Asian countries is also expected to drive the market growth of this region over the period of next eight years. In 2020, the Ministry of Health and Welfare (MOHW) in Taiwan approved 355 applications related to Investigational New Drug (IND). 78% of these applications were about multinational trials. Hence, Taiwan is growing as a regional hub for new drug discovery in the Asia Pacific region and procedures of quality checks and quality control are also rapidly progressing in the country.
Competitive Landscape Outlook:
The report provides both, qualitative as well as quantitative research of global pharmaceutical quality control market. It also provides comprehensive insights and development methods adopted by the key contenders. The report also offers extensive research on the key players in this market and details on the competitiveness of these players. Key business strategies such as affiliations, collaborations, mergers & acquisitions (M&A), and contracts adopted by these major market participants are also recognized and analyzed in the report. For each company, the report studies their global presence, competitors, service offerings and specification amongst others.
Some of the players operating in the pharmaceutical quality control market are Lucideon Limited, ThermoFisher Scientific, Merck KGaA, Charles River Laboratories, Eurofins Scientific, SGS Group Management SA, Toxikon Corporation, and Sartorius AG, amongst others.
Global Pharmaceutical Quality Control Market:
- By Application
- Allergens, antigens, vaccines and hormones
- Cytokines, enzymes and human whole-blood
- Plasma derivative
- Immune sera, immunoglobulins and products of fermentation
- Others (diagnostic agents for in vitro use, etc.)
- By Offering
- Consumables and Instruments
- Solutions
- Services
- By Techniques
- GC/MS
- LC/MS/ High Performance Liquid (HPLC)
- Ultra High Performance Liquid (UHPLC)
- Supercritical Fluid (SFC) chromatography
- Elemental analysis techniques
- Others
- By Testing Categories
- Elemental Impurities
- Volatile Organic Impurities
- Extractables and Leachables
- Semi-Volatile Organic Impurites
- Non-Volatile Organic Impurites
- Counterion Analysis
- Others
- By Analysis Type
- Pharmaceutical Microbiology
- Sterility Testing
- Antimicrobial Efficacy Testing (AET)
- Microbial Limits Testing
- Bioburden Determination
- Endotoxin Testing
- Environmental Monitoring and Identification
- Packaging Testing
- Pharmaceutical and Biopharmaceutical Release Testing
- Pharmaceutical Gas Testing
- Viral Safety Testing and Lot Release Testing
- Others (Water Analysis and etc.)
- Pharmaceutical Chemistry
- Elemental Impurities
- Volatile Organic Impurities
- Extractables and Leachables
- Semi-Volatile Organic Impurites
- Non-Volatile Organic Impurites
- Counterion Analysis
- Others
- By Region
- North America
- U.S.
- Canada
- Mexico
- Rest of North America
- Europe
- France
- The UK
- Spain
- Germany
- Italy
- Nordic Countries
- Denmark
- Finland
- Iceland
- Sweden
- Norway
- Benelux Union
- Belgium
- The Netherlands
- Luxembourg
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- New Zealand
- Australia
- South Korea
- Southeast Asia
- Indonesia
- Thailand
- Malaysia
- Singapore
- Rest of Southeast Asia
- Rest of Asia Pacific
- Middle East & Africa
- Saudi Arabia
- UAE
- Egypt
- Kuwait
- South Africa
- Rest of Middle East & Africa
- Latin America
- Brazil
- Argentina
- Rest of Latin America
Table of Contents
1. Market Scope
1.1. Market Segmentation
1.2. Years Considered
1.2.1. Historic Years: 2015 - 2019
1.2.2. Base Year: 2020
1.2.3. Forecast Years: 2021 – 2029
2. Key Target Audiences
3. Research Methodology
3.1. Primary Research
3.1.1. Research Questionnaire
3.1.2. Global Percentage Breakdown
3.1.3. Primary Interviews: Key Opinion Leaders (KOLs)
3.2. Secondary Research
3.2.1. Paid Databases
3.2.2. Secondary Sources
3.3. Market Size Estimates
3.3.1. Top-Down Approach
3.3.2. Bottom-Up Approach
3.4. Data Triangulation Methodology
3.5. Research Assumptions
4. Recommendations and Insights from AMI’s Perspective**
5. Holistic Overview of Pharmaceutical Quality Control Market
6. Market Synopsis:
Pharmaceutical Quality Control Market
7. Pharmaceutical Quality Control Market Analysis: Qualitative
Perspective
7.1. Introduction
7.1.1. Product Definition
7.1.2. Industry Development
7.2. Market Dynamics
7.2.1. Drivers
7.2.2. Restraints
7.2.3. Opportunities
7.3. Trends in Pharmaceutical Quality Control Market
7.4. Market Determinants Radar Chart
7.5. Macro-Economic and Micro-Economic Indicators: Pharmaceutical
Quality Control Market
7.6. Porter’s Five Force Analysis
7.7. Impact of Covid-19 on Pharmaceutical Quality Control Market
8. Global Pharmaceutical Quality Control Market Analysis and
Forecasts, 2021 – 2029
8.1. Overview
8.1.1. Global Pharmaceutical Quality Control Market Revenue (US$ Mn)
8.2. Global Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Application
8.2.1. Allergens, antigens, vaccines and hormones
8.2.1.1. Definition
8.2.1.2. Market Estimation and Penetration, 2015 - 2020
8.2.1.3. Market Forecast, 2021 - 2029
8.2.1.4. Compound Annual Growth Rate (CAGR)
8.2.1.5. Regional Bifurcation
8.2.1.5.1. North America
8.2.1.5.1.1. Market
Estimation, 2015 - 2020
8.2.1.5.1.2. Market
Forecast, 2021 - 2029
8.2.1.5.2. Europe
8.2.1.5.2.1. Market
Estimation, 2015 - 2020
8.2.1.5.2.2. Market
Forecast, 2021 - 2029
8.2.1.5.3. Asia Pacific
8.2.1.5.3.1. Market
Estimation, 2015 - 2020
8.2.1.5.3.2. Market
Forecast, 2021 - 2029
8.2.1.5.4. Middle East and Africa
8.2.1.5.4.1. Market
Estimation, 2015 - 2020
8.2.1.5.4.2. Market
Forecast, 2021 - 2029
8.2.1.5.5. Latin America
8.2.1.5.5.1. Market
Estimation, 2015 - 2020
8.2.1.5.5.2. Market
Forecast, 2021 - 2029
8.2.2. Cytokines, enzymes and human whole-blood
8.2.2.1. Definition
8.2.2.2. Market Estimation and Penetration, 2015 - 2020
8.2.2.3. Market Forecast, 2021 - 2029
8.2.2.4. Compound Annual Growth Rate (CAGR)
8.2.2.5. Regional Bifurcation
8.2.2.5.1. North America
8.2.2.5.1.1. Market
Estimation, 2015 - 2020
8.2.2.5.1.2. Market
Forecast, 2021 - 2029
8.2.2.5.2. Europe
8.2.2.5.2.1. Market
Estimation, 2015 - 2020
8.2.2.5.2.2. Market
Forecast, 2021 - 2029
8.2.2.5.3. Asia Pacific
8.2.2.5.3.1. Market
Estimation, 2015 - 2020
8.2.2.5.3.2. Market
Forecast, 2021 - 2029
8.2.2.5.4. Middle East and Africa
8.2.2.5.4.1. Market
Estimation, 2015 - 2020
8.2.2.5.4.2. Market
Forecast, 2021 - 2029
8.2.2.5.5. Latin America
8.2.2.5.5.1. Market
Estimation, 2015 - 2020
8.2.2.5.5.2. Market
Forecast, 2021 - 2029
8.2.3. Plasma derivative
8.2.3.1. Definition
8.2.3.2. Market Estimation and Penetration, 2015 - 2020
8.2.3.3. Market Forecast, 2021 - 2029
8.2.3.4. Compound Annual Growth Rate (CAGR)
8.2.3.5. Regional Bifurcation
8.2.3.5.1. North America
8.2.3.5.1.1. Market
Estimation, 2015 - 2020
8.2.3.5.1.2. Market
Forecast, 2021 - 2029
8.2.3.5.2. Europe
8.2.3.5.2.1. Market
Estimation, 2015 - 2020
8.2.3.5.2.2. Market
Forecast, 2021 - 2029
8.2.3.5.3. Asia Pacific
8.2.3.5.3.1. Market
Estimation, 2015 - 2020
8.2.3.5.3.2. Market
Forecast, 2021 - 2029
8.2.3.5.4. Middle East and Africa
8.2.3.5.4.1. Market
Estimation, 2015 - 2020
8.2.3.5.4.2. Market
Forecast, 2021 - 2029
8.2.3.5.5. Latin America
8.2.3.5.5.1. Market
Estimation, 2015 - 2020
8.2.3.5.5.2. Market
Forecast, 2021 - 2029
8.2.4. Immune sera, immunoglobulins and products of fermentation
8.2.4.1. Definition
8.2.4.2. Market Estimation and Penetration, 2015 - 2020
8.2.4.3. Market Forecast, 2021 - 2029
8.2.4.4. Compound Annual Growth Rate (CAGR)
8.2.4.5. Regional Bifurcation
8.2.4.5.1. North America
8.2.4.5.1.1. Market
Estimation, 2015 - 2020
8.2.4.5.1.2. Market
Forecast, 2021 - 2029
8.2.4.5.2. Europe
8.2.4.5.2.1. Market
Estimation, 2015 - 2020
8.2.4.5.2.2. Market
Forecast, 2021 - 2029
8.2.4.5.3. Asia Pacific
8.2.4.5.3.1. Market
Estimation, 2015 - 2020
8.2.4.5.3.2. Market
Forecast, 2021 - 2029
8.2.4.5.4. Middle East and Africa
8.2.4.5.4.1. Market
Estimation, 2015 - 2020
8.2.4.5.4.2. Market
Forecast, 2021 - 2029
8.2.4.5.5. Latin America
8.2.4.5.5.1. Market
Estimation, 2015 - 2020
8.2.4.5.5.2. Market
Forecast, 2021 - 2029
8.2.5. Others (diagnostic agents for in vitro use, etc.)
8.2.5.1. Definition
8.2.5.2. Market Estimation and Penetration, 2015 - 2020
8.2.5.3. Market Forecast, 2021 - 2029
8.2.5.4. Compound Annual Growth Rate (CAGR)
8.2.5.5. Regional Bifurcation
8.2.5.5.1. North America
8.2.5.5.1.1. Market
Estimation, 2015 - 2020
8.2.5.5.1.2. Market
Forecast, 2021 - 2029
8.2.5.5.2. Europe
8.2.5.5.2.1. Market
Estimation, 2015 - 2020
8.2.5.5.2.2. Market
Forecast, 2021 - 2029
8.2.5.5.3. Asia Pacific
8.2.5.5.3.1. Market
Estimation, 2015 - 2020
8.2.5.5.3.2. Market
Forecast, 2021 - 2029
8.2.5.5.4. Middle East and Africa
8.2.5.5.4.1. Market
Estimation, 2015 - 2020
8.2.5.5.4.2. Market
Forecast, 2021 - 2029
8.2.5.5.5. Latin America
8.2.5.5.5.1. Market
Estimation, 2015 - 2020
8.2.5.5.5.2. Market
Forecast, 2021 - 2029
8.3. Key Segment for Channeling Investments
8.3.1. By Application
9. Global Pharmaceutical Quality Control Market Analysis and
Forecasts, 2021 – 2029
9.1. Overview
9.2. Global Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Offering
9.2.1. Consumables and Instruments
9.2.1.1. Definition
9.2.1.2. Market Estimation and Penetration, 2015 - 2020
9.2.1.3. Market Forecast, 2021 - 2029
9.2.1.4. Compound Annual Growth Rate (CAGR)
9.2.1.5. Regional Bifurcation
9.2.1.5.1. North America
9.2.1.5.1.1. Market
Estimation, 2015 - 2020
9.2.1.5.1.2. Market
Forecast, 2021 - 2029
9.2.1.5.2. Europe
9.2.1.5.2.1. Market
Estimation, 2015 - 2020
9.2.1.5.2.2. Market
Forecast, 2021 - 2029
9.2.1.5.3. Asia Pacific
9.2.1.5.3.1. Market
Estimation, 2015 - 2020
9.2.1.5.3.2. Market
Forecast, 2021 - 2029
9.2.1.5.4. Middle East and Africa
9.2.1.5.4.1. Market
Estimation, 2015 - 2020
9.2.1.5.4.2. Market
Forecast, 2021 - 2029
9.2.1.5.5. Latin America
9.2.1.5.5.1. Market
Estimation, 2015 - 2020
9.2.1.5.5.2. Market
Forecast, 2021 - 2029
9.2.2. Solution
9.2.2.1. Definition
9.2.2.2. Market Estimation and Penetration, 2015 - 2020
9.2.2.3. Market Forecast, 2021 - 2029
9.2.2.4. Compound Annual Growth Rate (CAGR)
9.2.2.5. Regional Bifurcation
9.2.2.5.1. North America
9.2.2.5.1.1. Market
Estimation, 2015 - 2020
9.2.2.5.1.2. Market
Forecast, 2021 - 2029
9.2.2.5.2. Europe
9.2.2.5.2.1. Market
Estimation, 2015 - 2020
9.2.2.5.2.2. Market
Forecast, 2021 - 2029
9.2.2.5.3. Asia Pacific
9.2.2.5.3.1. Market
Estimation, 2015 - 2020
9.2.2.5.3.2. Market
Forecast, 2021 - 2029
9.2.2.5.4. Middle East and Africa
9.2.2.5.4.1. Market
Estimation, 2015 - 2020
9.2.2.5.4.2. Market
Forecast, 2021 - 2029
9.2.2.5.5. Latin America
9.2.2.5.5.1. Market
Estimation, 2015 - 2020
9.2.2.5.5.2. Market
Forecast, 2021 - 2029
9.2.3. Services
9.2.3.1. Definition
9.2.3.2. Market Estimation and Penetration, 2015 - 2020
9.2.3.3. Market Forecast, 2021 - 2029
9.2.3.4. Compound Annual Growth Rate (CAGR)
9.2.3.5. Regional Bifurcation
9.2.3.5.1. North America
9.2.3.5.1.1. Market
Estimation, 2015 - 2020
9.2.3.5.1.2. Market
Forecast, 2021 - 2029
9.2.3.5.2. Europe
9.2.3.5.2.1. Market
Estimation, 2015 - 2020
9.2.3.5.2.2. Market
Forecast, 2021 - 2029
9.2.3.5.3. Asia Pacific
9.2.3.5.3.1. Market
Estimation, 2015 - 2020
9.2.3.5.3.2. Market
Forecast, 2021 - 2029
9.2.3.5.4. Middle East and Africa
9.2.3.5.4.1. Market
Estimation, 2015 - 2020
9.2.3.5.4.2. Market
Forecast, 2021 - 2029
9.2.3.5.5. Latin America
9.2.3.5.5.1. Market
Estimation, 2015 - 2020
9.2.3.5.5.2. Market
Forecast, 2021 - 2029
9.3. Key Segment for Channeling Investments
9.3.1. By Offering
10. Global Pharmaceutical Quality Control Market Analysis and
Forecasts, 2021 – 2029
10.1. Overview
10.2. Global Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Techniques
10.2.1. GC/MS
10.2.1.1. Definition
10.2.1.2. Market Estimation and Penetration, 2015 - 2020
10.2.1.3. Market Forecast, 2021 - 2029
