Global Duchenne Muscular Dystrophy (DMD) Market By Diagnosis (Enzyme Tests, Blood test, Genetic Testing, Muscle biopsy, CT Scan, Electromyogram (EMG), Electrocardiogram (ECG or EKG), Others); By Stages (Early Ambulatory, Late Ambulatory, Early Non-Ambulatory, Late Non-Ambulatory); By Clinical Phase (Pre-clinical, Phase 1, Phase 2, Phase 3, Phase 4, Approval); By Age Group (1 to 4 years, 5 to 7 years, 8 to 11 years, 12 to 19 years, Adults); By Treatment (Medication – Corticosteroids, Heart medications, Therapy – Range-of-motion and stretching exercises, Braces, Mobility aids, Breathing assistance, Others, Surgery, Diet and Nutritional supplements, Others); By Region (North America (U.S., Canada, Mexico, Rest Of North America), Europe (France, The UK, Spain, Germany, Italy, Denmark, Finland, Iceland, Sweden, Norway, Belgium, The Netherlands, Luxembourg, Rest of Europe), Asia Pacific (China, Japan, India, New Zealand, Australia, South Korea, Southeast Asia, Indonesia, Thailand, Malaysia, Singapore, Rest of Southeast Asia, Rest of Asia Pacific, Middle East & Africa (Saudi Arabia, UAE, Egypt, Kuwait, South Africa, Rest of Middle East & Africa) Latin America (Brazil, Argentina, Rest of Latin America)) - Global Insights, Growth, Size, Comparative Analysis, Trends and Forecast, 2022 – 2030
Industry Trends
Duchenne muscular dystrophy (DMD) happens to be a genetic disorder that has been characterized through a progressive muscle degeneration and weakness owing to the alterations of a bodily protein known as dystrophin that aids in keeping muscle cells intact. Duchenne muscular dystrophy (DMD) happens to be one of four conditions called dystrophinopathies. Apart from this, the remaining three diseases part of this group are Becker Muscular dystrophy (BMD, which happens to be a mild form of DMD); which is an intermediate clinical presentation among the Duchenne muscular dystrophy and Becker Muscular dystrophy; along with the DMD-linked dilated cardiomyopathy, a heart-disease with very less clinical skeletal, or even voluntary, muscle dystrophy. The symptoms of this diseases are likely to be seen majorly in early childhood, generally in the ages 2 and 3. The DMD mainly affects boys, though the girls can also witness the symptoms of this disease.
This disease was first described by a neurologist originating from the country of France named Guillaume Benjamin Amand Duchenne in the years between 1860 and 1870, though even hundred years after this, very little was known about what causes this disease. Later in the year 1986, the identification of a particular gene on the X chromosome was done by couple of researchers. This gene, when gets affected or mutated turns into or causes DMD. In the next year, the protein linked with this gene was identified and was named as dystrophin. Lower levels of the dystrophin protein in muscle cells makes them fragile and can be damaged easily. One of the major symptoms of DMD is the muscle weakness. This dystrophy starts from the age of 2 or 3, initially affecting the muscles that are important for the body and later gets better of the distal limb muscles. Normally, the external muscles placed in the lower part of the body are affected before the upper ones. The kid affected with the disorder is likely to have difficulty in various activities like walking, running and jumping. On the other hand, other symptoms are inclusive of enlarged calves, along with waddling gait, and lumbar lordosis which is an inward curve of the spine. Ultimately the disorder also gets better of the heart and respiratory muscles affecting them. Rising prevalence of this disorder in the children across various nation is the reason why the global duchenne muscular dystrophy (DMD) market is growing aggressively.
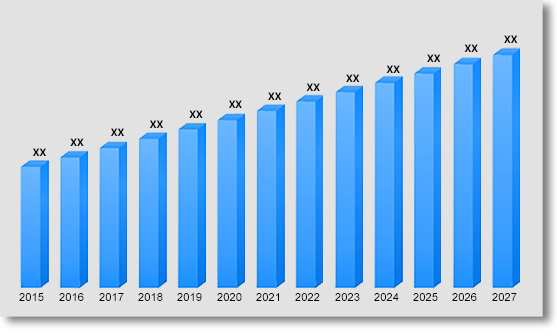
In terms of revenue, global duchenne muscular dystrophy (DMD) market was valued at US$ 1301.44 Mn in 2021 growing at a CAGR of 4.7% over the forecast period (2022 – 2030). The study analyses the market in terms of revenue across all the major regions, which have been bifurcated into countries.
Global Duchenne Muscular Dystrophy (DMD) Market Revenue & Forecast, (US$ Million), 2015 – 2030
Diagnosis Outlook
Based on the diagnosis, the global duchenne muscular dystrophy (DMD) market is segmented into enzyme tests, blood test, genetic testing, muscle biopsy, CT scan, electromyogram (EMG), electrocardiogram (ECG or EKG), and others. Genetic testing segment accounts for majority of revenue for this market. This type of testing involves checking one’s DNA that is the chemical database that brings along with the instructions for one’s functions of the human body. Genetic testing is capable of revealing changes (mutations) in the genes of humans that are likely to cause any type of disorder. It can provide key information for the purpose of diagnosing, treating and preventing any disease, though there are limitations. In case, of a healthy person, a positive result from genetic testing doesn't most of the time means one will develop a disease. Meanwhile, in certain situations, a negative result also indicates one having certain disorder. This is the reason, the Global Duchenne Muscular Dystrophy (DMD) Market makes most of the money from this segment.
Region Outlook
Based on region North America had the highest share in the global duchenne muscular dystrophy (DMD) market in the year 2021. Over the period of last several years, there has been a surge in the number of disorders amongst a major chunk of the population in nations like the United States and Canada. The prevalence of this disorder is likely to be 1 in every 3,500 live births that are male. Age of onset is generally among the age of 3 and 5 years. According to the reports, there are numerous muscular dystrophies and on a large scale are likely to affect nearly 250,000 American individuals. Owing to all this, the region will be minting massive amount of money for the market.
