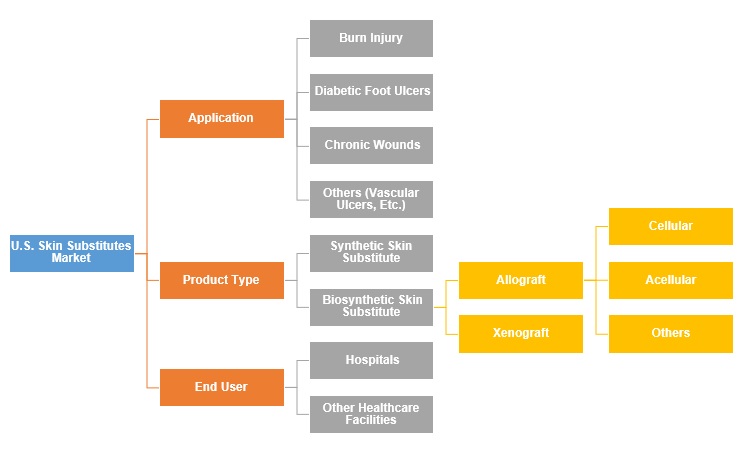
Skin Substitutes Market by Application (Burn Injury, Diabetic Foot Ulcers, Chronic Wounds, Others (Vascular Ulcers, Etc.); by Product Type (Synthetic Skin Substitute, Biosynthetic Skin Substitute, Biological Skin Substitute, Allograft {Cellular, Acellular, Others}, Xenograft); by End User (Hospitals, Other Healthcare Facilities) – U.S. Insights, Growth, Size, Comparative Analysis, Trends and Forecast, 2018-2026
Industry Trends
Skin is the largest organ in the human body and is made up of two main layers, the upper one known as epidermis and the lower one known as dermis. It is in direct contact with the outside world and this makes it most susceptible to injuries or damages. Hence, substitutes are necessary in case of skin wounds and disorders. Skin substitutes have been used as a form of wound closure since the earliest centuries of medical science. Substitutes are either permanent or temporary depending on their time period. Temporary substitutes are used for a short period to protect the wound area from bacterial contamination and external trauma and to promote easy wound healing. On the other hand, permanent skin substitutes are used to replace the full thickness of skin layers and improve the quality of skin at the wound or burn sites permanently. The usage of skin substitutes has shown to accelerate the wound healing thus helping to cope up with the problems of dehydration and infection. As per the American Burn Association (ABA), about 1 million burn injuries occur annually in the United States. This number has increased in the recent years due to which United States has become a prominent market for skin substitutes.
U.S. Skin Substitutes Market, By End User, 2017 & 2022 (USD Million)
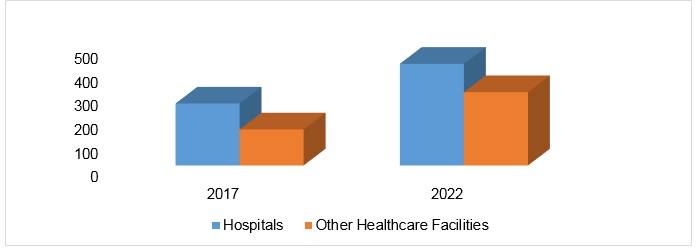
Plastic surgeons use skin substitutes to treat a wide variety of cutaneous defects as they are useful in healing wounds and minimizing the donor sites. They are commonly used for application in burns, acute wounds and chronic wounds. The skin substitutes are numerous and varied. The choice of skin substitutes depends on many factors which primarily includes the normal skin anatomy, the patient’s condition, medical and surgical comorbidities, the amount of skin requiring replacement and the level of contamination of the wound. The U.S. skin substitutes market was valued to be US$ 357.38 Mn in 2017 and is anticipated to be US$ 734.74 Mn by 2022.
Skin Substitutes Market, by Product Type
Among all the skin substitutes available in the market for wound healing and skin grafting, synthetic ones are the most preferred. They are generally artificially produced from synthesized materials, which are made on demand with specific characteristics required as per a patient’s skin conditions and purchasing power. Biobrane, Integra, Alloderm, and TransCyte are most commonly used synthetic skin substitutes.
Biobrane is composed of an ultrathin silicone film on its outer surface and a nylon filament on the inner side, upon which type I collagen peptides are strongly bonded. The semi-permeable silicone surface controls water vapor loss from the wound. It is therefore used as a temporary wound dressing and is removed upon healing or when autograft skin is available. It has been shown to reduce hospitalization time in case of pediatric patients with second degree burn injuries.
On the other hand, Integra is composed of a dermal layer made up of porous bovine collagen, chondroitin-6-sulfate GAG and an epidermal layer made of synthetic silicone polymer. It is currently used in patients with large burns and limited autograft donor sites.