10.2.1.4. Compound Annual Growth Rate (CAGR)
10.2.1.5. Regional Bifurcation
10.2.1.5.1. North America
10.2.1.5.1.1. Market
Estimation, 2015 - 2020
10.2.1.5.1.2. Market
Forecast, 2021 - 2029
10.2.1.5.2. Europe
10.2.1.5.2.1. Market
Estimation, 2015 - 2020
10.2.1.5.2.2. Market
Forecast, 2021 - 2029
10.2.1.5.3. Asia Pacific
10.2.1.5.3.1. Market
Estimation, 2015 - 2020
10.2.1.5.3.2. Market
Forecast, 2021 - 2029
10.2.1.5.4. Middle East and Africa
10.2.1.5.4.1. Market
Estimation, 2015 - 2020
10.2.1.5.4.2. Market
Forecast, 2021 - 2029
10.2.1.5.5. Latin America
10.2.1.5.5.1. Market
Estimation, 2015 - 2020
10.2.1.5.5.2. Market
Forecast, 2021 - 2029
10.2.2. LC/MS/ High Performance
Liquid (HPLC)
10.2.2.1. Definition
10.2.2.2. Market Estimation and Penetration, 2015 - 2020
10.2.2.3. Market Forecast, 2021 - 2029
10.2.2.4. Compound Annual Growth Rate (CAGR)
10.2.2.5. Regional Bifurcation
10.2.2.5.1. North America
10.2.2.5.1.1. Market
Estimation, 2015 - 2020
10.2.2.5.1.2. Market
Forecast, 2021 - 2029
10.2.2.5.2. Europe
10.2.2.5.2.1. Market
Estimation, 2015 - 2020
10.2.2.5.2.2. Market
Forecast, 2021 - 2029
10.2.2.5.3. Asia Pacific
10.2.2.5.3.1. Market
Estimation, 2015 - 2020
10.2.2.5.3.2. Market
Forecast, 2021 - 2029
10.2.2.5.4. Middle East and Africa
10.2.2.5.4.1. Market
Estimation, 2015 - 2020
10.2.2.5.4.2. Market
Forecast, 2021 - 2029
10.2.2.5.5. Latin America
10.2.2.5.5.1. Market
Estimation, 2015 - 2020
10.2.2.5.5.2. Market
Forecast, 2021 - 2029
10.2.3. Ultra High Performance Liquid (UHPLC)
10.2.3.1. Definition
10.2.3.2. Market Estimation and Penetration, 2015 - 2020
10.2.3.3. Market Forecast, 2021 - 2029
10.2.3.4. Compound Annual Growth Rate (CAGR)
10.2.3.5. Regional Bifurcation
10.2.3.5.1. North America
10.2.3.5.1.1. Market
Estimation, 2015 - 2020
10.2.3.5.1.2. Market
Forecast, 2021 - 2029
10.2.3.5.2. Europe
10.2.3.5.2.1. Market
Estimation, 2015 - 2020
10.2.3.5.2.2. Market
Forecast, 2021 - 2029
10.2.3.5.3. Asia Pacific
10.2.3.5.3.1. Market
Estimation, 2015 - 2020
10.2.3.5.3.2. Market
Forecast, 2021 - 2029
10.2.3.5.4. Middle East and Africa
10.2.3.5.4.1. Market
Estimation, 2015 - 2020
10.2.3.5.4.2. Market
Forecast, 2021 - 2029
10.2.3.5.5. Latin America
10.2.3.5.5.1. Market
Estimation, 2015 - 2020
10.2.3.5.5.2. Market
Forecast, 2021 - 2029
10.2.4. Supercritical Fluid (SFC) chromatography
10.2.4.1. Definition
10.2.4.2. Market Estimation and Penetration, 2015 - 2020
10.2.4.3. Market Forecast, 2021 - 2029
10.2.4.4. Compound Annual Growth Rate (CAGR)
10.2.4.5. Regional Bifurcation
10.2.4.5.1. North America
10.2.4.5.1.1. Market
Estimation, 2015 - 2020
10.2.4.5.1.2. Market
Forecast, 2021 - 2029
10.2.4.5.2. Europe
10.2.4.5.2.1. Market
Estimation, 2015 - 2020
10.2.4.5.2.2. Market
Forecast, 2021 - 2029
10.2.4.5.3. Asia Pacific
10.2.4.5.3.1. Market
Estimation, 2015 - 2020
10.2.4.5.3.2. Market
Forecast, 2021 - 2029
10.2.4.5.4. Middle East and Africa
10.2.4.5.4.1. Market
Estimation, 2015 - 2020
10.2.4.5.4.2. Market
Forecast, 2021 - 2029
10.2.4.5.5. Latin America
10.2.4.5.5.1. Market
Estimation, 2015 - 2020
10.2.4.5.5.2. Market
Forecast, 2021 - 2029
10.2.5. Elemental analysis techniques
10.2.5.1. Definition
10.2.5.2. Market Estimation and Penetration, 2015 - 2020
10.2.5.3. Market Forecast, 2021 - 2029
10.2.5.4. Compound Annual Growth Rate (CAGR)
10.2.5.5. Regional Bifurcation
10.2.5.5.1. North America
10.2.5.5.1.1. Market
Estimation, 2015 - 2020
10.2.5.5.1.2. Market
Forecast, 2021 - 2029
10.2.5.5.2. Europe
10.2.5.5.2.1. Market
Estimation, 2015 - 2020
10.2.5.5.2.2. Market
Forecast, 2021 - 2029
10.2.5.5.3. Asia Pacific
10.2.5.5.3.1. Market
Estimation, 2015 - 2020
10.2.5.5.3.2. Market
Forecast, 2021 - 2029
10.2.5.5.4. Middle East and Africa
10.2.5.5.4.1. Market
Estimation, 2015 - 2020
10.2.5.5.4.2. Market
Forecast, 2021 - 2029
10.2.5.5.5. Latin America
10.2.5.5.5.1. Market
Estimation, 2015 - 2020
10.2.5.5.5.2. Market
Forecast, 2021 - 2029
10.2.6. Others
10.2.6.1. Definition
10.2.6.2. Market Estimation and Penetration, 2015 - 2020
10.2.6.3. Market Forecast, 2021 - 2029
10.2.6.4. Compound Annual Growth Rate (CAGR)
10.2.6.5. Regional Bifurcation
10.2.6.5.1. North America
10.2.6.5.1.1. Market
Estimation, 2015 - 2020
10.2.6.5.1.2. Market
Forecast, 2021 - 2029
10.2.6.5.2. Europe
10.2.6.5.2.1. Market
Estimation, 2015 - 2020
10.2.6.5.2.2. Market
Forecast, 2021 - 2029
10.2.6.5.3. Asia Pacific
10.2.6.5.3.1. Market
Estimation, 2015 - 2020
10.2.6.5.3.2. Market
Forecast, 2021 - 2029
10.2.6.5.4. Middle East and Africa
10.2.6.5.4.1. Market
Estimation, 2015 - 2020
10.2.6.5.4.2. Market
Forecast, 2021 - 2029
10.2.6.5.5. Latin America
10.2.6.5.5.1. Market
Estimation, 2015 - 2020
10.2.6.5.5.2. Market
Forecast, 2021 - 2029
10.3. Key Segment for Channeling Investments
10.3.1. By Techniques
11. Global Pharmaceutical Quality Control Market Analysis and
Forecasts, 2021 – 2029
11.1. Overview
11.2. Global Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Testing Categories
11.2.1. Elemental Impurities
11.2.1.1. Definition
11.2.1.2. Market Estimation and Penetration, 2015 - 2020
11.2.1.3. Market Forecast, 2021 - 2029
11.2.1.4. Compound Annual Growth Rate (CAGR)
11.2.1.5. Regional Bifurcation
11.2.1.5.1. North America
11.2.1.5.1.1. Market
Estimation, 2015 - 2020
11.2.1.5.1.2. Market
Forecast, 2021 - 2029
11.2.1.5.2. Europe
11.2.1.5.2.1. Market
Estimation, 2015 - 2020
11.2.1.5.2.2. Market
Forecast, 2021 - 2029
11.2.1.5.3. Asia Pacific
11.2.1.5.3.1. Market
Estimation, 2015 - 2020
11.2.1.5.3.2. Market
Forecast, 2021 - 2029
11.2.1.5.4. Middle East and Africa
11.2.1.5.4.1. Market
Estimation, 2015 - 2020
11.2.1.5.4.2. Market
Forecast, 2021 - 2029
11.2.1.5.5. Latin America
11.2.1.5.5.1. Market
Estimation, 2015 - 2020
11.2.1.5.5.2. Market
Forecast, 2021 - 2029
11.2.2. Volatile Organic Impurities
11.2.2.1. Definition
11.2.2.2. Market Estimation and Penetration, 2015 - 2020
11.2.2.3. Market Forecast, 2021 - 2029
11.2.2.4. Compound Annual Growth Rate (CAGR)
11.2.2.5. Regional Bifurcation
11.2.2.5.1. North America
11.2.2.5.1.1. Market
Estimation, 2015 - 2020
11.2.2.5.1.2. Market
Forecast, 2021 - 2029
11.2.2.5.2. Europe
11.2.2.5.2.1. Market
Estimation, 2015 - 2020
11.2.2.5.2.2. Market
Forecast, 2021 - 2029
11.2.2.5.3. Asia Pacific
11.2.2.5.3.1. Market
Estimation, 2015 - 2020
11.2.2.5.3.2. Market
Forecast, 2021 - 2029
11.2.2.5.4. Middle East and Africa
11.2.2.5.4.1. Market
Estimation, 2015 - 2020
11.2.2.5.4.2. Market
Forecast, 2021 - 2029
11.2.2.5.5. Latin America
11.2.2.5.5.1. Market
Estimation, 2015 - 2020
11.2.2.5.5.2. Market
Forecast, 2021 - 2029
11.2.3. Extractables and Leachables
11.2.3.1. Definition
11.2.3.2. Market Estimation and Penetration, 2015 - 2020
11.2.3.3. Market Forecast, 2021 - 2029
11.2.3.4. Compound Annual Growth Rate (CAGR)
11.2.3.5. Regional Bifurcation
11.2.3.5.1. North America
11.2.3.5.1.1. Market
Estimation, 2015 - 2020
11.2.3.5.1.2. Market
Forecast, 2021 - 2029
11.2.3.5.2. Europe
11.2.3.5.2.1. Market
Estimation, 2015 - 2020
11.2.3.5.2.2. Market
Forecast, 2021 - 2029
11.2.3.5.3. Asia Pacific
11.2.3.5.3.1. Market
Estimation, 2015 - 2020
11.2.3.5.3.2. Market
Forecast, 2021 - 2029
11.2.3.5.4. Middle East and Africa
11.2.3.5.4.1. Market
Estimation, 2015 - 2020
11.2.3.5.4.2. Market
Forecast, 2021 - 2029
11.2.3.5.5. Latin America
11.2.3.5.5.1. Market
Estimation, 2015 - 2020
11.2.3.5.5.2. Market
Forecast, 2021 - 2029
11.2.4. Semi-Volatile Organic Impurites
11.2.4.1. Definition
11.2.4.2. Market Estimation and Penetration, 2015 - 2020
11.2.4.3. Market Forecast, 2021 - 2029
11.2.4.4. Compound Annual Growth Rate (CAGR)
11.2.4.5. Regional Bifurcation
11.2.4.5.1. North America
11.2.4.5.1.1. Market
Estimation, 2015 - 2020
11.2.4.5.1.2. Market
Forecast, 2021 - 2029
11.2.4.5.2. Europe
11.2.4.5.2.1. Market
Estimation, 2015 - 2020
11.2.4.5.2.2. Market
Forecast, 2021 - 2029
11.2.4.5.3. Asia Pacific
11.2.4.5.3.1. Market
Estimation, 2015 - 2020
11.2.4.5.3.2. Market
Forecast, 2021 - 2029
11.2.4.5.4. Middle East and Africa
11.2.4.5.4.1. Market
Estimation, 2015 - 2020
11.2.4.5.4.2. Market
Forecast, 2021 - 2029
11.2.4.5.5. Latin America
11.2.4.5.5.1. Market
Estimation, 2015 - 2020
11.2.4.5.5.2. Market
Forecast, 2021 - 2029
11.2.5. Non-Volatile Organic Impurites
11.2.5.1. Definition
11.2.5.2. Market Estimation and Penetration, 2015 - 2020
11.2.5.3. Market Forecast, 2021 - 2029
11.2.5.4. Compound Annual Growth Rate (CAGR)
11.2.5.5. Regional Bifurcation
11.2.5.5.1. North America
11.2.5.5.1.1. Market
Estimation, 2015 - 2020
11.2.5.5.1.2. Market
Forecast, 2021 - 2029
11.2.5.5.2. Europe
11.2.5.5.2.1. Market
Estimation, 2015 - 2020
11.2.5.5.2.2. Market
Forecast, 2021 - 2029
11.2.5.5.3. Asia Pacific
11.2.5.5.3.1. Market
Estimation, 2015 - 2020
11.2.5.5.3.2. Market
Forecast, 2021 - 2029
11.2.5.5.4. Middle East and Africa
11.2.5.5.4.1. Market
Estimation, 2015 - 2020
11.2.5.5.4.2. Market
Forecast, 2021 - 2029
11.2.5.5.5. Latin America
11.2.5.5.5.1. Market
Estimation, 2015 - 2020
11.2.5.5.5.2. Market
Forecast, 2021 - 2029
11.2.6. Counterion Analysis
11.2.6.1. Definition
11.2.6.2. Market Estimation and Penetration, 2015 - 2020
11.2.6.3. Market Forecast, 2021 - 2029
11.2.6.4. Compound Annual Growth Rate (CAGR)
11.2.6.5. Regional Bifurcation
11.2.6.5.1. North America
11.2.6.5.1.1. Market
Estimation, 2015 - 2020
11.2.6.5.1.2. Market
Forecast, 2021 - 2029
11.2.6.5.2. Europe
11.2.6.5.2.1. Market
Estimation, 2015 - 2020
11.2.6.5.2.2. Market
Forecast, 2021 - 2029
11.2.6.5.3. Asia Pacific
11.2.6.5.3.1. Market
Estimation, 2015 - 2020
11.2.6.5.3.2. Market
Forecast, 2021 - 2029
11.2.6.5.4. Middle East and Africa
11.2.6.5.4.1. Market
Estimation, 2015 - 2020
11.2.6.5.4.2. Market
Forecast, 2021 - 2029
11.2.6.5.5. Latin America
11.2.6.5.5.1. Market
Estimation, 2015 - 2020
11.2.6.5.5.2. Market
Forecast, 2021 - 2029
11.2.7. Others
11.2.7.1. Definition
11.2.7.2. Market Estimation and Penetration, 2015 - 2020
11.2.7.3. Market Forecast, 2021 - 2029
11.2.7.4. Compound Annual Growth Rate (CAGR)
11.2.7.5. Regional Bifurcation
11.2.7.5.1. North America
11.2.7.5.1.1. Market
Estimation, 2015 - 2020
11.2.7.5.1.2. Market
Forecast, 2021 - 2029
11.2.7.5.2. Europe
11.2.7.5.2.1. Market
Estimation, 2015 - 2020
11.2.7.5.2.2. Market
Forecast, 2021 - 2029
11.2.7.5.3. Asia Pacific
11.2.7.5.3.1. Market
Estimation, 2015 - 2020
11.2.7.5.3.2. Market
Forecast, 2021 - 2029
11.2.7.5.4. Middle East and Africa
11.2.7.5.4.1. Market
Estimation, 2015 - 2020
11.2.7.5.4.2. Market
Forecast, 2021 - 2029
11.2.7.5.5. Latin America
11.2.7.5.5.1. Market
Estimation, 2015 - 2020
11.2.7.5.5.2. Market
Forecast, 2021 - 2029
11.3. Key Segment for Channeling Investments
11.3.1. By Testing Categories
12. Global Pharmaceutical Quality Control Market Analysis and
Forecasts, 2021 – 2029
12.1. Overview
12.2. Global Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Analysis Type
12.2.1. Pharmaceutical Microbiology (Definition, Market Estimation and
Penetration, 2015 - 2020, Market Estimation (2015 - 2020), Market Forecast
(2021 - 2029), Compound Annual Growth Rate (CAGR), Regional Bifurcation (North
America, Europe, Asia Pacific, Middle East and Africa, Latin America) and
Information on Sterility Testing, Antimicrobial Efficacy Testing (AET),
Microbial Limits Testing, Bioburden Determination, Endotoxin Testing,
Environmental Monitoring and Identification, Packaging Testing, Pharmaceutical
and Biopharmaceutical Release Testing,
Pharmaceutical Gas Testing, Viral Safety Testing and Lot Release Testing,
Others (Water Analysis and etc.))
12.2.1.1. Sterility Testing
12.2.1.2. Antimicrobial Efficacy Testing (AET)
12.2.1.3. Microbial Limits Testing
12.2.1.4. Bioburden Determination
12.2.1.5. Endotoxin Testing
12.2.1.6. Environmental Monitoring and Identification
12.2.1.7. Packaging Testing
12.2.1.8. Pharmaceutical and Biopharmaceutical Release Testing
12.2.1.9. Pharmaceutical Gas Testing
12.2.1.10.