Competitive Landscape
The report provides both, qualitative and quantitative research of global duchenne muscular dystrophy (DMD) market, as well as provides comprehensive insights and development methods adopted by the key contenders. The report also offers extensive research on the key players in this market and details on the competitiveness of these players. Key business strategies such as mergers and acquisitions (M&A), affiliations, collaborations, and contracts adopted by these major market participants are also recognized and analysed in the report. For each company, the report studies their global presence, competitors, service offerings and specification amongst others.
Some of the players operating in the global duchenne muscular dystrophy (DMD) market are:
- Antisense Therapeutics Limited
- Capricor Therapeutics.
- DAIICHI SANKYO COMPANY, LIMITED
- Dyne Therapeutics
- Edgewise Therapeutics
- ENCELL Co, Ltd
- Entrada Therapeutics, Inc
- FibroGen, Inc.
- ITALFARMACO S.p.A
- NIPPON SHINYAKU CO. LTD
- PepGen Inc.
- Pfizer Inc.
- SANTHERA PHARMACEUTICALS
- Sarepta Therapeutics, Inc.
- Solid Biosciences Inc
- Taiho Pharmaceuticals Co. Ltd
- Wave Life Sciences
- Other Industry Participants
Global Duchenne Muscular Dystrophy (DMD) Market:
By Diagnosis
- Enzyme Tests
- Blood test
- Genetic Testing
- Muscle biopsy
- CT Scan
- Electromyogram (EMG)
- Electrocardiogram (ECG or EKG)
- Others
By Stages
- Early Ambulatory
- Late Ambulatory
- Early Non-Ambulatory
- Late Non-Ambulatory
By Clinical Phase
- Pre-clinical
- Phase 1
- Phase 2
- Phase 3
- Phase 4
- Approval
By Age Group
- 1 to 4 years
- 5 to 7 years
- 8 to 11 years
- 12 to 19 years
- Adults
By Treatment
- Medication
- Corticosteroids
- Heart medications
- Therapy
- Range-of-motion and stretching exercises
- Braces
- Mobility aids
- Breathing assistance
- Others
- Surgery
- Diet and Nutritional supplements
- Others
By Region
- North America (U.S., Canada, Mexico, Rest of North America)
- Europe (France, The UK, Spain, Germany, Italy, Nordic Countries (Denmark, Finland, Iceland, Sweden, Norway), Benelux Union (Belgium, The Netherlands, Luxembourg), Rest of Europe
- Asia Pacific (China, Japan, India, New Zealand, Australia, South Korea, Southeast Asia (Indonesia, Thailand, Malaysia, Singapore, Rest of Southeast Asia), Rest of Asia Pacific
- Middle East & Africa (Saudi Arabia, UAE, Egypt, Kuwait, South Africa, Rest of Middle East & Africa)
- Latin America (Brazil, Argentina, Rest of Latin America)
Table of Contents
1.
Market
Scope
1.1. Market Segmentation
1.2. Years Considered
1.2.1. Historic Years: 2015 - 2020
1.2.2. Base Year: 2021
1.2.3. Forecast Years: 2022 - 2030
2.
Key
Target Audiences
3.
Research
Methodology
3.1. Primary Research
3.1.1. Research Questionnaire
3.1.2. Global Percentage Breakdown
3.1.3. Primary Interviews: Key Opinion Leaders
(KOLs)
3.2. Secondary Research
3.2.1. Paid Databases
3.2.2. Secondary Sources
3.3. Market Size Estimates
3.3.1. Top-Down Approach
3.3.2. Bottom-Up Approach
3.4. Data Triangulation Methodology
3.5. Research Assumptions
4.
Recommendations
and Insights from AMI’s Perspective**
5.
Holistic
Overview of Duchenne Muscular Dystrophy (DMD) Market
6.
Market
Synopsis: Duchenne Muscular Dystrophy
(DMD) Market
7.
Duchenne
Muscular Dystrophy (DMD) Market Analysis: Qualitative Perspective
7.1. Introduction
7.1.1. Product Definition
7.1.2. Industry Development
7.2. Market Dynamics
7.2.1. Drivers
7.2.2. Restraints
7.2.3. Opportunities
7.2.4. Challenges
7.3. Trends in Duchenne Muscular Dystrophy (DMD)
Market
7.4. Market Determinants Radar Chart
7.5. Macro-Economic and Micro-Economic Indicators:
Duchenne Muscular Dystrophy (DMD) Market
7.6. Impact of Covid-19 on Duchenne Muscular
Dystrophy (DMD) Market
8.
Global Duchenne
Muscular Dystrophy (DMD) Market Analysis and Forecasts, 2022 - 2030
8.1. Overview
8.1.1. Global Duchenne Muscular Dystrophy (DMD)
Market Revenue (US$ Mn)
8.2. Global Duchenne Muscular Dystrophy (DMD)
Market Revenue (US$ Mn) and Forecasts, By Diagnosis
8.2.1. Enzyme Tests
8.2.1.1.
Definition
8.2.1.2.
Market
Estimation and Penetration, 2015 - 2021
8.2.1.3.
Market
Forecast, 2022 - 2030
8.2.1.4.
Compound
Annual Growth Rate (CAGR)
8.2.1.5.
Regional
Bifurcation
8.2.1.5.1.
North
America
8.2.1.5.1.1. Market Estimation, 2015 - 2021
8.2.1.5.1.2. Market Forecast, 2022 - 2030
8.2.1.5.2.
Europe
8.2.1.5.2.1. Market Estimation, 2015 - 2021
8.2.1.5.2.2. Market Forecast, 2022 - 2030
8.2.1.5.3.
Asia
Pacific
8.2.1.5.3.1. Market Estimation, 2015 - 2021
8.2.1.5.3.2. Market Forecast, 2022 - 2030
8.2.1.5.4.
Middle
East and Africa
8.2.1.5.4.1. Market Estimation, 2015 - 2021
8.2.1.5.4.2. Market Forecast, 2022 - 2030
8.2.1.5.5.
Latin
America
8.2.1.5.5.1. Market Estimation, 2015 - 2021
8.2.1.5.5.2. Market Forecast, 2022 - 2030
8.2.2. Blood Tests
8.2.2.1.
Definition
8.2.2.2.
Market
Estimation and Penetration, 2015 - 2021
8.2.2.3.