The working of Alloderm is similar to that of Integra and has been used as a temporary wound cover in third degree burnt wounds. TransCyte’s composition is similar to that of Biobrane and is used as a temporary cover for excised burns that await for placement of natural skin substitutes.
Skin Substitutes Market, by Application
Skin substitutes are widely used in the burn injuries and have become a major part of the burn care strategy. Extensive burn wounds are difficult to manage and repair. Several engineered skin substitutes have been developed to aid in this process. Burn wounds are caused by damage to the skin due to heat, chemicals, electricity or radiation. The burns that damage the epidermal, dermal and hypodermal layers of the skin are mostly referred to as first, second and third-degree burns, respectively. Second and third degree burns mostly cause fluid loss, drastic disturbances of ionic equilibrium, loss of temperature control, pain, immunodepression, bacterial invasion and in some cases substantial or permanent disability. According to the latest statistics, the United States exhibits 450,000 burn injuries per annum that require medical attention. Nearly 40,000 of these patients require hospitalization. The cost of burden on the healthcare system due to severe burn injuries is significant, with per patient hospital charges ranging from US$ 27,000 to US$ 500, 000 for severe wounds. During acute phase of skin burn treatments, skin substitutes have improved functional and cosmetic results and thus have increased the quality of life. In case of chronic wound treatments, skin substitutes are being used to reconstruct and improve burnt scars and other defects.
Competitive Landscape
The United States skin substitutes market report provides both, qualitative and quantitative research of the market, as well as strategic insights along with developments that are being adopted by the key contenders. The report also offers extensive research on the key players in this market and detailed insights on the competitiveness of these players. The key business strategies such as mergers and acquisition (M&A), affiliations, collaborations, and contracts adopted by the major players are also recognized and analyzed in the report. For each company, the report recognizes their research institutes, competitors, product type, application and specification, pricing, and gross margin.
Skin substitutes market participants include Stratatech a Mallinckrodt, PolyNovo North America LLC, Organogenesis Inc., Mölnlycke Health Care AB, Medtronic (Covidien), Integra Life Sciences Corporation, Genzyme Corporation, Cook Biotech, Inc., BSN Medical, C. R. Bard, Inc., AMARANTUS BIOSCIENCE HOLDINGS, INC., AlloSource, Advanced Tissue, Acelity L.P. Inc. (KCI- Kinetic Concepts Inc.), Soluble Systems, LLC (Alliqua BioMedical, Inc.), Smith & Nephew plc., Shire Regenerative Medicine, Inc. and Tissue Regenix Group Plc amongst others. In January 2014, Shire Regenerative Medicine, Inc., that was formerly known as Advanced BioHealing, Inc. entered into an agreement with United States based Organogenesis Inc. and sold its DERMAGRAFT assets to the company. DERMAGRAFT is a living skin substitute for use in the treatment of full-thickness diabetic foot ulcers and is approved for use in the U.S. and Canada.
U.S. Skin Substitutes Market Background
1. Introduction
1.1. Market Scope
1.2. Market Segmentation
1.3. Methodology
1.4. Assumptions
2. Skin Substitutes Market Snapshot
3. Executive Summary: Skin Substitutes Market
4. Qualitative Analysis: Skin Substitutes Market
4.1. Introduction
4.1.1. Product Definition
4.1.2. Industry Development
4.2. Market Dynamics
4.2.1. Drivers
4.2.2. Restraints
4.2.3. Opportunities
4.3. Trends in Market
5. U.S. Skin Substitutes Market Analysis and Forecasts, 2018 –
2026
5.1. Overview
5.1.1. U.S. Market Revenue (US$ Mn) and Forecasts
5.2. U.S. Skin Substitutes Market Revenue (US$ Mn) and Forecasts,
By Product Type
5.2.1. Synthetic Skin Substitute
5.2.1.1. Definition
5.2.1.2. Market Penetration
5.2.1.3. Market Revenue Expected to Increase by 2026
5.2.1.4. Compound Annual Growth Rate (CAGR)
5.2.2. Biosynthetic Skin Substitute
5.2.2.1. Definition
5.2.2.2. Market Penetration
5.2.2.3. Market Revenue Expected to Increase by 2026
5.2.2.4. Compound Annual Growth Rate (CAGR)
5.2.3. Biological Skin Substitute (Definition, Market Penetration,
Market Revenue Expected to Increase by 2026, Compound Annual Growth Rate (CAGR)
and information on Allograft, Xenograft)
5.2.3.1.