Viral Safety Testing and Lot
Release Testing
12.2.1.11.
Others (Water Analysis and etc.)
12.2.2. Pharmaceutical Chemistry (Definition, Market Estimation and
Penetration, 2015 - 2020, Market Estimation (2015 - 2020), Market Forecast
(2021 - 2029), Compound Annual Growth Rate (CAGR), Regional Bifurcation (North
America, Europe, Asia Pacific, Middle East and Africa, Latin America) and
Information on Elemental Impurities, Volatile Organic Impurities, Extractables
and Leachables , Semi-Volatile Organic Impurites, Non-Volatile Organic
Impurites, Counterion Analysis , Others)
12.2.2.1. Elemental Impurities
12.2.2.2. Volatile Organic Impurities
12.2.2.3. Extractables and Leachables
12.2.2.4. Semi-Volatile Organic Impurites
12.2.2.5. Non-Volatile Organic Impurites
12.2.2.6. Counterion Analysis
12.2.2.7. Others
12.3. Key Segment for Channeling Investments
12.3.1. By Analysis Type
13. North America Pharmaceutical Quality Control Market Analysis
and Forecasts, 2021 - 2029
13.1. Overview
13.1.1. North America Pharmaceutical Quality Control Market Revenue
(US$ Mn)
13.2. North America Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Application
13.2.1. Allergens, antigens, vaccines and hormones
13.2.2. Cytokines, enzymes and human whole-blood
13.2.3. Plasma derivative
13.2.4. Immune sera, immunoglobulins and products of fermentation
13.2.5. Others (diagnostic agents for in vitro use, etc.)
13.3. North America Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Offering
13.3.1. Consumables and Instruments
13.3.2. Solution
13.3.3. Services
13.4. North America Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Techniques
13.4.1. GC/MS
13.4.2. LC/MS/ High Performance
Liquid (HPLC)
13.4.3. Ultra High Performance Liquid (UHPLC)
13.4.4. Supercritical Fluid (SFC) chromatography
13.4.5. Elemental analysis techniques
13.4.6. Others
13.5. North America Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Testing Categories
13.5.1. Elemental Impurities
13.5.2. Volatile Organic Impurities
13.5.3. Extractables and Leachables
13.5.4. Semi-Volatile Organic Impurites
13.5.5. Non-Volatile Organic Impurites
13.5.6. Counterion Analysis
13.5.7. Others
13.6. North America Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Analysis Type
13.6.1. Pharmaceutical Microbiology
13.6.1.1. Sterility Testing
13.6.1.2. Antimicrobial Efficacy Testing (AET)
13.6.1.3. Microbial Limits Testing
13.6.1.4. Bioburden Determination
13.6.1.5. Endotoxin Testing
13.6.1.6. Environmental Monitoring and Identification
13.6.1.7. Packaging Testing
13.6.1.8. Pharmaceutical and Biopharmaceutical Release Testing
13.6.1.9. Pharmaceutical Gas Testing
13.6.1.10.
Viral Safety Testing and Lot
Release Testing
13.6.1.11.
Others (Water Analysis and etc.)
13.6.2. Pharmaceutical Chemistry
13.6.2.1. Elemental Impurities
13.6.2.2. Volatile Organic Impurities
13.6.2.3. Extractables and Leachables
13.6.2.4. Semi-Volatile Organic Impurites
13.6.2.5. Non-Volatile Organic Impurites
13.6.2.6. Counterion Analysis
13.6.2.7. Others
13.7. North America Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Country
13.7.1. U.S
13.7.1.1. U.S Pharmaceutical Quality Control Market Revenue (US$ Mn) and
Forecasts, By Application
13.7.1.1.1. Allergens, antigens, vaccines and hormones
13.7.1.1.2. Cytokines, enzymes and human whole-blood
13.7.1.1.3. Plasma derivative
13.7.1.1.4. Immune sera, immunoglobulins and products of fermentation
13.7.1.1.5. Others (diagnostic agents for in vitro use, etc.)
13.7.1.2. U.S Pharmaceutical Quality Control Market Revenue (US$ Mn) and
Forecasts, By Offering
13.7.1.2.1. Consumables and Instruments
13.7.1.2.2. Solution
13.7.1.2.3. Services
13.7.1.3. U.S Pharmaceutical Quality Control Market Revenue (US$ Mn) and
Forecasts, By Techniques
13.7.1.3.1. GC/MS
13.7.1.3.2. LC/MS/ High Performance
Liquid (HPLC)
13.7.1.3.3. Ultra High Performance Liquid (UHPLC)
13.7.1.3.4. Supercritical Fluid (SFC) chromatography
13.7.1.3.5. Elemental analysis techniques
13.7.1.3.6. Others
13.7.1.4. U.S Pharmaceutical Quality Control Market Revenue (US$ Mn) and
Forecasts, By Testing Categories
13.7.1.4.1. Elemental Impurities
13.7.1.4.2. Volatile Organic Impurities
13.7.1.4.3. Extractables and Leachables
13.7.1.4.4. Semi-Volatile Organic Impurites
13.7.1.4.5. Non-Volatile Organic Impurites
13.7.1.4.6. Counterion Analysis
13.7.1.4.7. Others
13.7.1.5. U.S Pharmaceutical Quality Control Market Revenue (US$ Mn) and
Forecasts, By Analysis Type
13.7.1.5.1. Pharmaceutical Microbiology
13.7.1.5.1.1. Sterility
Testing
13.7.1.5.1.2. Antimicrobial
Efficacy Testing (AET)
13.7.1.5.1.3. Microbial
Limits Testing
13.7.1.5.1.4. Bioburden
Determination
13.7.1.5.1.5. Endotoxin
Testing
13.7.1.5.1.6. Environmental
Monitoring and Identification
13.7.1.5.1.7. Packaging
Testing
13.7.1.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
13.7.1.5.1.9. Pharmaceutical
Gas Testing
13.7.1.5.1.10. Viral
Safety Testing and Lot Release Testing
13.7.1.5.1.11. Others
(Water Analysis and etc.)
13.7.1.5.2. Pharmaceutical Chemistry
13.7.1.5.2.1. Elemental
Impurities
13.7.1.5.2.2. Volatile
Organic Impurities
13.7.1.5.2.3. Extractables
and Leachables
13.7.1.5.2.4. Semi-Volatile
Organic Impurites
13.7.1.5.2.5. Non-Volatile
Organic Impurites
13.7.1.5.2.6. Counterion
Analysis
13.7.1.5.2.7. Others
13.7.2. Canada
13.7.2.1. Canada Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Application
13.7.2.1.1. Allergens, antigens, vaccines and hormones
13.7.2.1.2. Cytokines, enzymes and human whole-blood
13.7.2.1.3. Plasma derivative
13.7.2.1.4. Immune sera, immunoglobulins and products of fermentation
13.7.2.1.5. Others (diagnostic agents for in vitro use, etc.)
13.7.2.2. Canada Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Offering
13.7.2.2.1. Consumables and Instruments
13.7.2.2.2. Solution
13.7.2.2.3. Services
13.7.2.3. Canada Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Techniques
13.7.2.3.1. GC/MS
13.7.2.3.2. LC/MS/ High Performance
Liquid (HPLC)
13.7.2.3.3. Ultra High Performance Liquid (UHPLC)
13.7.2.3.4. Supercritical Fluid (SFC) chromatography
13.7.2.3.5. Elemental analysis techniques
13.7.2.3.6. Others
13.7.2.4. Canada Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Testing Categories
13.7.2.4.1. Elemental Impurities
13.7.2.4.2. Volatile Organic Impurities
13.7.2.4.3. Extractables and Leachables
13.7.2.4.4. Semi-Volatile Organic Impurites
13.7.2.4.5. Non-Volatile Organic Impurites
13.7.2.4.6. Counterion Analysis
13.7.2.4.7. Others
13.7.2.5. Canada Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Analysis Type
13.7.2.5.1. Pharmaceutical Microbiology
13.7.2.5.1.1. Sterility
Testing
13.7.2.5.1.2. Antimicrobial
Efficacy Testing (AET)
13.7.2.5.1.3. Microbial
Limits Testing
13.7.2.5.1.4. Bioburden
Determination
13.7.2.5.1.5. Endotoxin
Testing
13.7.2.5.1.6. Environmental
Monitoring and Identification
13.7.2.5.1.7. Packaging
Testing
13.7.2.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
13.7.2.5.1.9. Pharmaceutical
Gas Testing
13.7.2.5.1.10. Viral
Safety Testing and Lot Release Testing
13.7.2.5.1.11. Others
(Water Analysis and etc.)
13.7.2.5.2. Pharmaceutical Chemistry
13.7.2.5.2.1. Elemental
Impurities
13.7.2.5.2.2. Volatile
Organic Impurities
13.7.2.5.2.3. Extractables
and Leachables
13.7.2.5.2.4. Semi-Volatile
Organic Impurites
13.7.2.5.2.5. Non-Volatile
Organic Impurites
13.7.2.5.2.6. Counterion
Analysis
13.7.2.5.2.7. Others
13.7.3. Mexico
13.7.3.1. Mexico Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Application
13.7.3.1.1. Allergens, antigens, vaccines and hormones
13.7.3.1.2. Cytokines, enzymes and human whole-blood
13.7.3.1.3. Plasma derivative
13.7.3.1.4. Immune sera, immunoglobulins and products of fermentation
13.7.3.1.5. Others (diagnostic agents for in vitro use, etc.)
13.7.3.2. Mexico Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Offering
13.7.3.2.1. Consumables and Instruments
13.7.3.2.2. Solution
13.7.3.2.3. Services
13.7.3.3. Mexico Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Techniques
13.7.3.3.1. GC/MS
13.7.3.3.2. LC/MS/ High Performance
Liquid (HPLC)
13.7.3.3.3. Ultra High Performance Liquid (UHPLC)
13.7.3.3.4. Supercritical Fluid (SFC) chromatography
13.7.3.3.5. Elemental analysis techniques
13.7.3.3.6. Others
13.7.3.4. Mexico Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Testing Categories
13.7.3.4.1. Elemental Impurities
13.7.3.4.2. Volatile Organic Impurities
13.7.3.4.3. Extractables and Leachables
13.7.3.4.4. Semi-Volatile Organic Impurites
13.7.3.4.5. Non-Volatile Organic Impurites
13.7.3.4.6. Counterion Analysis
13.7.3.4.7. Others
13.7.3.5. Mexico Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Analysis Type
13.7.3.5.1. Pharmaceutical Microbiology
13.7.3.5.1.1. Sterility
Testing
13.7.3.5.1.2. Antimicrobial
Efficacy Testing (AET)
13.7.3.5.1.3. Microbial
Limits Testing
13.7.3.5.1.4. Bioburden
Determination
13.7.3.5.1.5. Endotoxin
Testing
13.7.3.5.1.6. Environmental
Monitoring and Identification
13.7.3.5.1.7. Packaging
Testing
13.7.3.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
13.7.3.5.1.9. Pharmaceutical
Gas Testing
13.7.3.5.1.10. Viral
Safety Testing and Lot Release Testing
13.7.3.5.1.11. Others
(Water Analysis and etc.)
13.7.3.5.2. Pharmaceutical Chemistry
13.7.3.5.2.1. Elemental
Impurities
13.7.3.5.2.2. Volatile
Organic Impurities
13.7.3.5.2.3. Extractables
and Leachables
13.7.3.5.2.4. Semi-Volatile
Organic Impurites
13.7.3.5.2.5. Non-Volatile
Organic Impurites
13.7.3.5.2.6. Counterion
Analysis
13.7.3.5.2.7. Others
13.7.4. Rest of North America
13.7.4.1. Rest of North America Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By Application
13.7.4.1.1. Allergens, antigens, vaccines and hormones
13.7.4.1.2. Cytokines, enzymes and human whole-blood
13.7.4.1.3. Plasma derivative
13.7.4.1.4. Immune sera, immunoglobulins and products of fermentation
13.7.4.1.5. Others (diagnostic agents for in vitro use, etc.)
13.7.4.2. Rest of North America Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By Offering
13.7.4.2.1. Consumables and Instruments
13.7.4.2.2. Solution
13.7.4.2.3. Services
13.7.4.3. Rest of North America Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By
Techniques
13.7.4.3.1. GC/MS
13.7.4.3.2. LC/MS/ High Performance
Liquid (HPLC)
13.7.4.3.3. Ultra High Performance Liquid (UHPLC)
13.7.4.3.4. Supercritical Fluid (SFC) chromatography
13.7.4.3.5. Elemental analysis techniques
13.7.4.3.6. Others
13.7.4.4. Rest of North America Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By Testing Categories
13.7.4.4.1. Elemental Impurities
13.7.4.4.2. Volatile Organic Impurities
13.7.4.4.3. Extractables and Leachables
13.7.4.4.4. Semi-Volatile Organic Impurites
13.7.4.4.5. Non-Volatile Organic Impurites
13.7.4.4.6. Counterion Analysis
13.7.4.4.7. Others
13.7.4.5. Rest of North America Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By Analysis Type
13.7.4.5.1. Pharmaceutical Microbiology
13.7.4.5.1.1. Sterility
Testing
13.7.4.5.1.2. Antimicrobial
Efficacy Testing (AET)
13.7.4.5.1.3. Microbial
Limits Testing
13.7.4.5.1.4. Bioburden
Determination
13.7.4.5.1.5. Endotoxin
Testing
13.7.4.5.1.6. Environmental
Monitoring and Identification
13.7.4.5.1.7. Packaging
Testing
13.7.4.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
13.7.4.5.1.9. Pharmaceutical
Gas Testing
13.7.4.5.1.10. Viral
Safety Testing and Lot Release Testing
13.7.4.5.1.11. Others
(Water Analysis and etc.)
13.7.4.5.2. Pharmaceutical Chemistry
13.7.4.5.2.1. Elemental
Impurities
13.7.4.5.2.2. Volatile
Organic Impurities
13.7.4.5.2.3. Extractables
and Leachables
13.7.4.5.2.4. Semi-Volatile
Organic Impurites
13.7.4.5.2.5. Non-Volatile
Organic Impurites
13.7.4.5.2.6. Counterion
Analysis
13.7.4.5.2.7. Others
13.8. Key Segment for Channeling Investments
13.8.1. By Country
13.8.2. By Application
13.8.3. By Offering
13.8.4. By Techniques
13.8.5. By Testing Categories
13.8.6. By Analysis Type
14. Europe Pharmaceutical Quality Control Market Analysis and
Forecasts, 2021 - 2029
14.1. Overview
14.1.1. Europe Pharmaceutical Quality Control Market Revenue (US$ Mn)
14.2. Europe Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Application
14.2.1. Allergens, antigens, vaccines and hormones
14.2.2. Cytokines, enzymes and human whole-blood
14.2.3. Plasma derivative
14.2.4. Immune sera, immunoglobulins and products of fermentation
14.2.5. Others (diagnostic agents for in vitro use, etc.)
14.3. Europe Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Offering
14.3.1. Consumables and Instruments
14.3.2. Solution
14.3.3. Services
14.4. Europe Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Techniques
14.4.1. GC/MS
14.4.2. LC/MS/ High Performance
Liquid (HPLC)
14.4.3. Ultra High Performance Liquid (UHPLC)
14.4.4. Supercritical Fluid (SFC) chromatography
14.4.5. Elemental analysis techniques
14.4.6. Others
14.5. Europe Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Testing Categories
14.5.1. Elemental Impurities
14.5.2. Volatile Organic Impurities
14.5.3. Extractables and Leachables
14.5.4. Semi-Volatile Organic Impurites
14.5.5. Non-Volatile Organic Impurites
14.5.6. Counterion Analysis
14.5.7. Others
14.6. Europe Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Analysis Type
14.6.1. Pharmaceutical Microbiology
14.6.1.1. Sterility Testing
14.6.1.2. Antimicrobial Efficacy Testing (AET)
14.6.1.3. Microbial Limits Testing
14.6.1.4. Bioburden Determination
14.6.1.5. Endotoxin Testing
14.6.1.6. Environmental Monitoring and Identification
14.6.1.7. Packaging Testing
14.6.1.8. Pharmaceutical and Biopharmaceutical Release Testing
14.6.1.9. Pharmaceutical Gas Testing
14.6.1.10.