Market
Forecast, 2022 - 2030
8.2.2.4.
Compound
Annual Growth Rate (CAGR)
8.2.2.5.
Regional
Bifurcation
8.2.2.5.1.
North
America
8.2.2.5.1.1. Market Estimation, 2015 - 2021
8.2.2.5.1.2. Market Forecast, 2022 - 2030
8.2.2.5.2.
Europe
8.2.2.5.2.1. Market Estimation, 2015 - 2021
8.2.2.5.2.2. Market Forecast, 2022 - 2030
8.2.2.5.3.
Asia
Pacific
8.2.2.5.3.1. Market Estimation, 2015 - 2021
8.2.2.5.3.2. Market Forecast, 2022 - 2030
8.2.2.5.4.
Middle
East and Africa
8.2.2.5.4.1. Market Estimation, 2015 - 2021
8.2.2.5.4.2. Market Forecast, 2022 - 2030
8.2.2.5.5.
Latin
America
8.2.2.5.5.1. Market Estimation, 2015 - 2021
8.2.2.5.5.2. Market Forecast, 2022 - 2030
8.2.3. Genetic Testing
8.2.3.1.
Definition
8.2.3.2.
Market
Estimation and Penetration, 2015 - 2021
8.2.3.3.
Market
Forecast, 2022 - 2030
8.2.3.4.
Compound
Annual Growth Rate (CAGR)
8.2.3.5.
Regional
Bifurcation
8.2.3.5.1.
North
America
8.2.3.5.1.1. Market Estimation, 2015 - 2021
8.2.3.5.1.2. Market Forecast, 2022 - 2030
8.2.3.5.2.
Europe
8.2.3.5.2.1. Market Estimation, 2015 - 2021
8.2.3.5.2.2. Market Forecast, 2022 - 2030
8.2.3.5.3.
Asia
Pacific
8.2.3.5.3.1. Market Estimation, 2015 - 2021
8.2.3.5.3.2. Market Forecast, 2022 - 2030
8.2.3.5.4.
Middle
East and Africa
8.2.3.5.4.1. Market Estimation, 2015 - 2021
8.2.3.5.4.2. Market Forecast, 2022 - 2030
8.2.3.5.5.
Latin
America
8.2.3.5.5.1. Market Estimation, 2015 - 2021
8.2.3.5.5.2. Market Forecast, 2022 - 2030
8.2.4. Muscle
biopsy
8.2.4.1.
Definition
8.2.4.2.
Market
Estimation and Penetration, 2015 - 2021
8.2.4.3.
Market
Forecast, 2022 - 2030
8.2.4.4.
Compound
Annual Growth Rate (CAGR)
8.2.4.5.
Regional
Bifurcation
8.2.4.5.1.
North
America
8.2.4.5.1.1. Market Estimation, 2015 - 2021
8.2.4.5.1.2. Market Forecast, 2022 - 2030
8.2.4.5.2.
Europe
8.2.4.5.2.1. Market Estimation, 2015 - 2021
8.2.4.5.2.2. Market Forecast, 2022 - 2030
8.2.4.5.3.
Asia
Pacific
8.2.4.5.3.1. Market Estimation, 2015 - 2021
8.2.4.5.3.2. Market Forecast, 2022 - 2030
8.2.4.5.4.
Middle
East and Africa
8.2.4.5.4.1. Market Estimation, 2015 - 2021
8.2.4.5.4.2. Market Forecast, 2022 - 2030
8.2.4.5.5.
Latin
America
8.2.4.5.5.1. Market Estimation, 2015 - 2021
8.2.4.5.5.2. Market Forecast, 2022 - 2030
8.2.5. CT Scan
8.2.5.1.
Definition
8.2.5.2.
Market
Estimation and Penetration, 2015 - 2021
8.2.5.3.
Market
Forecast, 2022 - 2030
8.2.5.4.
Compound
Annual Growth Rate (CAGR)
8.2.5.5.
Regional
Bifurcation
8.2.5.5.1.
North
America
8.2.5.5.1.1. Market Estimation, 2015 - 2021
8.2.5.5.1.2. Market Forecast, 2022 - 2030
8.2.5.5.2.
Europe
8.2.5.5.2.1. Market Estimation, 2015 - 2021
8.2.5.5.2.2. Market Forecast, 2022 - 2030
8.2.5.5.3.
Asia
Pacific
8.2.5.5.3.1. Market Estimation, 2015 - 2021
8.2.5.5.3.2. Market Forecast, 2022 - 2030
8.2.5.5.4.
Middle
East and Africa
8.2.5.5.4.1. Market Estimation, 2015 - 2021
8.2.5.5.4.2. Market Forecast, 2022 - 2030
8.2.5.5.5.
Latin
America
8.2.5.5.5.1. Market Estimation, 2015 - 2021
8.2.5.5.5.2. Market Forecast, 2022 - 2030
8.2.6. Electromyogram (EMG)
8.2.6.1.
Definition
8.2.6.2.
Market
Estimation and Penetration, 2015 - 2021
8.2.6.3.
Market
Forecast, 2022 - 2030
8.2.6.4.
Compound
Annual Growth Rate (CAGR)
8.2.6.5.
Regional
Bifurcation
8.2.6.5.1.
North
America
8.2.6.5.1.1. Market Estimation, 2015 - 2021
8.2.6.5.1.2. Market Forecast, 2022 - 2030
8.2.6.5.2.
Europe
8.2.6.5.2.1. Market Estimation, 2015 - 2021
8.2.6.5.2.2. Market Forecast, 2022 - 2030
8.2.6.5.3.
Asia
Pacific
8.2.6.5.3.1. Market Estimation, 2015 - 2021
8.2.6.5.3.2. Market Forecast, 2022 - 2030
8.2.6.5.4.
Middle
East and Africa
8.2.6.5.4.1. Market Estimation, 2015 - 2021
8.2.6.5.4.2. Market Forecast, 2022 - 2030
8.2.6.5.5.
Latin
America
8.2.6.5.5.1. Market Estimation, 2015 - 2021
8.2.6.5.5.2. Market Forecast, 2022 - 2030
8.2.7. Electrocardiogram (ECG or EKG)
8.2.7.1.