Allograft
5.2.3.1.1. Cellular
5.2.3.1.2. Acellular
5.2.3.1.3. Others
5.2.3.2. Xenograft
5.3. Key Segment for Channeling Investments
5.3.1. By Product Type
6. U.S. Skin Substitutes Market Analysis and Forecasts, 2018 –
2026
6.1. Overview
6.2. U.S. Skin Substitutes Market Revenue (US$ Mn) and Forecasts,
By Application
6.2.1. Burn Injury
6.2.1.1. Definition
6.2.1.2. Market Penetration
6.2.1.3. Market Revenue Expected to Increase by 2026
6.2.1.4. Compound Annual Growth Rate (CAGR)
6.2.2. Diabetic Foot Ulcers
6.2.2.1. Definition
6.2.2.2. Market Penetration
6.2.2.3. Market Revenue Expected to Increase by 2026
6.2.2.4. Compound Annual Growth Rate (CAGR)
6.2.3. Chronic Wounds
6.2.3.1. Definition
6.2.3.2. Market Penetration
6.2.3.3. Market Revenue Expected to Increase by 2026
6.2.3.4. Compound Annual Growth Rate (CAGR)
6.2.4. Others (Vascular Ulcers, Plastic Surgery, Etc.)
6.2.4.1. Definition
6.2.4.2. Market Penetration
6.2.4.3. Market Revenue Expected to Increase by 2026
6.2.4.4. Compound Annual Growth Rate (CAGR)
6.3. Key Segment for Channeling Investments
6.3.1. By Application
7. U.S. Skin Substitutes Market Analysis and Forecasts, 2018 –
2026
7.1. Overview
7.2. U.S. Skin Substitutes Market Revenue (US$ Mn) and Forecasts,
By End User
7.2.1. Hospitals
7.2.1.1. Definition
7.2.1.2. Market Penetration
7.2.1.3. Market Revenue Expected to Increase by 2026
7.2.1.4. Compound Annual Growth Rate (CAGR)
7.2.2. Other Healthcare Facilities
7.2.2.1. Definition
7.2.2.2. Market Penetration
7.2.2.3. Market Revenue Expected to Increase by 2026
7.2.2.4. Compound Annual Growth Rate (CAGR)
7.3. Key Segment for Channeling Investments
7.3.1. By End User
8. Competitive Benchmarking
8.1. Player Positioning Analysis
8.2. U.S. Presence and Growth Strategies
9. Player Profiles
9.1. Acelity L.P. Inc.
9.1.1. Company Details
9.1.2. Company Overview
9.1.3. Product Offerings
9.1.4. Key Developments
9.1.5. Financial Analysis
9.1.6. SWOT Analysis
9.1.7. Business Strategies
9.2. AlloSource Advanced Tissue AMARANTUS BIOSCIENCE HOLDINGS,
INC.,
9.2.1. Company Details
9.2.2. Company Overview
9.2.3. Product Offerings
9.2.4. Key Developments
9.2.5. Financial Analysis
9.2.6. SWOT Analysis
9.2.7. Business Strategies
9.3. Advanced Tissue
9.3.1. Company Details
9.3.2. Company Overview
9.3.3. Product Offerings
9.3.4. Key Developments
9.3.5. Financial Analysis
9.3.6. SWOT Analysis
9.3.7. Business Strategies
9.4. AMARANTUS BIOSCIENCE HOLDINGS, INC.
9.4.1. Company Details
9.4.2. Company Overview
9.4.3. Product Offerings
9.4.4. Key Developments
9.4.5. Financial Analysis
9.4.6. SWOT Analysis
9.4.7. Business Strategies
9.5. BSN Medical
9.5.1. Company Details
9.5.2. Company Overview
9.5.3. Product Offerings
9.5.4. Key Developments
9.5.5. Financial Analysis
9.5.6. SWOT Analysis
9.5.7. Business Strategies
9.6. C. R. Bard, Inc.
9.6.1. Company Details
9.6.2. Company Overview
9.6.3. Product Offerings
9.6.4. Key Developments
9.6.5. Financial Analysis
9.6.6. SWOT Analysis
9.6.7. Business Strategies
9.7. Cook Biotech, Inc.
9.7.1. Company Details
9.7.2. Company Overview
9.7.3. Product Offerings
9.7.4. Key Developments
9.7.5. Financial Analysis
9.7.6. SWOT Analysis
9.7.7. Business Strategies
9.8. Genzyme Corporation
9.8.1. Company Details
9.8.2. Company Overview
9.8.3. Product Offerings
9.8.4. Key Developments
9.8.5. Financial Analysis
9.8.6. SWOT Analysis
9.8.7. Business Strategies
9.9. Integra Life Sciences Corporation
9.9.1. Company Details
9.9.2. Company Overview
9.9.3. Product Offerings
9.9.4. Key Developments
9.9.5. Financial Analysis
9.9.6. SWOT Analysis
9.9.7. Business Strategies
9.10. Medtronic (Covidien)
9.10.1. Company Details
9.10.2. Company Overview
9.10.3. Product Offerings
9.10.4. Key Developments
9.10.5. Financial Analysis
9.10.6. SWOT Analysis
9.10.7. Business Strategies
9.11. Mölnlycke Health Care AB
9.11.1. Company Details
9.11.2. Company Overview
9.11.3. Product Offerings
9.11.4. Key Developments
9.11.5. Financial Analysis
9.11.6. SWOT Analysis
9.11.7. Business Strategies
9.12. Organogenesis Inc.
9.12.1. Company Details
9.12.2. Company Overview
9.12.3. Product Offerings
9.12.4. Key Developments
9.12.5. Financial Analysis
9.12.6. SWOT Analysis
9.12.7. Business Strategies
9.13. PolyNovo North America LLC
9.13.1. Company Details