Viral Safety Testing and Lot
Release Testing
14.6.1.11.
Others (Water Analysis and etc.)
14.6.2. Pharmaceutical Chemistry
14.6.2.1. Elemental Impurities
14.6.2.2. Volatile Organic Impurities
14.6.2.3. Extractables and Leachables
14.6.2.4. Semi-Volatile Organic Impurites
14.6.2.5. Non-Volatile Organic Impurites
14.6.2.6. Counterion Analysis
14.6.2.7. Others
14.7. Europe Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Country
14.7.1. France
14.7.1.1. France Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Application
14.7.1.1.1. Allergens, antigens, vaccines and hormones
14.7.1.1.2. Cytokines, enzymes and human whole-blood
14.7.1.1.3. Plasma derivative
14.7.1.1.4. Immune sera, immunoglobulins and products of fermentation
14.7.1.1.5. Others (diagnostic agents for in vitro use, etc.)
14.7.1.2. France Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Offering
14.7.1.2.1. Consumables and Instruments
14.7.1.2.2. Solution
14.7.1.2.3. Services
14.7.1.3. France Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Techniques
14.7.1.3.1. GC/MS
14.7.1.3.2. LC/MS/ High Performance
Liquid (HPLC)
14.7.1.3.3. Ultra High Performance Liquid (UHPLC)
14.7.1.3.4. Supercritical Fluid (SFC) chromatography
14.7.1.3.5. Elemental analysis techniques
14.7.1.3.6. Others
14.7.1.4. France Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Testing Categories
14.7.1.4.1. Elemental Impurities
14.7.1.4.2. Volatile Organic Impurities
14.7.1.4.3. Extractables and Leachables
14.7.1.4.4. Semi-Volatile Organic Impurites
14.7.1.4.5. Non-Volatile Organic Impurites
14.7.1.4.6. Counterion Analysis
14.7.1.4.7. Others
14.7.1.5. France Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Analysis Type
14.7.1.5.1. Pharmaceutical Microbiology
14.7.1.5.1.1. Sterility
Testing
14.7.1.5.1.2. Antimicrobial
Efficacy Testing (AET)
14.7.1.5.1.3. Microbial
Limits Testing
14.7.1.5.1.4. Bioburden
Determination
14.7.1.5.1.5. Endotoxin
Testing
14.7.1.5.1.6. Environmental
Monitoring and Identification
14.7.1.5.1.7. Packaging
Testing
14.7.1.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
14.7.1.5.1.9. Pharmaceutical
Gas Testing
14.7.1.5.1.10. Viral
Safety Testing and Lot Release Testing
14.7.1.5.1.11. Others
(Water Analysis and etc.)
14.7.1.5.2. Pharmaceutical Chemistry
14.7.1.5.2.1. Elemental
Impurities
14.7.1.5.2.2. Volatile
Organic Impurities
14.7.1.5.2.3. Extractables
and Leachables
14.7.1.5.2.4. Semi-Volatile
Organic Impurites
14.7.1.5.2.5. Non-Volatile
Organic Impurites
14.7.1.5.2.6. Counterion
Analysis
14.7.1.5.2.7. Others
14.7.2. The UK
14.7.2.1. The UK Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Application
14.7.2.1.1. Allergens, antigens, vaccines and hormones
14.7.2.1.2. Cytokines, enzymes and human whole-blood
14.7.2.1.3. Plasma derivative
14.7.2.1.4. Immune sera, immunoglobulins and products of fermentation
14.7.2.1.5. Others (diagnostic agents for in vitro use, etc.)
14.7.2.2. The UK Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Offering
14.7.2.2.1. Consumables and Instruments
14.7.2.2.2. Solution
14.7.2.2.3. Services
14.7.2.3. The UK Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Techniques
14.7.2.3.1. GC/MS
14.7.2.3.2. LC/MS/ High Performance
Liquid (HPLC)
14.7.2.3.3. Ultra High Performance Liquid (UHPLC)
14.7.2.3.4. Supercritical Fluid (SFC) chromatography
14.7.2.3.5. Elemental analysis techniques
14.7.2.3.6. Others
14.7.2.4. The UK Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Testing Categories
14.7.2.4.1. Elemental Impurities
14.7.2.4.2. Volatile Organic Impurities
14.7.2.4.3. Extractables and Leachables
14.7.2.4.4. Semi-Volatile Organic Impurites
14.7.2.4.5. Non-Volatile Organic Impurites
14.7.2.4.6. Counterion Analysis
14.7.2.4.7. Others
14.7.2.5. The UK Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Analysis Type
14.7.2.5.1. Pharmaceutical Microbiology
14.7.2.5.1.1. Sterility
Testing
14.7.2.5.1.2. Antimicrobial
Efficacy Testing (AET)
14.7.2.5.1.3. Microbial
Limits Testing
14.7.2.5.1.4. Bioburden
Determination
14.7.2.5.1.5. Endotoxin
Testing
14.7.2.5.1.6. Environmental
Monitoring and Identification
14.7.2.5.1.7. Packaging
Testing
14.7.2.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
14.7.2.5.1.9. Pharmaceutical
Gas Testing
14.7.2.5.1.10. Viral
Safety Testing and Lot Release Testing
14.7.2.5.1.11. Others
(Water Analysis and etc.)
14.7.2.5.2. Pharmaceutical Chemistry
14.7.2.5.2.1. Elemental
Impurities
14.7.2.5.2.2. Volatile
Organic Impurities
14.7.2.5.2.3. Extractables
and Leachables
14.7.2.5.2.4. Semi-Volatile
Organic Impurites
14.7.2.5.2.5. Non-Volatile
Organic Impurites
14.7.2.5.2.6. Counterion
Analysis
14.7.2.5.2.7. Others
14.7.3. Spain
14.7.3.1. Spain Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Application
14.7.3.1.1. Allergens, antigens, vaccines and hormones
14.7.3.1.2. Cytokines, enzymes and human whole-blood
14.7.3.1.3. Plasma derivative
14.7.3.1.4. Immune sera, immunoglobulins and products of fermentation
14.7.3.1.5. Others (diagnostic agents for in vitro use, etc.)
14.7.3.2. Spain Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Offering
14.7.3.2.1. Consumables and Instruments
14.7.3.2.2. Solution
14.7.3.2.3. Services
14.7.3.3. Spain Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Techniques
14.7.3.3.1. GC/MS
14.7.3.3.2. LC/MS/ High Performance
Liquid (HPLC)
14.7.3.3.3. Ultra High Performance Liquid (UHPLC)
14.7.3.3.4. Supercritical Fluid (SFC) chromatography
14.7.3.3.5. Elemental analysis techniques
14.7.3.3.6. Others
14.7.3.4. Spain Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Testing Categories
14.7.3.4.1. Elemental Impurities
14.7.3.4.2. Volatile Organic Impurities
14.7.3.4.3. Extractables and Leachables
14.7.3.4.4. Semi-Volatile Organic Impurites
14.7.3.4.5. Non-Volatile Organic Impurites
14.7.3.4.6. Counterion Analysis
14.7.3.4.7. Others
14.7.3.5. Spain Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Analysis Type
14.7.3.5.1. Pharmaceutical Microbiology
14.7.3.5.1.1. Sterility
Testing
14.7.3.5.1.2. Antimicrobial
Efficacy Testing (AET)
14.7.3.5.1.3. Microbial
Limits Testing
14.7.3.5.1.4. Bioburden
Determination
14.7.3.5.1.5. Endotoxin
Testing
14.7.3.5.1.6. Environmental
Monitoring and Identification
14.7.3.5.1.7. Packaging
Testing
14.7.3.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
14.7.3.5.1.9. Pharmaceutical
Gas Testing
14.7.3.5.1.10. Viral
Safety Testing and Lot Release Testing
14.7.3.5.1.11. Others
(Water Analysis and etc.)
14.7.3.5.2. Pharmaceutical Chemistry
14.7.3.5.2.1. Elemental
Impurities
14.7.3.5.2.2. Volatile
Organic Impurities
14.7.3.5.2.3. Extractables
and Leachables
14.7.3.5.2.4. Semi-Volatile
Organic Impurites
14.7.3.5.2.5. Non-Volatile
Organic Impurites
14.7.3.5.2.6. Counterion
Analysis
14.7.3.5.2.7. Others
14.7.4. Germany
14.7.4.1. Germany Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Application
14.7.4.1.1. Allergens, antigens, vaccines and hormones
14.7.4.1.2. Cytokines, enzymes and human whole-blood
14.7.4.1.3. Plasma derivative
14.7.4.1.4. Immune sera, immunoglobulins and products of fermentation
14.7.4.1.5. Others (diagnostic agents for in vitro use, etc.)
14.7.4.2. Germany Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Offering
14.7.4.2.1. Consumables and Instruments
14.7.4.2.2. Solution
14.7.4.2.3. Services
14.7.4.3. Germany Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Techniques
14.7.4.3.1. GC/MS
14.7.4.3.2. LC/MS/ High Performance
Liquid (HPLC)
14.7.4.3.3. Ultra High Performance Liquid (UHPLC)
14.7.4.3.4. Supercritical Fluid (SFC) chromatography
14.7.4.3.5. Elemental analysis techniques
14.7.4.3.6. Others
14.7.4.4. Germany Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Testing Categories
14.7.4.4.1. Elemental Impurities
14.7.4.4.2. Volatile Organic Impurities
14.7.4.4.3. Extractables and Leachables
14.7.4.4.4. Semi-Volatile Organic Impurites
14.7.4.4.5. Non-Volatile Organic Impurites
14.7.4.4.6. Counterion Analysis
14.7.4.4.7. Others
14.7.4.5. Germany Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Analysis Type
14.7.4.5.1. Pharmaceutical Microbiology
14.7.4.5.1.1. Sterility
Testing
14.7.4.5.1.2. Antimicrobial
Efficacy Testing (AET)
14.7.4.5.1.3. Microbial
Limits Testing
14.7.4.5.1.4. Bioburden
Determination
14.7.4.5.1.5. Endotoxin
Testing
14.7.4.5.1.6. Environmental
Monitoring and Identification
14.7.4.5.1.7. Packaging
Testing
14.7.4.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
14.7.4.5.1.9. Pharmaceutical
Gas Testing
14.7.4.5.1.10. Viral
Safety Testing and Lot Release Testing
14.7.4.5.1.11. Others
(Water Analysis and etc.)
14.7.4.5.2. Pharmaceutical Chemistry
14.7.4.5.2.1. Elemental
Impurities
14.7.4.5.2.2. Volatile
Organic Impurities
14.7.4.5.2.3. Extractables
and Leachables
14.7.4.5.2.4. Semi-Volatile
Organic Impurites
14.7.4.5.2.5. Non-Volatile
Organic Impurites
14.7.4.5.2.6. Counterion
Analysis
14.7.4.5.2.7. Others
14.7.5. Italy
14.7.5.1. Italy Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Application
14.7.5.1.1. Allergens, antigens, vaccines and hormones
14.7.5.1.2. Cytokines, enzymes and human whole-blood
14.7.5.1.3. Plasma derivative
14.7.5.1.4. Immune sera, immunoglobulins and products of fermentation
14.7.5.1.5. Others (diagnostic agents for in vitro use, etc.)
14.7.5.2. Italy Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Offering
14.7.5.2.1. Consumables and Instruments
14.7.5.2.2. Solution
14.7.5.2.3. Services
14.7.5.3. Italy Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Techniques
14.7.5.3.1. GC/MS
14.7.5.3.2. LC/MS/ High Performance
Liquid (HPLC)
14.7.5.3.3. Ultra High Performance Liquid (UHPLC)
14.7.5.3.4. Supercritical Fluid (SFC) chromatography
14.7.5.3.5. Elemental analysis techniques
14.7.5.3.6. Others
14.7.5.4. Italy Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Testing Categories
14.7.5.4.1. Elemental Impurities
14.7.5.4.2. Volatile Organic Impurities
14.7.5.4.3. Extractables and Leachables
14.7.5.4.4. Semi-Volatile Organic Impurites
14.7.5.4.5. Non-Volatile Organic Impurites
14.7.5.4.6. Counterion Analysis
14.7.5.4.7. Others
14.7.5.5. Italy Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Analysis Type
14.7.5.5.1. Pharmaceutical Microbiology
14.7.5.5.1.1. Sterility
Testing
14.7.5.5.1.2. Antimicrobial
Efficacy Testing (AET)
14.7.5.5.1.3. Microbial
Limits Testing
14.7.5.5.1.4. Bioburden
Determination
14.7.5.5.1.5. Endotoxin
Testing
14.7.5.5.1.6. Environmental
Monitoring and Identification
14.7.5.5.1.7. Packaging
Testing
14.7.5.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
14.7.5.5.1.9. Pharmaceutical
Gas Testing
14.7.5.5.1.10. Viral
Safety Testing and Lot Release Testing
14.7.5.5.1.11. Others
(Water Analysis and etc.)
14.7.5.5.2. Pharmaceutical Chemistry
14.7.5.5.2.1. Elemental
Impurities
14.7.5.5.2.2. Volatile
Organic Impurities
14.7.5.5.2.3. Extractables
and Leachables
14.7.5.5.2.4. Semi-Volatile
Organic Impurites
14.7.5.5.2.5. Non-Volatile
Organic Impurites
14.7.5.5.2.6. Counterion
Analysis
14.7.5.5.2.7. Others
14.7.6. Nordic Countries
14.7.6.1. Nordic Countries Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Application
14.7.6.1.1. Allergens, antigens, vaccines and hormones
14.7.6.1.2. Cytokines, enzymes and human whole-blood
14.7.6.1.3. Plasma derivative
14.7.6.1.4. Immune sera, immunoglobulins and products of fermentation
14.7.6.1.5. Others (diagnostic agents for in vitro use, etc.)
14.7.6.2. Nordic Countries Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Offering
14.7.6.2.1. Consumables and Instruments
14.7.6.2.2. Solution
14.7.6.2.3. Services
14.7.6.3. Nordic Countries Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Techniques
14.7.6.3.1. GC/MS
14.7.6.3.2. LC/MS/ High Performance
Liquid (HPLC)
14.7.6.3.3. Ultra High Performance Liquid (UHPLC)
14.7.6.3.4. Supercritical Fluid (SFC) chromatography
14.7.6.3.5. Elemental analysis techniques
14.7.6.3.6. Others
14.7.6.4. Nordic Countries Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Testing Categories
14.7.6.4.1. Elemental Impurities
14.7.6.4.2. Volatile Organic Impurities
14.7.6.4.3. Extractables and Leachables
14.7.6.4.4. Semi-Volatile Organic Impurites
14.7.6.4.5. Non-Volatile Organic Impurites
14.7.6.4.6. Counterion Analysis
14.7.6.4.7. Others
14.7.6.5. Nordic Countries Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Analysis Type
14.7.6.5.1. Pharmaceutical Microbiology
14.7.6.5.1.1. Sterility
Testing
14.7.6.5.1.2. Antimicrobial
Efficacy Testing (AET)
14.7.6.5.1.3. Microbial
Limits Testing
14.7.6.5.1.4. Bioburden
Determination
14.7.6.5.1.5. Endotoxin
Testing
14.7.6.5.1.6. Environmental
Monitoring and Identification
14.7.6.5.1.7. Packaging
Testing
14.7.6.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
14.7.6.5.1.9. Pharmaceutical
Gas Testing
14.7.6.5.1.10. Viral
Safety Testing and Lot Release Testing
14.7.6.5.1.11. Others
(Water Analysis and etc.)