Definition
8.2.7.2.
Market
Estimation and Penetration, 2015 - 2021
8.2.7.3.
Market
Forecast, 2022 - 2030
8.2.7.4.
Compound
Annual Growth Rate (CAGR)
8.2.7.5.
Regional
Bifurcation
8.2.7.5.1.
North
America
8.2.7.5.1.1. Market Estimation, 2015 - 2021
8.2.7.5.1.2. Market Forecast, 2022 - 2030
8.2.7.5.2.
Europe
8.2.7.5.2.1. Market Estimation, 2015 - 2021
8.2.7.5.2.2. Market Forecast, 2022 - 2030
8.2.7.5.3.
Asia
Pacific
8.2.7.5.3.1. Market Estimation, 2015 - 2021
8.2.7.5.3.2. Market Forecast, 2022 - 2030
8.2.7.5.4.
Middle
East and Africa
8.2.7.5.4.1. Market Estimation, 2015 - 2021
8.2.7.5.4.2. Market Forecast, 2022 - 2030
8.2.7.5.5.
Latin
America
8.2.7.5.5.1. Market Estimation, 2015 - 2021
8.2.7.5.5.2. Market Forecast, 2022 - 2030
8.2.8. Others
8.2.8.1.
Definition
8.2.8.2.
Market
Estimation and Penetration, 2015 - 2021
8.2.8.3.
Market
Forecast, 2022 - 2030
8.2.8.4.
Compound
Annual Growth Rate (CAGR)
8.2.8.5.
Regional
Bifurcation
8.2.8.5.1.
North
America
8.2.8.5.1.1. Market Estimation, 2015 - 2021
8.2.8.5.1.2. Market Forecast, 2022 - 2030
8.2.8.5.2.
Europe
8.2.8.5.2.1. Market Estimation, 2015 - 2021
8.2.8.5.2.2. Market Forecast, 2022 - 2030
8.2.8.5.3.
Asia
Pacific
8.2.8.5.3.1. Market Estimation, 2015 - 2021
8.2.8.5.3.2. Market Forecast, 2022 - 2030
8.2.8.5.4.
Middle
East and Africa
8.2.8.5.4.1. Market Estimation, 2015 - 2021
8.2.8.5.4.2. Market Forecast, 2022 - 2030
8.2.8.5.5.
Latin
America
8.2.8.5.5.1. Market Estimation, 2015 - 2021
8.2.8.5.5.2. Market Forecast, 2022 - 2030
8.3. Key Segment for Channeling Investments
8.3.1. By Diagnosis
9.
Global Duchenne
Muscular Dystrophy (DMD) Market Analysis and Forecasts, 2022 - 2030
9.1. Overview
9.2. Global Duchenne Muscular Dystrophy (DMD)
Market Revenue (US$ Mn) and Forecasts, By Stages
9.2.1. Early Ambulatory
9.2.1.1.
Definition
9.2.1.2.
Market
Estimation and Penetration, 2015 - 2021
9.2.1.3.
Market
Forecast, 2022 - 2030
9.2.1.4.
Compound
Annual Growth Rate (CAGR)
9.2.1.5.
Regional
Bifurcation
9.2.1.5.1.
North
America
9.2.1.5.1.1. Market Estimation, 2015 - 2021
9.2.1.5.1.2. Market Forecast, 2022 - 2030
9.2.1.5.2.
Europe
9.2.1.5.2.1. Market Estimation, 2015 - 2021
9.2.1.5.2.2. Market Forecast, 2022 - 2030
9.2.1.5.3.
Asia
Pacific
9.2.1.5.3.1. Market Estimation, 2015 - 2021
9.2.1.5.3.2. Market Forecast, 2022 - 2030
9.2.1.5.4.
Middle
East and Africa
9.2.1.5.4.1. Market Estimation, 2015 - 2021
9.2.1.5.4.2. Market Forecast, 2022 - 2030
9.2.1.5.5.
Latin
America
9.2.1.5.5.1. Market Estimation, 2015 - 2021
9.2.1.5.5.2. Market Forecast, 2022 - 2030
9.2.2. Late Ambulatory
9.2.2.1.
Definition
9.2.2.2.
Market
Estimation and Penetration, 2015 - 2021
9.2.2.3.
Market
Forecast, 2022 - 2030
9.2.2.4.
Compound
Annual Growth Rate (CAGR)
9.2.2.5.
Regional
Bifurcation
9.2.2.5.1.
North
America
9.2.2.5.1.1. Market Estimation, 2015 - 2021
9.2.2.5.1.2. Market Forecast, 2022 - 2030
9.2.2.5.2.
Europe
9.2.2.5.2.1. Market Estimation, 2015 - 2021
9.2.2.5.2.2. Market Forecast, 2022 - 2030
9.2.2.5.3.
Asia
Pacific
9.2.2.5.3.1. Market Estimation, 2015 - 2021
9.2.2.5.3.2. Market Forecast, 2022 - 2030
9.2.2.5.4.
Middle
East and Africa
9.2.2.5.4.1. Market Estimation, 2015 - 2021
9.2.2.5.4.2. Market Forecast, 2022 - 2030
9.2.2.5.5.
Latin
America
9.2.2.5.5.1. Market Estimation, 2015 - 2021
9.2.2.5.5.2. Market Forecast, 2022 - 2030
9.2.3. Early Non-Ambulatory
9.2.3.1.
Definition
9.2.3.2.
Market
Estimation and Penetration, 2015 - 2021
9.2.3.3.
Market
Forecast, 2022 - 2030
9.2.3.4.
Compound
Annual Growth Rate (CAGR)
9.2.3.5.
Regional
Bifurcation
9.2.3.5.1.
North
America
9.2.3.5.1.1. Market Estimation, 2015 - 2021
9.2.3.5.1.2. Market Forecast, 2022 - 2030
9.2.3.5.2.
Europe
9.2.3.5.2.1. Market Estimation, 2015 - 2021
9.2.3.5.2.2. Market Forecast, 2022 - 2030
9.2.3.5.3.
Asia
Pacific
9.2.3.5.3.1. Market Estimation, 2015 - 2021
9.2.3.5.3.2. Market Forecast, 2022 - 2030
9.2.3.5.4.