9.13.2. Company Overview
9.13.3. Product Offerings
9.13.4. Key Developments
9.13.5. Financial Analysis
9.13.6. SWOT Analysis
9.13.7. Business Strategies
9.14. Shire Regenerative Medicine, Inc. (formerly Advanced
BioHealing, Inc.)
9.14.1. Company Details
9.14.2. Company Overview
9.14.3. Product Offerings
9.14.4. Key Developments
9.14.5. Financial Analysis
9.14.6. SWOT Analysis
9.14.7. Business Strategies
9.15. Smith & Nephew plc.
9.15.1. Company Details
9.15.2. Company Overview
9.15.3. Product Offerings
9.15.4. Key Developments
9.15.5. Financial Analysis
9.15.6. SWOT Analysis
9.15.7. Business Strategies
9.16. Soluble Systems, LLC (Alliqua BioMedical, Inc.)
9.16.1. Company Details
9.16.2. Company Overview
9.16.3. Product Offerings
9.16.4. Key Developments
9.16.5. Financial Analysis
9.16.6. SWOT Analysis
9.16.7. Business Strategies
9.17. Stratatech a Mallinckrodt
9.17.1. Company Details
9.17.2. Company Overview
9.17.3. Product Offerings
9.17.4. Key Developments
9.17.5. Financial Analysis
9.17.6. SWOT Analysis
9.17.7. Business Strategies
9.18. Tissue Regenix Group Plc
9.18.1. Company Details
9.18.2. Company Overview
9.18.3. Product Offerings
9.18.4. Key Developments
9.18.5. Financial Analysis
9.18.6. SWOT Analysis
9.18.7. Business Strategies
Note:
This ToC is tentative and can be changed according to the research study
conducted during the course of report completion.
At Absolute Markets Insights, we are engaged in building both global as well as country specific reports. As a result, the approach taken for deriving the estimation and forecast for a specific country is a bit unique and different in comparison to the global research studies. In this case, we not only study the concerned market factors & trends prevailing in a particular country (from secondary research) but we also tend to calculate the actual market size & forecast from the revenue generated from the market participants involved in manufacturing or distributing the any concerned product. These companies can also be service providers. For analyzing any country specifically, we do consider the growth factors prevailing under the states/cities/county for the same. For instance, if we are analyzing an industry specific to United States, we primarily need to study about the states present under the same(where the product/service has the highest growth). Similar analysis will be followed by other countries. Our scope of the report changes with different markets.
Our research study is mainly implement through a mix of both secondary and primary research. Various sources such as industry magazines, trade journals, and government websites and trade associations are reviewed for gathering precise data. Primary interviews are conducted to validate the market size derived from secondary research. Industry experts, major manufacturers and distributors are contacted for further validation purpose on the current market penetration and growth trends.
Prominent participants in our primary research process include:
- Key Opinion Leaders namely the CEOs, CSOs, VPs, purchasing managers, amongst others
- Research and development participants, distributors/suppliers and subject matter experts
Secondary Research includes data extracted from paid data sources:
- Reuters
- Factiva
- Bloomberg
- One Source
- Hoovers
Research Methodology
Key Inclusions

Reach to us
Call us on
+91-74002-42424
Drop us an email at
sales@absolutemarketsinsights.com
Why Absolute Markets Insights?
An effective strategy is the entity that influences a business to stand out of the crowd. An organization with a phenomenal strategy for success dependably has the edge over the rivals in the market. It offers the organizations a head start in planning their strategy. Absolute Market Insights is the new initiation in the industry that will furnish you with the lead your business needs. Absolute Market Insights is the best destination for your business intelligence and analytical solutions; essentially because our qualitative and quantitative sources of information are competent to give one-stop solutions. We inventively combine qualitative and quantitative research in accurate proportions to have the best report, which not only gives the most recent insights but also assists you to grow.