14.7.6.5.2. Pharmaceutical Chemistry
14.7.6.5.2.1. Elemental
Impurities
14.7.6.5.2.2. Volatile
Organic Impurities
14.7.6.5.2.3. Extractables
and Leachables
14.7.6.5.2.4. Semi-Volatile
Organic Impurites
14.7.6.5.2.5. Non-Volatile
Organic Impurites
14.7.6.5.2.6. Counterion
Analysis
14.7.6.5.2.7. Others
14.7.6.6. Nordic Countries Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Country
14.7.6.6.1. Denmark
14.7.6.6.2. Finland
14.7.6.6.3. Iceland
14.7.6.6.4. Sweden
14.7.6.6.5. Norway
14.7.7. Benelux Union
14.7.7.1. Benelux Union Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Application
14.7.7.1.1. Allergens, antigens, vaccines and hormones
14.7.7.1.2. Cytokines, enzymes and human whole-blood
14.7.7.1.3. Plasma derivative
14.7.7.1.4. Immune sera, immunoglobulins and products of fermentation
14.7.7.1.5. Others (diagnostic agents for in vitro use, etc.)
14.7.7.2. Benelux Union Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Offering
14.7.7.2.1. Consumables and Instruments
14.7.7.2.2. Solution
14.7.7.2.3. Services
14.7.7.3. Benelux Union Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Techniques
14.7.7.3.1. GC/MS
14.7.7.3.2. LC/MS/ High Performance
Liquid (HPLC)
14.7.7.3.3. Ultra High Performance Liquid (UHPLC)
14.7.7.3.4. Supercritical Fluid (SFC) chromatography
14.7.7.3.5. Elemental analysis techniques
14.7.7.3.6. Others
14.7.7.4. Benelux Union Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Testing Categories
14.7.7.4.1. Elemental Impurities
14.7.7.4.2. Volatile Organic Impurities
14.7.7.4.3. Extractables and Leachables
14.7.7.4.4. Semi-Volatile Organic Impurites
14.7.7.4.5. Non-Volatile Organic Impurites
14.7.7.4.6. Counterion Analysis
14.7.7.4.7. Others
14.7.7.5. Benelux Union Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Analysis Type
14.7.7.5.1. Pharmaceutical Microbiology
14.7.7.5.1.1. Sterility
Testing
14.7.7.5.1.2. Antimicrobial
Efficacy Testing (AET)
14.7.7.5.1.3. Microbial
Limits Testing
14.7.7.5.1.4. Bioburden
Determination
14.7.7.5.1.5. Endotoxin
Testing
14.7.7.5.1.6. Environmental
Monitoring and Identification
14.7.7.5.1.7. Packaging
Testing
14.7.7.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
14.7.7.5.1.9. Pharmaceutical
Gas Testing
14.7.7.5.1.10. Viral
Safety Testing and Lot Release Testing
14.7.7.5.1.11. Others
(Water Analysis and etc.)
14.7.7.5.2. Pharmaceutical Chemistry
14.7.7.5.2.1. Elemental
Impurities
14.7.7.5.2.2. Volatile
Organic Impurities
14.7.7.5.2.3. Extractables
and Leachables
14.7.7.5.2.4. Semi-Volatile
Organic Impurites
14.7.7.5.2.5. Non-Volatile
Organic Impurites
14.7.7.5.2.6. Counterion
Analysis
14.7.7.5.2.7. Others
14.7.7.6. Benelux Union Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Country
14.7.7.6.1. Belgium
14.7.7.6.2. The Netherlands
14.7.7.6.3. Luxembourg
14.7.8. Rest of Europe
14.7.8.1. Rest of Europe Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Application
14.7.8.1.1. Allergens, antigens, vaccines and hormones
14.7.8.1.2. Cytokines, enzymes and human whole-blood
14.7.8.1.3. Plasma derivative
14.7.8.1.4. Immune sera, immunoglobulins and products of fermentation
14.7.8.1.5. Others (diagnostic agents for in vitro use, etc.)
14.7.8.2. Rest of Europe Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Offering
14.7.8.2.1. Consumables and Instruments
14.7.8.2.2. Solution
14.7.8.2.3. Services
14.7.8.3. Rest of Europe Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Techniques
14.7.8.3.1. GC/MS
14.7.8.3.2. LC/MS/ High Performance
Liquid (HPLC)
14.7.8.3.3. Ultra High Performance Liquid (UHPLC)
14.7.8.3.4. Supercritical Fluid (SFC) chromatography
14.7.8.3.5. Elemental analysis techniques
14.7.8.3.6. Others
14.7.8.4. Rest of Europe Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Testing Categories
14.7.8.4.1. Elemental Impurities
14.7.8.4.2. Volatile Organic Impurities
14.7.8.4.3. Extractables and Leachables
14.7.8.4.4. Semi-Volatile Organic Impurites
14.7.8.4.5. Non-Volatile Organic Impurites
14.7.8.4.6. Counterion Analysis
14.7.8.4.7. Others
14.7.8.5. Rest of Europe Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Analysis Type
14.7.8.5.1. Pharmaceutical Microbiology
14.7.8.5.1.1. Sterility
Testing
14.7.8.5.1.2. Antimicrobial
Efficacy Testing (AET)
14.7.8.5.1.3. Microbial
Limits Testing
14.7.8.5.1.4. Bioburden
Determination
14.7.8.5.1.5. Endotoxin
Testing
14.7.8.5.1.6. Environmental
Monitoring and Identification
14.7.8.5.1.7. Packaging
Testing
14.7.8.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
14.7.8.5.1.9. Pharmaceutical
Gas Testing
14.7.8.5.1.10. Viral
Safety Testing and Lot Release Testing
14.7.8.5.1.11. Others
(Water Analysis and etc.)
14.7.8.5.2. Pharmaceutical Chemistry
14.7.8.5.2.1. Elemental
Impurities
14.7.8.5.2.2. Volatile
Organic Impurities
14.7.8.5.2.3. Extractables
and Leachables
14.7.8.5.2.4. Semi-Volatile
Organic Impurites
14.7.8.5.2.5. Non-Volatile
Organic Impurites
14.7.8.5.2.6. Counterion
Analysis
14.7.8.5.2.7. Others
14.8. Key Segment for Channeling Investments
14.8.1. By Country
14.8.2. By Application
14.8.3. By Offering
14.8.4. By Techniques
14.8.5. By Testing Categories
14.8.6. By Analysis Type
15. Asia Pacific Pharmaceutical Quality Control Market Analysis
and Forecasts, 2021 - 2029
15.1. Overview
15.1.1. Asia Pacific Pharmaceutical Quality Control Market Revenue
(US$ Mn)
15.2. Asia Pacific Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Application
15.2.1. Allergens, antigens, vaccines and hormones
15.2.2. Cytokines, enzymes and human whole-blood
15.2.3. Plasma derivative
15.2.4. Immune sera, immunoglobulins and products of fermentation
15.2.5. Others (diagnostic agents for in vitro use, etc.)
15.3. Asia Pacific Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Offering
15.3.1. Consumables and Instruments
15.3.2. Solution
15.3.3. Services
15.4. Asia Pacific Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Techniques
15.4.1. GC/MS
15.4.2. LC/MS/ High Performance
Liquid (HPLC)
15.4.3. Ultra High Performance Liquid (UHPLC)
15.4.4. Supercritical Fluid (SFC) chromatography
15.4.5. Elemental analysis techniques
15.4.6. Others
15.5. Asia Pacific Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Testing Categories
15.5.1. Elemental Impurities
15.5.2. Volatile Organic Impurities
15.5.3. Extractables and Leachables
15.5.4. Semi-Volatile Organic Impurites
15.5.5. Non-Volatile Organic Impurites
15.5.6. Counterion Analysis
15.5.7. Others
15.6. Asia Pacific Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Analysis Type
15.6.1. Pharmaceutical Microbiology
15.6.1.1. Sterility Testing
15.6.1.2. Antimicrobial Efficacy Testing (AET)
15.6.1.3. Microbial Limits Testing
15.6.1.4. Bioburden Determination
15.6.1.5. Endotoxin Testing
15.6.1.6. Environmental Monitoring and Identification
15.6.1.7. Packaging Testing
15.6.1.8. Pharmaceutical and Biopharmaceutical Release Testing
15.6.1.9. Pharmaceutical Gas Testing
15.6.1.10.
Viral Safety Testing and Lot
Release Testing
15.6.1.11.
Others (Water Analysis and etc.)
15.6.2. Pharmaceutical Chemistry
15.6.2.1. Elemental Impurities
15.6.2.2. Volatile Organic Impurities
15.6.2.3. Extractables and Leachables
15.6.2.4. Semi-Volatile Organic Impurites
15.6.2.5. Non-Volatile Organic Impurites
15.6.2.6. Counterion Analysis
15.6.2.7. Others
15.7. Asia Pacific Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Country
15.7.1. China
15.7.1.1. China Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Application
15.7.1.1.1. Allergens, antigens, vaccines and hormones
15.7.1.1.2. Cytokines, enzymes and human whole-blood
15.7.1.1.3. Plasma derivative
15.7.1.1.4. Immune sera, immunoglobulins and products of fermentation
15.7.1.1.5. Others (diagnostic agents for in vitro use, etc.)
15.7.1.2. China Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Offering
15.7.1.2.1. Consumables and Instruments
15.7.1.2.2. Solution
15.7.1.2.3. Services
15.7.1.3. China Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Techniques
15.7.1.3.1. GC/MS
15.7.1.3.2. LC/MS/ High Performance
Liquid (HPLC)
15.7.1.3.3. Ultra High Performance Liquid (UHPLC)
15.7.1.3.4. Supercritical Fluid (SFC) chromatography
15.7.1.3.5. Elemental analysis techniques
15.7.1.3.6. Others
15.7.1.4. China Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Testing Categories
15.7.1.4.1. Elemental Impurities
15.7.1.4.2. Volatile Organic Impurities
15.7.1.4.3. Extractables and Leachables
15.7.1.4.4. Semi-Volatile Organic Impurites
15.7.1.4.5. Non-Volatile Organic Impurites
15.7.1.4.6. Counterion Analysis
15.7.1.4.7. Others
15.7.1.5. China Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Analysis Type
15.7.1.5.1. Pharmaceutical Microbiology
15.7.1.5.1.1. Sterility
Testing
15.7.1.5.1.2. Antimicrobial
Efficacy Testing (AET)
15.7.1.5.1.3. Microbial
Limits Testing
15.7.1.5.1.4. Bioburden
Determination
15.7.1.5.1.5. Endotoxin
Testing
15.7.1.5.1.6. Environmental
Monitoring and Identification
15.7.1.5.1.7. Packaging
Testing
15.7.1.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
15.7.1.5.1.9. Pharmaceutical
Gas Testing
15.7.1.5.1.10. Viral
Safety Testing and Lot Release Testing
15.7.1.5.1.11. Others
(Water Analysis and etc.)
15.7.1.5.2. Pharmaceutical Chemistry
15.7.1.5.2.1. Elemental
Impurities
15.7.1.5.2.2. Volatile
Organic Impurities
15.7.1.5.2.3. Extractables
and Leachables
15.7.1.5.2.4. Semi-Volatile
Organic Impurites
15.7.1.5.2.5. Non-Volatile
Organic Impurites
15.7.1.5.2.6. Counterion
Analysis
15.7.1.5.2.7. Others
15.7.2. Japan
15.7.2.1. Japan Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Application
15.7.2.1.1. Allergens, antigens, vaccines and hormones
15.7.2.1.2. Cytokines, enzymes and human whole-blood
15.7.2.1.3. Plasma derivative
15.7.2.1.4. Immune sera, immunoglobulins and products of fermentation
15.7.2.1.5. Others (diagnostic agents for in vitro use, etc.)
15.7.2.2. Japan Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Offering
15.7.2.2.1. Consumables and Instruments
15.7.2.2.2. Solution
15.7.2.2.3. Services
15.7.2.3. Japan Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Techniques
15.7.2.3.1. GC/MS
15.7.2.3.2. LC/MS/ High Performance
Liquid (HPLC)
15.7.2.3.3. Ultra High Performance Liquid (UHPLC)
15.7.2.3.4. Supercritical Fluid (SFC) chromatography
15.7.2.3.5. Elemental analysis techniques
15.7.2.3.6. Others
15.7.2.4. Japan Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Testing Categories
15.7.2.4.1. Elemental Impurities
15.7.2.4.2. Volatile Organic Impurities
15.7.2.4.3. Extractables and Leachables
15.7.2.4.4. Semi-Volatile Organic Impurites
15.7.2.4.5. Non-Volatile Organic Impurites
15.7.2.4.6. Counterion Analysis
15.7.2.4.7. Others
15.7.2.5. Japan Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Analysis Type
15.7.2.5.1. Pharmaceutical Microbiology
15.7.2.5.1.1. Sterility
Testing
15.7.2.5.1.2. Antimicrobial
Efficacy Testing (AET)
15.7.2.5.1.3. Microbial
Limits Testing
15.7.2.5.1.4. Bioburden
Determination
15.7.2.5.1.5. Endotoxin
Testing
15.7.2.5.1.6. Environmental
Monitoring and Identification
15.7.2.5.1.7. Packaging
Testing
15.7.2.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
15.7.2.5.1.9. Pharmaceutical
Gas Testing
15.7.2.5.1.10. Viral
Safety Testing and Lot Release Testing
15.7.2.5.1.11. Others
(Water Analysis and etc.)
15.7.2.5.2. Pharmaceutical Chemistry
15.7.2.5.2.1. Elemental
Impurities
15.7.2.5.2.2. Volatile
Organic Impurities
15.7.2.5.2.3. Extractables
and Leachables
15.7.2.5.2.4. Semi-Volatile
Organic Impurites
15.7.2.5.2.5. Non-Volatile
Organic Impurites
15.7.2.5.2.6. Counterion
Analysis
15.7.2.5.2.7. Others
15.7.3. India
15.7.3.1. India Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Application
15.7.3.1.1. Allergens, antigens, vaccines and hormones
15.7.3.1.2. Cytokines, enzymes and human whole-blood
15.7.3.1.3. Plasma derivative
15.7.3.1.4. Immune sera, immunoglobulins and products of fermentation
15.7.3.1.5. Others (diagnostic agents for in vitro use, etc.)
15.7.3.2. India Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Offering
15.7.3.2.1. Consumables and Instruments
15.7.3.2.2. Solution
15.7.3.2.3. Services
15.7.3.3. India Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Techniques
15.7.3.3.1. GC/MS
15.7.3.3.2. LC/MS/ High Performance
Liquid (HPLC)
15.7.3.3.3. Ultra High Performance Liquid (UHPLC)
15.7.3.3.4. Supercritical Fluid (SFC) chromatography
15.7.3.3.5. Elemental analysis techniques
15.7.3.3.6. Others
15.7.3.4. India Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Testing Categories
15.7.3.4.1. Elemental Impurities
15.7.3.4.2. Volatile Organic Impurities
15.7.3.4.3. Extractables and Leachables
15.7.3.4.4. Semi-Volatile Organic Impurites
15.7.3.4.5. Non-Volatile Organic Impurites
15.7.3.4.6. Counterion Analysis
15.7.3.4.7. Others
15.7.3.5. India Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Analysis Type
15.7.3.5.1. Pharmaceutical Microbiology
15.7.3.5.1.1. Sterility
Testing
15.7.3.5.1.2. Antimicrobial
Efficacy Testing (AET)
15.7.3.5.1.3. Microbial
Limits Testing
15.7.3.5.1.4. Bioburden
Determination
15.7.3.5.1.5. Endotoxin
Testing
15.7.3.5.1.6. Environmental
Monitoring and Identification
15.7.3.5.1.7. Packaging
Testing
15.7.3.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
15.7.3.5.1.9. Pharmaceutical
Gas Testing
15.7.3.5.1.10. Viral
Safety Testing and Lot Release Testing
15.7.3.5.1.11. Others
(Water Analysis and etc.)