Middle
East and Africa
9.2.3.5.4.1. Market Estimation, 2015 - 2021
9.2.3.5.4.2. Market Forecast, 2022 - 2030
9.2.3.5.5.
Latin
America
9.2.3.5.5.1. Market Estimation, 2015 - 2021
9.2.3.5.5.2. Market Forecast, 2022 - 2030
9.2.4. Late Non-Ambulatory
9.2.4.1.
Definition
9.2.4.2.
Market
Estimation and Penetration, 2015 - 2021
9.2.4.3.
Market
Forecast, 2022 - 2030
9.2.4.4.
Compound
Annual Growth Rate (CAGR)
9.2.4.5.
Regional
Bifurcation
9.2.4.5.1.
North
America
9.2.4.5.1.1. Market Estimation, 2015 - 2021
9.2.4.5.1.2. Market Forecast, 2022 - 2030
9.2.4.5.2.
Europe
9.2.4.5.2.1. Market Estimation, 2015 - 2021
9.2.4.5.2.2. Market Forecast, 2022 - 2030
9.2.4.5.3.
Asia
Pacific
9.2.4.5.3.1. Market Estimation, 2015 - 2021
9.2.4.5.3.2. Market Forecast, 2022 - 2030
9.2.4.5.4.
Middle
East and Africa
9.2.4.5.4.1. Market Estimation, 2015 - 2021
9.2.4.5.4.2. Market Forecast, 2022 - 2030
9.2.4.5.5.
Latin
America
9.2.4.5.5.1. Market Estimation, 2015 - 2021
9.2.4.5.5.2. Market Forecast, 2022 - 2030
9.3. Key Segment for Channeling Investments
9.3.1. By Stages
10. Global Duchenne Muscular Dystrophy (DMD)
Market Analysis and Forecasts, 2022 - 2030
10.1. Overview
10.2. Global Duchenne Muscular Dystrophy (DMD)
Market Revenue (US$ Mn) and Forecasts, By Clinical Phase
10.2.1. Pre-clinical
10.2.1.1.
Definition
10.2.1.2.
Market
Estimation and Penetration, 2015 - 2021
10.2.1.3.
Market
Forecast, 2022 - 2030
10.2.1.4.
Compound
Annual Growth Rate (CAGR)
10.2.1.5.
Regional
Bifurcation
10.2.1.5.1.
North
America
10.2.1.5.1.1. Market Estimation, 2015 - 2021
10.2.1.5.1.2. Market Forecast, 2022 - 2030
10.2.1.5.2.
Europe
10.2.1.5.2.1. Market Estimation, 2015 - 2021
10.2.1.5.2.2. Market Forecast, 2022 - 2030
10.2.1.5.3.
Asia
Pacific
10.2.1.5.3.1. Market Estimation, 2015 - 2021
10.2.1.5.3.2. Market Forecast, 2022 - 2030
10.2.1.5.4.
Middle
East and Africa
10.2.1.5.4.1. Market Estimation, 2015 - 2021
10.2.1.5.4.2. Market Forecast, 2022 - 2030
10.2.1.5.5.
Latin
America
10.2.1.5.5.1. Market Estimation, 2015 - 2021
10.2.1.5.5.2. Market Forecast, 2022 - 2030
10.2.2. Phase 1
10.2.2.1.
Definition
10.2.2.2.
Market
Estimation and Penetration, 2015 - 2021
10.2.2.3.
Market
Forecast, 2022 - 2030
10.2.2.4.
Compound
Annual Growth Rate (CAGR)
10.2.2.5.
Regional
Bifurcation
10.2.2.5.1.
North
America
10.2.2.5.1.1. Market Estimation, 2015 - 2021
10.2.2.5.1.2. Market Forecast, 2022 - 2030
10.2.2.5.2.
Europe
10.2.2.5.2.1. Market Estimation, 2015 - 2021
10.2.2.5.2.2. Market Forecast, 2022 - 2030
10.2.2.5.3.
Asia
Pacific
10.2.2.5.3.1. Market Estimation, 2015 - 2021
10.2.2.5.3.2. Market Forecast, 2022 - 2030
10.2.2.5.4.
Middle
East and Africa
10.2.2.5.4.1. Market Estimation, 2015 - 2021
10.2.2.5.4.2. Market Forecast, 2022 - 2030
10.2.2.5.5.
Latin
America
10.2.2.5.5.1. Market Estimation, 2015 - 2021
10.2.2.5.5.2. Market Forecast, 2022 - 2030
10.2.3. Phase 2
10.2.3.1.
Definition
10.2.3.2.
Market
Estimation and Penetration, 2015 - 2021
10.2.3.3.
Market
Forecast, 2022 - 2030
10.2.3.4.
Compound
Annual Growth Rate (CAGR)
10.2.3.5.
Regional
Bifurcation
10.2.3.5.1.
North
America
10.2.3.5.1.1. Market Estimation, 2015 - 2021
10.2.3.5.1.2. Market Forecast, 2022 - 2030
10.2.3.5.2.
Europe
10.2.3.5.2.1. Market Estimation, 2015 - 2021
10.2.3.5.2.2. Market Forecast, 2022 - 2030
10.2.3.5.3.
Asia
Pacific
10.2.3.5.3.1. Market Estimation, 2015 - 2021
10.2.3.5.3.2. Market Forecast, 2022 - 2030
10.2.3.5.4.
Middle
East and Africa
10.2.3.5.4.1. Market Estimation, 2015 - 2021
10.2.3.5.4.2. Market Forecast, 2022 - 2030
10.2.3.5.5.
Latin
America
10.2.3.5.5.1. Market Estimation, 2015 - 2021
10.2.3.5.5.2. Market Forecast, 2022 - 2030
10.2.4. Phase 3
10.2.4.1.
Definition
10.2.4.2.
Market
Estimation and Penetration, 2015 - 2021
10.2.4.3.
Market
Forecast, 2022 - 2030
10.2.4.4.
Compound
Annual Growth Rate (CAGR)
10.2.4.5.
Regional
Bifurcation
10.2.4.5.1.