15.7.3.5.2. Pharmaceutical Chemistry
15.7.3.5.2.1. Elemental
Impurities
15.7.3.5.2.2. Volatile
Organic Impurities
15.7.3.5.2.3. Extractables
and Leachables
15.7.3.5.2.4. Semi-Volatile
Organic Impurites
15.7.3.5.2.5. Non-Volatile
Organic Impurites
15.7.3.5.2.6. Counterion
Analysis
15.7.3.5.2.7. Others
15.7.4. New Zealand
15.7.4.1. New Zealand Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Application
15.7.4.1.1. Allergens, antigens, vaccines and hormones
15.7.4.1.2. Cytokines, enzymes and human whole-blood
15.7.4.1.3. Plasma derivative
15.7.4.1.4. Immune sera, immunoglobulins and products of fermentation
15.7.4.1.5. Others (diagnostic agents for in vitro use, etc.)
15.7.4.2. New Zealand Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Offering
15.7.4.2.1. Consumables and Instruments
15.7.4.2.2. Solution
15.7.4.2.3. Services
15.7.4.3. New Zealand Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Techniques
15.7.4.3.1. GC/MS
15.7.4.3.2. LC/MS/ High Performance
Liquid (HPLC)
15.7.4.3.3. Ultra High Performance Liquid (UHPLC)
15.7.4.3.4. Supercritical Fluid (SFC) chromatography
15.7.4.3.5. Elemental analysis techniques
15.7.4.3.6. Others
15.7.4.4. New Zealand Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Testing Categories
15.7.4.4.1. Elemental Impurities
15.7.4.4.2. Volatile Organic Impurities
15.7.4.4.3. Extractables and Leachables
15.7.4.4.4. Semi-Volatile Organic Impurites
15.7.4.4.5. Non-Volatile Organic Impurites
15.7.4.4.6. Counterion Analysis
15.7.4.4.7. Others
15.7.4.5. New Zealand Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Analysis Type
15.7.4.5.1. Pharmaceutical Microbiology
15.7.4.5.1.1. Sterility
Testing
15.7.4.5.1.2. Antimicrobial
Efficacy Testing (AET)
15.7.4.5.1.3. Microbial
Limits Testing
15.7.4.5.1.4. Bioburden
Determination
15.7.4.5.1.5. Endotoxin
Testing
15.7.4.5.1.6. Environmental
Monitoring and Identification
15.7.4.5.1.7. Packaging
Testing
15.7.4.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
15.7.4.5.1.9. Pharmaceutical
Gas Testing
15.7.4.5.1.10. Viral
Safety Testing and Lot Release Testing
15.7.4.5.1.11. Others
(Water Analysis and etc.)
15.7.4.5.2. Pharmaceutical Chemistry
15.7.4.5.2.1. Elemental
Impurities
15.7.4.5.2.2. Volatile
Organic Impurities
15.7.4.5.2.3. Extractables
and Leachables
15.7.4.5.2.4. Semi-Volatile
Organic Impurites
15.7.4.5.2.5. Non-Volatile
Organic Impurites
15.7.4.5.2.6. Counterion
Analysis
15.7.4.5.2.7. Others
15.7.5. Australia
15.7.5.1. Australia Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Application
15.7.5.1.1. Allergens, antigens, vaccines and hormones
15.7.5.1.2. Cytokines, enzymes and human whole-blood
15.7.5.1.3. Plasma derivative
15.7.5.1.4. Immune sera, immunoglobulins and products of fermentation
15.7.5.1.5. Others (diagnostic agents for in vitro use, etc.)
15.7.5.2. Australia Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Offering
15.7.5.2.1. Consumables and Instruments
15.7.5.2.2. Solution
15.7.5.2.3. Services
15.7.5.3. Australia Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Techniques
15.7.5.3.1. GC/MS
15.7.5.3.2. LC/MS/ High Performance
Liquid (HPLC)
15.7.5.3.3. Ultra High Performance Liquid (UHPLC)
15.7.5.3.4. Supercritical Fluid (SFC) chromatography
15.7.5.3.5. Elemental analysis techniques
15.7.5.3.6. Others
15.7.5.4. Australia Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Testing Categories
15.7.5.4.1. Elemental Impurities
15.7.5.4.2. Volatile Organic Impurities
15.7.5.4.3. Extractables and Leachables
15.7.5.4.4. Semi-Volatile Organic Impurites
15.7.5.4.5. Non-Volatile Organic Impurites
15.7.5.4.6. Counterion Analysis
15.7.5.4.7. Others
15.7.5.5. Australia Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Analysis Type
15.7.5.5.1. Pharmaceutical Microbiology
15.7.5.5.1.1. Sterility
Testing
15.7.5.5.1.2. Antimicrobial
Efficacy Testing (AET)
15.7.5.5.1.3. Microbial
Limits Testing
15.7.5.5.1.4. Bioburden
Determination
15.7.5.5.1.5. Endotoxin
Testing
15.7.5.5.1.6. Environmental
Monitoring and Identification
15.7.5.5.1.7. Packaging
Testing
15.7.5.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
15.7.5.5.1.9. Pharmaceutical
Gas Testing
15.7.5.5.1.10. Viral
Safety Testing and Lot Release Testing
15.7.5.5.1.11. Others
(Water Analysis and etc.)
15.7.5.5.2. Pharmaceutical Chemistry
15.7.5.5.2.1. Elemental
Impurities
15.7.5.5.2.2. Volatile
Organic Impurities
15.7.5.5.2.3. Extractables
and Leachables
15.7.5.5.2.4. Semi-Volatile
Organic Impurites
15.7.5.5.2.5. Non-Volatile
Organic Impurites
15.7.5.5.2.6. Counterion
Analysis
15.7.5.5.2.7. Others
15.7.6. South Korea
15.7.6.1. South Korea Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Application
15.7.6.1.1. Allergens, antigens, vaccines and hormones
15.7.6.1.2. Cytokines, enzymes and human whole-blood
15.7.6.1.3. Plasma derivative
15.7.6.1.4. Immune sera, immunoglobulins and products of fermentation
15.7.6.1.5. Others (diagnostic agents for in vitro use, etc.)
15.7.6.2. South Korea Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Offering
15.7.6.2.1. Consumables and Instruments
15.7.6.2.2. Solution
15.7.6.2.3. Services
15.7.6.3. South Korea Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Techniques
15.7.6.3.1. GC/MS
15.7.6.3.2. LC/MS/ High Performance
Liquid (HPLC)
15.7.6.3.3. Ultra High Performance Liquid (UHPLC)
15.7.6.3.4. Supercritical Fluid (SFC) chromatography
15.7.6.3.5. Elemental analysis techniques
15.7.6.3.6. Others
15.7.6.4. South Korea Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Testing Categories
15.7.6.4.1. Elemental Impurities
15.7.6.4.2. Volatile Organic Impurities
15.7.6.4.3. Extractables and Leachables
15.7.6.4.4. Semi-Volatile Organic Impurites
15.7.6.4.5. Non-Volatile Organic Impurites
15.7.6.4.6. Counterion Analysis
15.7.6.4.7. Others
15.7.6.5. South Korea Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Analysis Type
15.7.6.5.1. Pharmaceutical Microbiology
15.7.6.5.1.1. Sterility
Testing
15.7.6.5.1.2. Antimicrobial
Efficacy Testing (AET)
15.7.6.5.1.3. Microbial
Limits Testing
15.7.6.5.1.4. Bioburden
Determination
15.7.6.5.1.5. Endotoxin
Testing
15.7.6.5.1.6. Environmental
Monitoring and Identification
15.7.6.5.1.7. Packaging
Testing
15.7.6.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
15.7.6.5.1.9. Pharmaceutical
Gas Testing
15.7.6.5.1.10. Viral
Safety Testing and Lot Release Testing
15.7.6.5.1.11. Others
(Water Analysis and etc.)
15.7.6.5.2. Pharmaceutical Chemistry
15.7.6.5.2.1. Elemental
Impurities
15.7.6.5.2.2. Volatile
Organic Impurities
15.7.6.5.2.3. Extractables
and Leachables
15.7.6.5.2.4. Semi-Volatile
Organic Impurites
15.7.6.5.2.5. Non-Volatile
Organic Impurites
15.7.6.5.2.6. Counterion
Analysis
15.7.6.5.2.7. Others
15.7.7. Southeast Asia
15.7.7.1. Southeast Asia Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Application
15.7.7.1.1. Allergens, antigens, vaccines and hormones
15.7.7.1.2. Cytokines, enzymes and human whole-blood
15.7.7.1.3. Plasma derivative
15.7.7.1.4. Immune sera, immunoglobulins and products of fermentation
15.7.7.1.5. Others (diagnostic agents for in vitro use, etc.)
15.7.7.2. Southeast Asia Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Offering
15.7.7.2.1. Consumables and Instruments
15.7.7.2.2. Solution
15.7.7.2.3. Services
15.7.7.3. Southeast Asia Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Techniques
15.7.7.3.1. GC/MS
15.7.7.3.2. LC/MS/ High Performance
Liquid (HPLC)
15.7.7.3.3. Ultra High Performance Liquid (UHPLC)
15.7.7.3.4. Supercritical Fluid (SFC) chromatography
15.7.7.3.5. Elemental analysis techniques
15.7.7.3.6. Others
15.7.7.4. Southeast Asia Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Testing Categories
15.7.7.4.1. Elemental Impurities
15.7.7.4.2. Volatile Organic Impurities
15.7.7.4.3. Extractables and Leachables
15.7.7.4.4. Semi-Volatile Organic Impurites
15.7.7.4.5. Non-Volatile Organic Impurites
15.7.7.4.6. Counterion Analysis
15.7.7.4.7. Others
15.7.7.5. Southeast Asia Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Analysis Type
15.7.7.5.1. Pharmaceutical Microbiology
15.7.7.5.1.1. Sterility
Testing
15.7.7.5.1.2. Antimicrobial
Efficacy Testing (AET)
15.7.7.5.1.3. Microbial
Limits Testing
15.7.7.5.1.4. Bioburden
Determination
15.7.7.5.1.5. Endotoxin
Testing
15.7.7.5.1.6. Environmental
Monitoring and Identification
15.7.7.5.1.7. Packaging
Testing
15.7.7.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
15.7.7.5.1.9. Pharmaceutical
Gas Testing
15.7.7.5.1.10. Viral
Safety Testing and Lot Release Testing
15.7.7.5.1.11. Others
(Water Analysis and etc.)
15.7.7.5.2. Pharmaceutical Chemistry
15.7.7.5.2.1. Elemental
Impurities
15.7.7.5.2.2. Volatile
Organic Impurities
15.7.7.5.2.3. Extractables
and Leachables
15.7.7.5.2.4. Semi-Volatile
Organic Impurites
15.7.7.5.2.5. Non-Volatile
Organic Impurites
15.7.7.5.2.6. Counterion
Analysis
15.7.7.5.2.7. Others
15.7.7.6. Southeast Asia Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Country
15.7.7.6.1. Indonesia
15.7.7.6.2. Thailand
15.7.7.6.3. Malaysia
15.7.7.6.4. Singapore
15.7.7.6.5. Rest of Southeast Asia
15.7.8. Rest of Asia Pacific
15.7.8.1. Rest of Asia Pacific Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By Application
15.7.8.1.1. Allergens, antigens, vaccines and hormones
15.7.8.1.2. Cytokines, enzymes and human whole-blood
15.7.8.1.3. Plasma derivative
15.7.8.1.4. Immune sera, immunoglobulins and products of fermentation
15.7.8.1.5. Others (diagnostic agents for in vitro use, etc.)
15.7.8.2. Rest of Asia Pacific Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By Offering
15.7.8.2.1. Consumables and Instruments
15.7.8.2.2. Solution
15.7.8.2.3. Services
15.7.8.3. Rest of Asia Pacific Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By
Techniques
15.7.8.3.1. GC/MS
15.7.8.3.2. LC/MS/ High Performance
Liquid (HPLC)
15.7.8.3.3. Ultra High Performance Liquid (UHPLC)
15.7.8.3.4. Supercritical Fluid (SFC) chromatography
15.7.8.3.5. Elemental analysis techniques
15.7.8.3.6. Others
15.7.8.4. Rest of Asia Pacific Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By Testing Categories
15.7.8.4.1. Elemental Impurities
15.7.8.4.2. Volatile Organic Impurities
15.7.8.4.3. Extractables and Leachables
15.7.8.4.4. Semi-Volatile Organic Impurites
15.7.8.4.5. Non-Volatile Organic Impurites
15.7.8.4.6. Counterion Analysis
15.7.8.4.7. Others
15.7.8.5. Rest of Asia Pacific Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By Analysis Type
15.7.8.5.1. Pharmaceutical Microbiology
15.7.8.5.1.1. Sterility
Testing
15.7.8.5.1.2. Antimicrobial
Efficacy Testing (AET)
15.7.8.5.1.3. Microbial
Limits Testing
15.7.8.5.1.4. Bioburden
Determination
15.7.8.5.1.5. Endotoxin
Testing
15.7.8.5.1.6. Environmental
Monitoring and Identification
15.7.8.5.1.7. Packaging
Testing
15.7.8.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
15.7.8.5.1.9. Pharmaceutical
Gas Testing
15.7.8.5.1.10. Viral
Safety Testing and Lot Release Testing
15.7.8.5.1.11. Others
(Water Analysis and etc.)
15.7.8.5.2. Pharmaceutical Chemistry
15.7.8.5.2.1. Elemental
Impurities
15.7.8.5.2.2. Volatile
Organic Impurities
15.7.8.5.2.3. Extractables
and Leachables
15.7.8.5.2.4. Semi-Volatile
Organic Impurites
15.7.8.5.2.5. Non-Volatile
Organic Impurites
15.7.8.5.2.6. Counterion
Analysis
15.7.8.5.2.7. Others
15.8. Key Segment for Channeling Investments
15.8.1. By Country
15.8.2. By Application
15.8.3. By Offering
15.8.4. By Techniques
15.8.5. By Testing Categories
15.8.6. By Analysis Type
16. Middle East and Africa Pharmaceutical Quality Control Market
Analysis and Forecasts, 2021 - 2029
16.1. Overview
16.1.1. Middle East and Africa Pharmaceutical Quality Control Market
Revenue (US$ Mn)
16.2. Middle East and Africa Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By Application
16.2.1. Allergens, antigens, vaccines and hormones
16.2.2. Cytokines, enzymes and human whole-blood
16.2.3. Plasma derivative
16.2.4. Immune sera, immunoglobulins and products of fermentation
16.2.5. Others (diagnostic agents for in vitro use, etc.)
16.3. Middle East and Africa Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By Offering
16.3.1. Consumables and Instruments
16.3.2. Solution
16.3.3. Services
16.4. Middle East and Africa Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By
Techniques
16.4.1. GC/MS
16.4.2. LC/MS/ High Performance
Liquid (HPLC)
16.4.3. Ultra High Performance Liquid (UHPLC)
16.4.4. Supercritical Fluid (SFC) chromatography
16.4.5. Elemental analysis techniques
16.4.6. Others
16.5. Middle East and Africa Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By Testing Categories
16.5.1. Elemental Impurities
16.5.2. Volatile Organic Impurities
16.5.3. Extractables and Leachables
16.5.4. Semi-Volatile Organic Impurites
16.5.5. Non-Volatile Organic Impurites
16.5.6. Counterion Analysis
16.5.7. Others
16.6. Middle East and Africa Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By Analysis Type
16.6.1. Pharmaceutical Microbiology
16.6.1.1. Sterility Testing
16.6.1.2. Antimicrobial Efficacy Testing (AET)
16.6.1.3. Microbial Limits Testing
16.6.1.4. Bioburden Determination
16.6.1.5. Endotoxin Testing
16.6.1.6. Environmental Monitoring and Identification
16.6.1.7. Packaging Testing
16.6.1.8. Pharmaceutical and Biopharmaceutical Release Testing
16.6.1.9. Pharmaceutical Gas Testing
16.6.1.10.
Viral Safety Testing and Lot
Release Testing
16.6.1.11.
Others (Water Analysis and etc.)
16.6.2. Pharmaceutical Chemistry
16.6.2.1. Elemental Impurities
16.6.2.2. Volatile Organic Impurities
16.6.2.3. Extractables and Leachables
16.6.2.4. Semi-Volatile Organic Impurites
16.6.2.5. Non-Volatile Organic Impurites
16.6.2.6. Counterion Analysis
16.6.2.7. Others
16.7. Middle East and Africa Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By Country
16.7.1. Saudi Arabia
16.7.1.1. Saudi Arabia Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Application
16.7.1.1.1. Allergens, antigens, vaccines and hormones
16.7.1.1.2. Cytokines, enzymes and human whole-blood
16.7.1.1.3. Plasma derivative
16.7.1.1.4. Immune sera, immunoglobulins and products of fermentation
16.7.1.1.5. Others (diagnostic agents for in vitro use, etc.)
16.7.1.2. Saudi Arabia Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Offering
16.7.1.2.1. Consumables and Instruments
16.7.1.2.2. Solution
16.7.1.2.3. Services
16.7.1.3. Saudi Arabia Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Techniques
16.7.1.3.1. GC/MS
16.7.1.3.2. LC/MS/ High Performance
Liquid (HPLC)
16.7.1.3.3. Ultra High Performance Liquid (UHPLC)
16.7.1.3.4. Supercritical Fluid (SFC) chromatography
16.7.1.3.5. Elemental analysis techniques
16.7.1.3.6. Others
16.7.1.4. Saudi Arabia Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Testing Categories
16.7.1.4.1. Elemental Impurities
16.7.1.4.2. Volatile Organic Impurities
16.7.1.4.3. Extractables and Leachables
16.7.1.4.4. Semi-Volatile Organic Impurites
16.7.1.4.5. Non-Volatile Organic Impurites
16.7.1.4.6. Counterion Analysis
16.7.1.4.7. Others
16.7.1.5. Saudi Arabia Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Analysis Type
16.7.1.5.1. Pharmaceutical Microbiology
16.7.1.5.1.1. Sterility
Testing
16.7.1.5.1.2. Antimicrobial
Efficacy Testing (AET)
16.7.1.5.1.3. Microbial
Limits Testing
16.7.1.5.1.4. Bioburden
Determination
16.7.1.5.1.5. Endotoxin
Testing
16.7.1.5.1.6. Environmental
Monitoring and Identification
16.7.1.5.1.7. Packaging
Testing
16.7.1.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
16.7.1.5.1.9. Pharmaceutical
Gas Testing
16.7.1.5.1.10. Viral
Safety Testing and Lot Release Testing
16.7.1.5.1.11. Others
(Water Analysis and etc.)
16.7.1.5.2. Pharmaceutical Chemistry
16.7.1.5.2.1. Elemental
Impurities
16.7.1.5.2.2. Volatile
Organic Impurities
16.7.1.5.2.3. Extractables
and Leachables
16.7.1.5.2.4. Semi-Volatile
Organic Impurites
16.7.1.5.2.5. Non-Volatile
Organic Impurites
16.7.1.5.2.6. Counterion
Analysis
16.7.1.5.2.7. Others
16.7.2. UAE
16.7.2.1. UAE Pharmaceutical Quality Control Market Revenue (US$ Mn) and
Forecasts, By Application
16.7.2.1.1. Allergens, antigens, vaccines and hormones
16.7.2.1.2. Cytokines, enzymes and human whole-blood
16.7.2.1.3. Plasma derivative
16.7.2.1.4. Immune sera, immunoglobulins and products of fermentation
16.7.2.1.5. Others (diagnostic agents for in vitro use, etc.)
16.7.2.2. UAE Pharmaceutical Quality Control Market Revenue (US$ Mn) and
Forecasts, By Offering
16.7.2.2.1. Consumables and Instruments
16.7.2.2.2. Solution
16.7.2.2.3. Services
16.7.2.3. UAE Pharmaceutical Quality Control Market Revenue (US$ Mn) and
Forecasts, By Techniques
16.7.2.3.1. GC/MS
16.7.2.3.2. LC/MS/ High Performance
Liquid (HPLC)
16.7.2.3.3. Ultra High Performance Liquid (UHPLC)
16.7.2.3.4. Supercritical Fluid (SFC) chromatography
16.7.2.3.5. Elemental analysis techniques
16.7.2.3.6. Others
16.7.2.4. UAE Pharmaceutical Quality Control Market Revenue (US$ Mn) and
Forecasts, By Testing Categories
16.7.2.4.1. Elemental Impurities
16.7.2.4.2. Volatile Organic Impurities
16.7.2.4.3. Extractables and Leachables
16.7.2.4.4. Semi-Volatile Organic Impurites
16.7.2.4.5. Non-Volatile Organic Impurites
16.7.2.4.6. Counterion Analysis
16.7.2.4.7. Others
16.7.2.5. UAE Pharmaceutical Quality Control Market Revenue (US$ Mn) and
Forecasts, By Analysis Type
16.7.2.5.1. Pharmaceutical Microbiology
16.7.2.5.1.1. Sterility
Testing
16.7.2.5.1.2. Antimicrobial
Efficacy Testing (AET)
16.7.2.5.1.3. Microbial
Limits Testing
16.7.2.5.1.4. Bioburden
Determination
16.7.2.5.1.5. Endotoxin
Testing
16.7.2.5.1.6. Environmental
Monitoring and Identification
16.7.2.5.1.7. Packaging
Testing
16.7.2.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
16.7.2.5.1.9. Pharmaceutical
Gas Testing
16.7.2.5.1.10. Viral
Safety Testing and Lot Release Testing
16.7.2.5.1.11. Others
(Water Analysis and etc.)
16.7.2.5.2. Pharmaceutical Chemistry
16.7.2.5.2.1. Elemental
Impurities
16.7.2.5.2.2. Volatile
Organic Impurities
16.7.2.5.2.3. Extractables
and Leachables
16.7.2.5.2.4. Semi-Volatile
Organic Impurites
16.7.2.5.2.5. Non-Volatile
Organic Impurites
16.7.2.5.2.6. Counterion
Analysis
16.7.2.5.2.7. Others
16.7.3. Egypt
16.7.3.1. Egypt Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Application
16.7.3.1.1. Allergens, antigens, vaccines and hormones
16.7.3.1.2. Cytokines, enzymes and human whole-blood
16.7.3.1.3. Plasma derivative
16.7.3.1.4. Immune sera, immunoglobulins and products of fermentation
16.7.3.1.5. Others (diagnostic agents for in vitro use, etc.)
16.7.3.2. Egypt Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Offering
16.7.3.2.1. Consumables and Instruments
16.7.3.2.2. Solution
16.7.3.2.3. Services
16.7.3.3. Egypt Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Techniques
16.7.3.3.1. GC/MS
16.7.3.3.2. LC/MS/ High Performance
Liquid (HPLC)
16.7.3.3.3. Ultra High Performance Liquid (UHPLC)
16.7.3.3.4. Supercritical Fluid (SFC) chromatography
16.7.3.3.5. Elemental analysis techniques
16.7.3.3.6. Others
16.7.3.4. Egypt Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Testing Categories
16.7.3.4.1. Elemental Impurities
16.7.3.4.2. Volatile Organic Impurities
16.7.3.4.3. Extractables and Leachables
16.7.3.4.4. Semi-Volatile Organic Impurites
16.7.3.4.5. Non-Volatile Organic Impurites
16.7.3.4.6. Counterion Analysis
16.7.3.4.7. Others
16.7.3.5. Egypt Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Analysis Type
16.7.3.5.1. Pharmaceutical Microbiology
16.7.3.5.1.1. Sterility
Testing
16.7.3.5.1.2. Antimicrobial
Efficacy Testing (AET)
16.7.3.5.1.3. Microbial
Limits Testing
16.7.3.5.1.4. Bioburden
Determination
16.7.3.5.1.5. Endotoxin
Testing
16.7.3.5.1.6. Environmental
Monitoring and Identification
16.7.3.5.1.7. Packaging
Testing
16.7.3.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
16.7.3.5.1.9. Pharmaceutical
Gas Testing
16.7.3.5.1.10. Viral
Safety Testing and Lot Release Testing
16.7.3.5.1.11. Others
(Water Analysis and etc.)
16.7.3.5.2. Pharmaceutical Chemistry
16.7.3.5.2.1. Elemental
Impurities
16.7.3.5.2.2. Volatile
Organic Impurities
16.7.3.5.2.3. Extractables
and Leachables
16.7.3.5.2.4. Semi-Volatile
Organic Impurites
16.7.3.5.2.5. Non-Volatile
Organic Impurites
16.7.3.5.2.6. Counterion
Analysis
16.7.3.5.2.7. Others
16.7.4. Kuwait
16.7.4.1. Kuwait Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Application
16.7.4.1.1. Allergens, antigens, vaccines and hormones
16.7.4.1.2. Cytokines, enzymes and human whole-blood
16.7.4.1.3. Plasma derivative
16.7.4.1.4. Immune sera, immunoglobulins and products of fermentation
16.7.4.1.5. Others (diagnostic agents for in vitro use, etc.)
16.7.4.2. Kuwait Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Offering
16.7.4.2.1. Consumables and Instruments
16.7.4.2.2. Solution
16.7.4.2.3. Services
16.7.4.3. Kuwait Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Techniques
16.7.4.3.1. GC/MS
16.7.4.3.2. LC/MS/ High Performance
Liquid (HPLC)
16.7.4.3.3. Ultra High Performance Liquid (UHPLC)
16.7.4.3.4. Supercritical Fluid (SFC) chromatography
16.7.4.3.5. Elemental analysis techniques
16.7.4.3.6. Others
16.7.4.4. Kuwait Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Testing Categories
16.7.4.4.1. Elemental Impurities
16.7.4.4.2. Volatile Organic Impurities
16.7.4.4.3. Extractables and Leachables
16.7.4.4.4. Semi-Volatile Organic Impurites
16.7.4.4.5. Non-Volatile Organic Impurites
16.7.4.4.6. Counterion Analysis
16.7.4.4.7. Others
16.7.4.5. Kuwait Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Analysis Type
16.7.4.5.1. Pharmaceutical Microbiology
16.7.4.5.1.1. Sterility
Testing
16.7.4.5.1.2. Antimicrobial
Efficacy Testing (AET)
16.7.4.5.1.3. Microbial
Limits Testing
16.7.4.5.1.4. Bioburden
Determination
16.7.4.5.1.5. Endotoxin
Testing
16.7.4.5.1.6. Environmental
Monitoring and Identification
16.7.4.5.1.7. Packaging
Testing
16.7.4.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
16.7.4.5.1.9. Pharmaceutical
Gas Testing
16.7.4.5.1.10. Viral
Safety Testing and Lot Release Testing
16.7.4.5.1.11. Others
(Water Analysis and etc.)
16.7.4.5.2. Pharmaceutical Chemistry
16.7.4.5.2.1. Elemental
Impurities
16.7.4.5.2.2. Volatile
Organic Impurities
16.7.4.5.2.3. Extractables
and Leachables
16.7.4.5.2.4. Semi-Volatile
Organic Impurites
16.7.4.5.2.5. Non-Volatile
Organic Impurites
16.7.4.5.2.6. Counterion
Analysis
16.7.4.5.2.7. Others
16.7.5. South Africa
16.7.5.1. South Africa Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Application
16.7.5.1.1. Allergens, antigens, vaccines and hormones
16.7.5.1.2. Cytokines, enzymes and human whole-blood
16.7.5.1.3. Plasma derivative
16.7.5.1.4. Immune sera, immunoglobulins and products of fermentation
16.7.5.1.5. Others (diagnostic agents for in vitro use, etc.)
16.7.5.2. South Africa Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Offering
16.7.5.2.1. Consumables and Instruments
16.7.5.2.2. Solution
16.7.5.2.3. Services
16.7.5.3. South Africa Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Techniques
16.7.5.3.1. GC/MS
16.7.5.3.2. LC/MS/ High Performance
Liquid (HPLC)
16.7.5.3.3. Ultra High Performance Liquid (UHPLC)
16.7.5.3.4. Supercritical Fluid (SFC) chromatography
16.7.5.3.5. Elemental analysis techniques
16.7.5.3.6. Others
16.7.5.4. South Africa Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Testing Categories
16.7.5.4.1. Elemental Impurities
16.7.5.4.2. Volatile Organic Impurities
16.7.5.4.3. Extractables and Leachables
16.7.5.4.4. Semi-Volatile Organic Impurites
16.7.5.4.5. Non-Volatile Organic Impurites
16.7.5.4.6. Counterion Analysis
16.7.5.4.7. Others
16.7.5.5. South Africa Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Analysis Type
16.7.5.5.1. Pharmaceutical Microbiology
16.7.5.5.1.1. Sterility
Testing
16.7.5.5.1.2. Antimicrobial
Efficacy Testing (AET)
16.7.5.5.1.3. Microbial
Limits Testing
16.7.5.5.1.4. Bioburden
Determination
16.7.5.5.1.5. Endotoxin
Testing
16.7.5.5.1.6. Environmental
Monitoring and Identification
16.7.5.5.1.7. Packaging
Testing
16.7.5.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
16.7.5.5.1.9. Pharmaceutical
Gas Testing
16.7.5.5.1.10. Viral
Safety Testing and Lot Release Testing
16.7.5.5.1.11. Others
(Water Analysis and etc.)
16.7.5.5.2. Pharmaceutical Chemistry
16.7.5.5.2.1. Elemental
Impurities
16.7.5.5.2.2. Volatile
Organic Impurities
16.7.5.5.2.3. Extractables
and Leachables
16.7.5.5.2.4. Semi-Volatile
Organic Impurites
16.7.5.5.2.5. Non-Volatile
Organic Impurites
16.7.5.5.2.6. Counterion
Analysis
16.7.5.5.2.7. Others
16.7.6. Rest of Middle East & Africa
16.7.6.1. Rest of Middle East & Africa Pharmaceutical Quality Control
Market Revenue (US$ Mn) and Forecasts, By Application
16.7.6.1.1. Allergens, antigens, vaccines and hormones
16.7.6.1.2. Cytokines, enzymes and human whole-blood
16.7.6.1.3. Plasma derivative
16.7.6.1.4. Immune sera, immunoglobulins and products of fermentation
16.7.6.1.5. Others (diagnostic agents for in vitro use, etc.)
16.7.6.2. Rest of Middle East & Africa Pharmaceutical Quality
Control Market Revenue (US$ Mn) and Forecasts, By Offering
16.7.6.2.1. Consumables and Instruments
16.7.6.2.2. Solution
16.7.6.2.3. Services
16.7.6.3. Rest of Middle East & Africa Pharmaceutical Quality
Control Market Revenue (US$ Mn) and Forecasts, By Techniques
16.7.6.3.1. GC/MS
16.7.6.3.2. LC/MS/ High Performance
Liquid (HPLC)
16.7.6.3.3. Ultra High Performance Liquid (UHPLC)
16.7.6.3.4. Supercritical Fluid (SFC) chromatography
16.7.6.3.5. Elemental analysis techniques
16.7.6.3.6. Others
16.7.6.4. Rest of Middle East & Africa Pharmaceutical Quality
Control Market Revenue (US$ Mn) and Forecasts, By Testing Categories
16.7.6.4.1. Elemental Impurities
16.7.6.4.2. Volatile Organic Impurities
16.7.6.4.3. Extractables and Leachables
16.7.6.4.4. Semi-Volatile Organic Impurites
16.7.6.4.5. Non-Volatile Organic Impurites
16.7.6.4.6. Counterion Analysis
16.7.6.4.7. Others
16.7.6.5. Rest of Middle East & Africa Pharmaceutical Quality
Control Market Revenue (US$ Mn) and Forecasts, By Analysis Type
16.7.6.5.1. Pharmaceutical Microbiology
16.7.6.5.1.1. Sterility
Testing
16.7.6.5.1.2. Antimicrobial
Efficacy Testing (AET)
16.7.6.5.1.3. Microbial
Limits Testing
16.7.6.5.1.4. Bioburden
Determination
16.7.6.5.1.5. Endotoxin
Testing
16.7.6.5.1.6. Environmental
Monitoring and Identification
16.7.6.5.1.7. Packaging
Testing
16.7.6.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
16.7.6.5.1.9. Pharmaceutical
Gas Testing
16.7.6.5.1.10. Viral
Safety Testing and Lot Release Testing
16.7.6.5.1.11. Others
(Water Analysis and etc.)
16.7.6.5.2. Pharmaceutical Chemistry
16.7.6.5.2.1. Elemental
Impurities
16.7.6.5.2.2. Volatile
Organic Impurities
16.7.6.5.2.3. Extractables
and Leachables
16.7.6.5.2.4. Semi-Volatile
Organic Impurites
16.7.6.5.2.5. Non-Volatile
Organic Impurites
16.7.6.5.2.6. Counterion
Analysis
16.7.6.5.2.7. Others
16.8. Key Segment for Channeling Investments
16.8.1. By Country
16.8.2. By Application
16.8.3. By Offering
16.8.4. By Techniques
16.8.5. By Testing Categories
16.8.6. By Analysis Type
17. Latin America Pharmaceutical Quality Control Market Analysis
and Forecasts, 2021 - 2029
17.1. Overview
17.1.1. Latin America Pharmaceutical Quality Control Market Revenue
(US$ Mn)
17.2. Latin America Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Application
17.2.1. Allergens, antigens, vaccines and hormones
17.2.2. Cytokines, enzymes and human whole-blood
17.2.3. Plasma derivative
17.2.4. Immune sera, immunoglobulins and products of fermentation
17.2.5. Others (diagnostic agents for in vitro use, etc.)
17.3. Latin America Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Offering
17.3.1. Consumables and Instruments
17.3.2. Solution
17.3.3. Services
17.4. Latin America Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Techniques
17.4.1. GC/MS
17.4.2. LC/MS/ High Performance
Liquid (HPLC)
17.4.3. Ultra High Performance Liquid (UHPLC)
17.4.4. Supercritical Fluid (SFC) chromatography
17.4.5. Elemental analysis techniques
17.4.6. Others
17.5. Latin America Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Testing Categories
17.5.1. Elemental Impurities
17.5.2. Volatile Organic Impurities
17.5.3. Extractables and Leachables
17.5.4. Semi-Volatile Organic Impurites
17.5.5. Non-Volatile Organic Impurites
17.5.6. Counterion Analysis
17.5.7. Others
17.6. Latin America Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Analysis Type
17.6.1. Pharmaceutical Microbiology
17.6.1.1. Sterility Testing
17.6.1.2. Antimicrobial Efficacy Testing (AET)
17.6.1.3. Microbial Limits Testing
17.6.1.4. Bioburden Determination
17.6.1.5. Endotoxin Testing
17.6.1.6. Environmental Monitoring and Identification
17.6.1.7. Packaging Testing
17.6.1.8. Pharmaceutical and Biopharmaceutical Release Testing
17.6.1.9. Pharmaceutical Gas Testing
17.6.1.10.