North
America
10.2.4.5.1.1. Market Estimation, 2015 - 2021
10.2.4.5.1.2. Market Forecast, 2022 - 2030
10.2.4.5.2.
Europe
10.2.4.5.2.1. Market Estimation, 2015 - 2021
10.2.4.5.2.2. Market Forecast, 2022 - 2030
10.2.4.5.3.
Asia
Pacific
10.2.4.5.3.1. Market Estimation, 2015 - 2021
10.2.4.5.3.2. Market Forecast, 2022 - 2030
10.2.4.5.4.
Middle
East and Africa
10.2.4.5.4.1. Market Estimation, 2015 - 2021
10.2.4.5.4.2. Market Forecast, 2022 - 2030
10.2.4.5.5.
Latin
America
10.2.4.5.5.1. Market Estimation, 2015 - 2021
10.2.4.5.5.2. Market Forecast, 2022 - 2030
10.2.5. Phase 4
10.2.5.1.
Definition
10.2.5.2.
Market
Estimation and Penetration, 2015 - 2021
10.2.5.3.
Market
Forecast, 2022 - 2030
10.2.5.4.
Compound
Annual Growth Rate (CAGR)
10.2.5.5.
Regional
Bifurcation
10.2.5.5.1.
North
America
10.2.5.5.1.1. Market Estimation, 2015 - 2021
10.2.5.5.1.2. Market Forecast, 2022 - 2030
10.2.5.5.2.
Europe
10.2.5.5.2.1. Market Estimation, 2015 - 2021
10.2.5.5.2.2. Market Forecast, 2022 - 2030
10.2.5.5.3.
Asia
Pacific
10.2.5.5.3.1. Market Estimation, 2015 - 2021
10.2.5.5.3.2. Market Forecast, 2022 - 2030
10.2.5.5.4.
Middle
East and Africa
10.2.5.5.4.1. Market Estimation, 2015 - 2021
10.2.5.5.4.2. Market Forecast, 2022 - 2030
10.2.5.5.5.
Latin
America
10.2.5.5.5.1. Market Estimation, 2015 - 2021
10.2.5.5.5.2. Market Forecast, 2022 - 2030
10.2.6. Approval
10.2.6.1.
Definition
10.2.6.2.
Market
Estimation and Penetration, 2015 - 2021
10.2.6.3.
Market
Forecast, 2022 - 2030
10.2.6.4.
Compound
Annual Growth Rate (CAGR)
10.2.6.5.
Regional
Bifurcation
10.2.6.5.1.
North
America
10.2.6.5.1.1. Market Estimation, 2015 - 2021
10.2.6.5.1.2. Market Forecast, 2022 - 2030
10.2.6.5.2.
Europe
10.2.6.5.2.1. Market Estimation, 2015 - 2021
10.2.6.5.2.2. Market Forecast, 2022 - 2030
10.2.6.5.3.
Asia
Pacific
10.2.6.5.3.1. Market Estimation, 2015 - 2021
10.2.6.5.3.2. Market Forecast, 2022 - 2030
10.2.6.5.4.
Middle
East and Africa
10.2.6.5.4.1. Market Estimation, 2015 - 2021
10.2.6.5.4.2. Market Forecast, 2022 - 2030
10.2.6.5.5.
Latin
America
10.2.6.5.5.1. Market Estimation, 2015 - 2021
10.2.6.5.5.2. Market Forecast, 2022 - 2030
10.3. Key Segment for Channeling Investments
10.3.1. By Clinical Phase
11. Global Duchenne Muscular Dystrophy (DMD)
Market Analysis and Forecasts, 2022 - 2030
11.1. Overview
11.2. Global Duchenne Muscular Dystrophy (DMD)
Market Revenue (US$ Mn) and Forecasts, By Age Group
11.2.1. 1 to 4 years
11.2.1.1.
Definition
11.2.1.2.
Market
Estimation and Penetration, 2015 - 2021
11.2.1.3.
Market
Forecast, 2022 - 2030
11.2.1.4.
Compound
Annual Growth Rate (CAGR)
11.2.1.5.
Regional
Bifurcation
11.2.1.5.1.
North
America
11.2.1.5.1.1. Market Estimation, 2015 - 2021
11.2.1.5.1.2. Market Forecast, 2022 - 2030
11.2.1.5.2.
Europe
11.2.1.5.2.1. Market Estimation, 2015 - 2021
11.2.1.5.2.2. Market Forecast, 2022 - 2030
11.2.1.5.3.
Asia
Pacific
11.2.1.5.3.1. Market Estimation, 2015 - 2021
11.2.1.5.3.2. Market Forecast, 2022 - 2030
11.2.1.5.4.
Middle
East and Africa
11.2.1.5.4.1. Market Estimation, 2015 - 2021
11.2.1.5.4.2. Market Forecast, 2022 - 2030
11.2.1.5.5.
Latin
America
11.2.1.5.5.1. Market Estimation, 2015 - 2021
11.2.1.5.5.2. Market Forecast, 2022 - 2030
11.2.2. 5 to 7 years
11.2.2.1.
Definition
11.2.2.2.
Market
Estimation and Penetration, 2015 - 2021
11.2.2.3.
Market
Forecast, 2022 - 2030
11.2.2.4.
Compound
Annual Growth Rate (CAGR)
11.2.2.5.
Regional
Bifurcation
11.2.2.5.1.
North
America
11.2.2.5.1.1. Market Estimation, 2015 - 2021
11.2.2.5.1.2. Market Forecast, 2022 - 2030
11.2.2.5.2.
Europe
11.2.2.5.2.1. Market Estimation, 2015 - 2021
11.2.2.5.2.2. Market Forecast, 2022 - 2030
11.2.2.5.3.
Asia Pacific
11.2.2.5.3.1. Market Estimation, 2015 - 2021
11.2.2.5.3.2. Market Forecast, 2022 - 2030
11.2.2.5.4.
Middle
East and Africa
11.2.2.5.4.1. Market Estimation, 2015 - 2021
11.2.2.5.4.2. Market Forecast, 2022 - 2030
11.2.2.5.5.