Viral Safety Testing and Lot
Release Testing
17.6.1.11.
Others (Water Analysis and etc.)
17.6.2. Pharmaceutical Chemistry
17.6.2.1. Elemental Impurities
17.6.2.2. Volatile Organic Impurities
17.6.2.3. Extractables and Leachables
17.6.2.4. Semi-Volatile Organic Impurites
17.6.2.5. Non-Volatile Organic Impurites
17.6.2.6. Counterion Analysis
17.6.2.7. Others
17.7. Latin America Pharmaceutical Quality Control Market Revenue
(US$ Mn) and Forecasts, By Country
17.7.1. Brazil
17.7.1.1. Brazil Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Application
17.7.1.1.1. Allergens, antigens, vaccines and hormones
17.7.1.1.2. Cytokines, enzymes and human whole-blood
17.7.1.1.3. Plasma derivative
17.7.1.1.4. Immune sera, immunoglobulins and products of fermentation
17.7.1.1.5. Others (diagnostic agents for in vitro use, etc.)
17.7.1.2. Brazil Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Offering
17.7.1.2.1. Consumables and Instruments
17.7.1.2.2. Solution
17.7.1.2.3. Services
17.7.1.3. Brazil Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Techniques
17.7.1.3.1. GC/MS
17.7.1.3.2. LC/MS/ High Performance
Liquid (HPLC)
17.7.1.3.3. Ultra High Performance Liquid (UHPLC)
17.7.1.3.4. Supercritical Fluid (SFC) chromatography
17.7.1.3.5. Elemental analysis techniques
17.7.1.3.6. Others
17.7.1.4. Brazil Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Testing Categories
17.7.1.4.1. Elemental Impurities
17.7.1.4.2. Volatile Organic Impurities
17.7.1.4.3. Extractables and Leachables
17.7.1.4.4. Semi-Volatile Organic Impurites
17.7.1.4.5. Non-Volatile Organic Impurites
17.7.1.4.6. Counterion Analysis
17.7.1.4.7. Others
17.7.1.5. Brazil Pharmaceutical Quality Control Market Revenue (US$ Mn)
and Forecasts, By Analysis Type
17.7.1.5.1. Pharmaceutical Microbiology
17.7.1.5.1.1. Sterility
Testing
17.7.1.5.1.2. Antimicrobial
Efficacy Testing (AET)
17.7.1.5.1.3. Microbial
Limits Testing
17.7.1.5.1.4. Bioburden
Determination
17.7.1.5.1.5. Endotoxin
Testing
17.7.1.5.1.6. Environmental
Monitoring and Identification
17.7.1.5.1.7. Packaging
Testing
17.7.1.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
17.7.1.5.1.9. Pharmaceutical
Gas Testing
17.7.1.5.1.10. Viral
Safety Testing and Lot Release Testing
17.7.1.5.1.11. Others
(Water Analysis and etc.)
17.7.1.5.2. Pharmaceutical Chemistry
17.7.1.5.2.1. Elemental
Impurities
17.7.1.5.2.2. Volatile
Organic Impurities
17.7.1.5.2.3. Extractables
and Leachables
17.7.1.5.2.4. Semi-Volatile
Organic Impurites
17.7.1.5.2.5. Non-Volatile
Organic Impurites
17.7.1.5.2.6. Counterion
Analysis
17.7.1.5.2.7. Others
17.7.2. Argentina
17.7.2.1. Argentina Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Application
17.7.2.1.1. Allergens, antigens, vaccines and hormones
17.7.2.1.2. Cytokines, enzymes and human whole-blood
17.7.2.1.3. Plasma derivative
17.7.2.1.4. Immune sera, immunoglobulins and products of fermentation
17.7.2.1.5. Others (diagnostic agents for in vitro use, etc.)
17.7.2.2. Argentina Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Offering
17.7.2.2.1. Consumables and Instruments
17.7.2.2.2. Solution
17.7.2.2.3. Services
17.7.2.3. Argentina Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Techniques
17.7.2.3.1. GC/MS
17.7.2.3.2. LC/MS/ High Performance
Liquid (HPLC)
17.7.2.3.3. Ultra High Performance Liquid (UHPLC)
17.7.2.3.4. Supercritical Fluid (SFC) chromatography
17.7.2.3.5. Elemental analysis techniques
17.7.2.3.6. Others
17.7.2.4. Argentina Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Testing Categories
17.7.2.4.1. Elemental Impurities
17.7.2.4.2. Volatile Organic Impurities
17.7.2.4.3. Extractables and Leachables
17.7.2.4.4. Semi-Volatile Organic Impurites
17.7.2.4.5. Non-Volatile Organic Impurites
17.7.2.4.6. Counterion Analysis
17.7.2.4.7. Others
17.7.2.5. Argentina Pharmaceutical Quality Control Market Revenue (US$
Mn) and Forecasts, By Analysis Type
17.7.2.5.1. Pharmaceutical Microbiology
17.7.2.5.1.1. Sterility
Testing
17.7.2.5.1.2. Antimicrobial
Efficacy Testing (AET)
17.7.2.5.1.3. Microbial
Limits Testing
17.7.2.5.1.4. Bioburden
Determination
17.7.2.5.1.5. Endotoxin
Testing
17.7.2.5.1.6. Environmental
Monitoring and Identification
17.7.2.5.1.7. Packaging
Testing
17.7.2.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
17.7.2.5.1.9. Pharmaceutical
Gas Testing
17.7.2.5.1.10. Viral
Safety Testing and Lot Release Testing
17.7.2.5.1.11. Others
(Water Analysis and etc.)
17.7.2.5.2. Pharmaceutical Chemistry
17.7.2.5.2.1. Elemental
Impurities
17.7.2.5.2.2. Volatile
Organic Impurities
17.7.2.5.2.3. Extractables
and Leachables
17.7.2.5.2.4. Semi-Volatile
Organic Impurites
17.7.2.5.2.5. Non-Volatile
Organic Impurites
17.7.2.5.2.6. Counterion
Analysis
17.7.2.5.2.7. Others
17.7.3. Rest of Latin America
17.7.3.1. Rest of Latin America Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By Application
17.7.3.1.1. Allergens, antigens, vaccines and hormones
17.7.3.1.2. Cytokines, enzymes and human whole-blood
17.7.3.1.3. Plasma derivative
17.7.3.1.4. Immune sera, immunoglobulins and products of fermentation
17.7.3.1.5. Others (diagnostic agents for in vitro use, etc.)
17.7.3.2. Rest of Latin America Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By Offering
17.7.3.2.1. Consumables and Instruments
17.7.3.2.2. Solution
17.7.3.2.3. Services
17.7.3.3. Rest of Latin America Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By
Techniques
17.7.3.3.1. GC/MS
17.7.3.3.2. LC/MS/ High Performance
Liquid (HPLC)
17.7.3.3.3. Ultra High Performance Liquid (UHPLC)
17.7.3.3.4. Supercritical Fluid (SFC) chromatography
17.7.3.3.5. Elemental analysis techniques
17.7.3.3.6. Others
17.7.3.4. Rest of Latin America Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By Testing Categories
17.7.3.4.1. Elemental Impurities
17.7.3.4.2. Volatile Organic Impurities
17.7.3.4.3. Extractables and Leachables
17.7.3.4.4. Semi-Volatile Organic Impurites
17.7.3.4.5. Non-Volatile Organic Impurites
17.7.3.4.6. Counterion Analysis
17.7.3.4.7. Others
17.7.3.5. Rest of Latin America Pharmaceutical Quality Control Market
Revenue (US$ Mn) and Forecasts, By Analysis Type
17.7.3.5.1. Pharmaceutical Microbiology
17.7.3.5.1.1. Sterility
Testing
17.7.3.5.1.2. Antimicrobial
Efficacy Testing (AET)
17.7.3.5.1.3. Microbial
Limits Testing
17.7.3.5.1.4. Bioburden
Determination
17.7.3.5.1.5. Endotoxin
Testing
17.7.3.5.1.6. Environmental
Monitoring and Identification
17.7.3.5.1.7. Packaging
Testing
17.7.3.5.1.8. Pharmaceutical
and Biopharmaceutical Release Testing
17.7.3.5.1.9. Pharmaceutical
Gas Testing
17.7.3.5.1.10. Viral
Safety Testing and Lot Release Testing
17.7.3.5.1.11. Others
(Water Analysis and etc.)
17.7.3.5.2. Pharmaceutical Chemistry
17.7.3.5.2.1. Elemental
Impurities
17.7.3.5.2.2. Volatile
Organic Impurities
17.7.3.5.2.3. Extractables
and Leachables
17.7.3.5.2.4. Semi-Volatile
Organic Impurites
17.7.3.5.2.5. Non-Volatile
Organic Impurites
17.7.3.5.2.6. Counterion
Analysis
17.7.3.5.2.7. Others
17.8. Key Segment for Channeling Investments
17.8.1. By Country
17.8.2. By Application
17.8.3. By Offering
17.8.4. By Techniques
17.8.5. By Testing Categories
17.8.6. By Analysis Type
18. Competitive Benchmarking
18.1. Market Share Analysis, 2020
18.2. Global Presence and Growth Strategies
18.2.1. Mergers and Acquisitions
18.2.2. Product Launches
18.2.3. Investments Trends
18.2.4. R&D Initiatives
19. Player Profiles
19.1. Lucideon Limited
19.1.1. Company Details
19.1.2. Company Overview
19.1.3. Product Offerings
19.1.4. Key Developments
19.1.5. Financial Analysis
19.1.6. SWOT Analysis
19.1.7. Business Strategies
19.2. ThermoFisher Scientific
19.2.1. Company Details
19.2.2. Company Overview
19.2.3. Product Offerings
19.2.4. Key Developments
19.2.5. Financial Analysis
19.2.6. SWOT Analysis
19.2.7. Business Strategies
19.3. Merck KGaA
19.3.1. Company Details
19.3.2. Company Overview
19.3.3. Product Offerings
19.3.4. Key Developments
19.3.5. Financial Analysis
19.3.6. SWOT Analysis
19.3.7. Business Strategies
19.4. Charles River Laboratories
19.4.1. Company Details
19.4.2. Company Overview
19.4.3. Product Offerings
19.4.4. Key Developments
19.4.5. Financial Analysis
19.4.6. SWOT Analysis
19.4.7. Business Strategies
19.5. Eurofins Scientific
19.5.1. Company Details
19.5.2. Company Overview
19.5.3. Product Offerings
19.5.4. Key Developments
19.5.5. Financial Analysis
19.5.6. SWOT Analysis
19.5.7. Business Strategies
19.6. SGS Group Management SA
19.6.1. Company Details
19.6.2. Company Overview
19.6.3. Product Offerings
19.6.4. Key Developments
19.6.5. Financial Analysis
19.6.6. SWOT Analysis
19.6.7. Business Strategies
19.7. Toxikon Corporation
19.7.1. Company Details
19.7.2. Company Overview
19.7.3. Product Offerings
19.7.4. Key Developments
19.7.5. Financial Analysis
19.7.6. SWOT Analysis
19.7.7. Business Strategies
19.8. Sartorius AG
19.8.1. Company Details
19.8.2. Company Overview
19.8.3. Product Offerings
19.8.4. Key Developments
19.8.5. Financial Analysis
19.8.6. SWOT Analysis
19.8.7. Business Strategies
19.9. Other Market Participants
20. Key Findings
At Absolute Markets Insights, we are engaged in building both global as well as country specific reports. As a result, the approach taken for deriving the estimation and forecast for a specific country is a bit unique and different in comparison to the global research studies. In this case, we not only study the concerned market factors & trends prevailing in a particular country (from secondary research) but we also tend to calculate the actual market size & forecast from the revenue generated from the market participants involved in manufacturing or distributing the any concerned product. These companies can also be service providers. For analyzing any country specifically, we do consider the growth factors prevailing under the states/cities/county for the same. For instance, if we are analyzing an industry specific to United States, we primarily need to study about the states present under the same(where the product/service has the highest growth). Similar analysis will be followed by other countries. Our scope of the report changes with different markets.
Our research study is mainly implement through a mix of both secondary and primary research. Various sources such as industry magazines, trade journals, and government websites and trade associations are reviewed for gathering precise data. Primary interviews are conducted to validate the market size derived from secondary research. Industry experts, major manufacturers and distributors are contacted for further validation purpose on the current market penetration and growth trends.
Prominent participants in our primary research process include:
- Key Opinion Leaders namely the CEOs, CSOs, VPs, purchasing managers, amongst others
- Research and development participants, distributors/suppliers and subject matter experts
Secondary Research includes data extracted from paid data sources:
- Reuters
- Factiva
- Bloomberg
- One Source
- Hoovers
Research Methodology
Key Inclusions

Reach to us
Call us on
+91-74002-42424
Drop us an email at
sales@absolutemarketsinsights.com
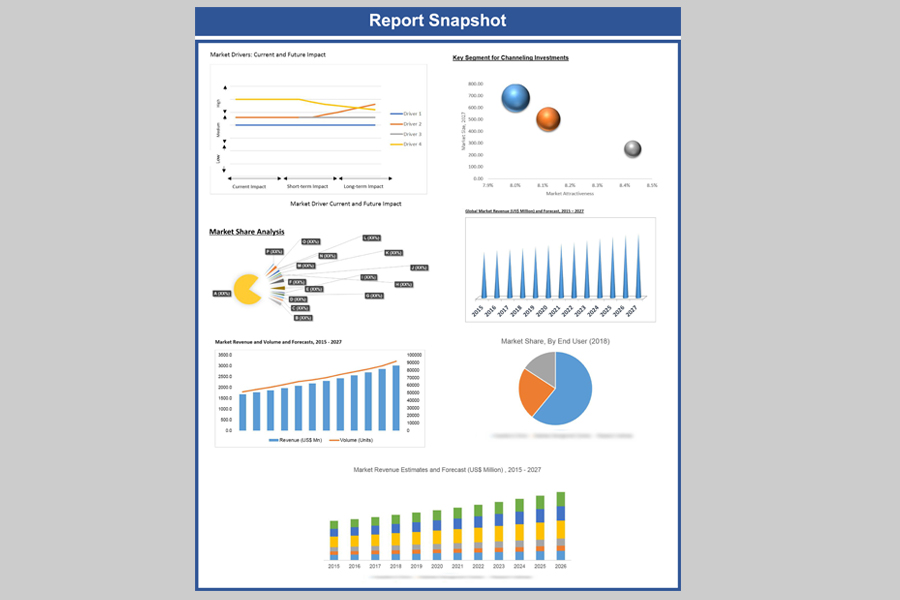
Why Absolute Markets Insights?
An effective strategy is the entity that influences a business to stand out of the crowd. An organization with a phenomenal strategy for success dependably has the edge over the rivals in the market. It offers the organizations a head start in planning their strategy. Absolute Market Insights is the new initiation in the industry that will furnish you with the lead your business needs. Absolute Market Insights is the best destination for your business intelligence and analytical solutions; essentially because our qualitative and quantitative sources of information are competent to give one-stop solutions. We inventively combine qualitative and quantitative research in accurate proportions to have the best report, which not only gives the most recent insights but also assists you to grow.