Latin
America
11.2.2.5.5.1. Market Estimation, 2015 - 2021
11.2.2.5.5.2. Market Forecast, 2022 - 2030
11.2.3. 8 to 11 years
11.2.3.1.
Definition
11.2.3.2.
Market
Estimation and Penetration, 2015 - 2021
11.2.3.3.
Market
Forecast, 2022 - 2030
11.2.3.4.
Compound
Annual Growth Rate (CAGR)
11.2.3.5.
Regional
Bifurcation
11.2.3.5.1.
North
America
11.2.3.5.1.1. Market Estimation, 2015 - 2021
11.2.3.5.1.2. Market Forecast, 2022 - 2030
11.2.3.5.2.
Europe
11.2.3.5.2.1. Market Estimation, 2015 - 2021
11.2.3.5.2.2. Market Forecast, 2022 - 2030
11.2.3.5.3.
Asia
Pacific
11.2.3.5.3.1. Market Estimation, 2015 - 2021
11.2.3.5.3.2. Market Forecast, 2022 - 2030
11.2.3.5.4.
Middle
East and Africa
11.2.3.5.4.1. Market Estimation, 2015 - 2021
11.2.3.5.4.2. Market Forecast, 2022 - 2030
11.2.3.5.5.
Latin
America
11.2.3.5.5.1. Market Estimation, 2015 - 2021
11.2.3.5.5.2. Market Forecast, 2022 - 2030
11.2.4. 12 to 19 years
11.2.4.1.
Definition
11.2.4.2.
Market
Estimation and Penetration, 2015 - 2021
11.2.4.3.
Market
Forecast, 2022 - 2030
11.2.4.4.
Compound
Annual Growth Rate (CAGR)
11.2.4.5.
Regional
Bifurcation
11.2.4.5.1.
North
America
11.2.4.5.1.1. Market Estimation, 2015 - 2021
11.2.4.5.1.2. Market Forecast, 2022 - 2030
11.2.4.5.2.
Europe
11.2.4.5.2.1. Market Estimation, 2015 - 2021
11.2.4.5.2.2. Market Forecast, 2022 - 2030
11.2.4.5.3.
Asia
Pacific
11.2.4.5.3.1. Market Estimation, 2015 - 2021
11.2.4.5.3.2. Market Forecast, 2022 - 2030
11.2.4.5.4.
Middle
East and Africa
11.2.4.5.4.1. Market Estimation, 2015 - 2021
11.2.4.5.4.2. Market Forecast, 2022 - 2030
11.2.4.5.5.
Latin
America
11.2.4.5.5.1. Market Estimation, 2015 - 2021
11.2.4.5.5.2. Market Forecast, 2022 - 2030
11.2.5. Adults
11.2.5.1.
Definition
11.2.5.2.
Market
Estimation and Penetration, 2015 - 2021
11.2.5.3.
Market
Forecast, 2022 - 2030
11.2.5.4.
Compound
Annual Growth Rate (CAGR)
11.2.5.5.
Regional
Bifurcation
11.2.5.5.1.
North
America
11.2.5.5.1.1. Market Estimation, 2015 - 2021
11.2.5.5.1.2. Market Forecast, 2022 - 2030
11.2.5.5.2.
Europe
11.2.5.5.2.1. Market Estimation, 2015 - 2021
11.2.5.5.2.2. Market Forecast, 2022 - 2030
11.2.5.5.3.
Asia
Pacific
11.2.5.5.3.1. Market Estimation, 2015 - 2021
11.2.5.5.3.2. Market Forecast, 2022 - 2030
11.2.5.5.4.
Middle
East and Africa
11.2.5.5.4.1. Market Estimation, 2015 - 2021
11.2.5.5.4.2. Market Forecast, 2022 - 2030
11.2.5.5.5.
Latin
America
11.2.5.5.5.1. Market Estimation, 2015 - 2021
11.2.5.5.5.2. Market Forecast, 2022 - 2030
11.3. Key Segment for Channeling Investments
11.3.1. By Age Group
12. Global Duchenne Muscular Dystrophy (DMD)
Market Analysis and Forecasts, 2022 - 2030
12.1. Overview
12.2. Global Duchenne Muscular Dystrophy (DMD)
Market Revenue (US$ Mn) and Forecasts, By Treatment
12.2.1. Medications (Definition, Market Estimation
and Penetration, 2015 - 2021, Market Estimation (2015 - 2021), Market Forecast
(2022 - 2030), Compound Annual Growth Rate (CAGR), Regional Bifurcation (North
America, Europe, Asia Pacific, Middle East and Africa, Latin America) and
Information on Corticosteroids, Heart medications)
12.2.1.1.
Corticosteroids
12.2.1.2.
Heart
medications
12.2.2. Therapy
(Definition, Market Estimation and Penetration, 2015 - 2021, Market
Estimation (2015 - 2021), Market Forecast (2022 - 2030), Compound Annual Growth
Rate (CAGR), Regional Bifurcation (North America, Europe, Asia Pacific, Middle
East and Africa, Latin America) and Information on Range-of-motion and
stretching exercises, Braces, Mobility aids, Breathing assistance, Others)
12.2.2.1.
Range-of-motion
and stretching exercises
12.2.2.2.
Braces
12.2.2.3.
Mobility
aids
12.2.2.4.
Breathing
assistance
12.2.2.5.
Others
12.2.3. Surgery
12.2.3.1.
Definition
12.2.3.2.
Market
Estimation and Penetration, 2015 - 2021
12.2.3.3.
Market
Forecast, 2022 - 2030
12.2.3.4.
Compound
Annual Growth Rate (CAGR)
12.2.3.5.
Regional
Bifurcation
12.2.3.5.1.
North
America
12.2.3.5.1.1. Market Estimation, 2015 - 2021
12.2.3.5.1.2. Market Forecast, 2022 - 2030
12.2.3.5.2.
Europe
12.2.3.5.2.1. Market Estimation, 2015 - 2021
12.2.3.5.2.2. Market Forecast, 2022 - 2030
12.2.3.5.3.
Asia
Pacific
12.2.3.5.3.1. Market Estimation, 2015 - 2021
12.2.3.5.3.2. Market Forecast, 2022 - 2030
12.2.3.5.4.
Middle
East and Africa
12.2.3.5.4.1. Market Estimation, 2015 - 2021
12.2.3.5.4.2. Market Forecast, 2022 - 2030
12.2.3.5.5.
Latin
America
12.2.3.5.5.1. Market Estimation, 2015 - 2021
12.2.3.5.5.2. Market Forecast, 2022 - 2030
12.2.4. Diet and Nutritional supplements
12.2.4.1.
Definition
12.2.4.2.
Market
Estimation and Penetration, 2015 - 2021
12.2.4.3.
Market
Forecast, 2022 - 2030
12.2.4.4.
Compound
Annual Growth Rate (CAGR)
12.2.4.5.
Regional
Bifurcation
12.2.4.5.1.
North
America
12.2.4.5.1.1. Market Estimation, 2015 - 2021
12.2.4.5.1.2. Market Forecast, 2022 - 2030
12.2.4.5.2.
Europe
12.2.4.5.2.1. Market Estimation, 2015 - 2021
12.2.4.5.2.2. Market Forecast, 2022 - 2030
12.2.4.5.3.
Asia
Pacific
12.2.4.5.3.1. Market Estimation, 2015 - 2021
12.2.4.5.3.2. Market Forecast, 2022 - 2030
12.2.4.5.4.
Middle
East and Africa
12.2.4.5.4.1. Market Estimation, 2015 - 2021
12.2.4.5.4.2. Market Forecast, 2022 - 2030
12.2.4.5.5.
Latin
America
12.2.4.5.5.1. Market Estimation, 2015 - 2021
12.2.4.5.5.2. Market Forecast, 2022 - 2030
12.2.5. Others
12.2.5.1.
Definition
12.2.5.2.
Market
Estimation and Penetration, 2015 - 2021
12.2.5.3.
Market
Forecast, 2022 - 2030
12.2.5.4.
Compound
Annual Growth Rate (CAGR)
12.2.5.5.
Regional
Bifurcation
12.2.5.5.1.
North
America
12.2.5.5.1.1. Market Estimation, 2015 - 2021
12.2.5.5.1.2. Market Forecast, 2022 - 2030
12.2.5.5.2.
Europe
12.2.5.5.2.1. Market Estimation, 2015 - 2021
12.2.5.5.2.2. Market Forecast, 2022 - 2030
12.2.5.5.3.
Asia
Pacific
12.2.5.5.3.1. Market Estimation, 2015 - 2021
12.2.5.5.3.2. Market Forecast, 2022 - 2030
12.2.5.5.4.
Middle
East and Africa
12.2.5.5.4.1. Market Estimation, 2015 - 2021
12.2.5.5.4.2. Market Forecast, 2022 - 2030
12.2.5.5.5.
Latin
America
12.2.5.5.5.1. Market Estimation, 2015 - 2021
12.2.5.5.5.2. Market Forecast, 2022 - 2030
12.3. Key Segment for Channeling Investments
12.3.1. By Treatment
13. North America Duchenne Muscular Dystrophy
(DMD) Market Analysis and Forecasts, 2022 - 2030
13.1. Overview
13.1.1. North America Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn)
13.2. North America Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn) and Forecasts, By Diagnosis
13.2.1. Enzyme Tests
13.2.2. Blood Tests
13.2.3. Genetic Testing
13.2.4. Muscle
biopsy
13.2.5. CT Scan
13.2.6. Electromyogram (EMG)
13.2.7. Electrocardiogram (ECG or EKG)
13.2.8. Others
13.3. North America Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn) and Forecasts, By Stages
13.3.1. Early Ambulatory
13.3.2. Late Ambulatory
13.3.3. Early Non-Ambulatory
13.3.4. Late Non-Ambulatory
13.4. North America Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn) and Forecasts, By Clinical Phase
13.4.1. Pre-clinical
13.4.2. Phase 1
13.4.3. Phase 2
13.4.4. Phase 3
13.4.5. Phase 4
13.4.6. Approval
13.5. North America Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn) and Forecasts, By Age Group
13.5.1. 1 to 4 years
13.5.2. 5 to 7 years
13.5.3. 8 to 11 years
13.5.4. 12 to 19 years
13.5.5. Adults
13.6. North America Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn) and Forecasts, By Treatment
13.6.1. Medications
13.6.1.1.
Corticosteroids
13.6.1.2.
Heart
medications
13.6.2. Therapy
13.6.2.1.
Range-of-motion
and stretching exercises
13.6.2.2.
Braces
13.6.2.3.
Mobility
aids
13.6.2.4.
Breathing
assistance
13.6.2.5.
Others
13.6.3. Surgery
13.6.4. Diet and Nutritional supplements
13.6.5. Others
13.7. North America Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn) and Forecasts, By Country
13.7.1. U.S
13.7.1.1.
U.S Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Diagnosis
13.7.1.1.1.
Enzyme
Tests
13.7.1.1.2.
Blood
Tests
13.7.1.1.3.
Genetic
Testing
13.7.1.1.4.
Muscle biopsy
13.7.1.1.5.
CT Scan
13.7.1.1.6.
Electromyogram
(EMG)
13.7.1.1.7.
Electrocardiogram
(ECG or EKG)
13.7.1.1.8.
Others
13.7.1.2.
U.S Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Stages
13.7.1.2.1.
Early
Ambulatory
13.7.1.2.2.
Late
Ambulatory
13.7.1.2.3.
Early
Non-Ambulatory
13.7.1.2.4.
Late
Non-Ambulatory
13.7.1.3.
U.S Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Clinical
Phase
13.7.1.3.1.
Pre-clinical
13.7.1.3.2.
Phase 1
13.7.1.3.3.
Phase 2
13.7.1.3.4.
Phase 3
13.7.1.3.5.
Phase 4
13.7.1.3.6.
Approval
13.7.1.4.
U.S Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Age Group
13.7.1.4.1.
1 to 4
years
13.7.1.4.2.
5 to 7
years
13.7.1.4.3.
8 to 11
years
13.7.1.4.4.
12 to 19
years
13.7.1.4.5.
Adults
13.7.1.5.
U.S Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Treatment
13.7.1.5.1.
Medications
13.7.1.5.1.1. Corticosteroids
13.7.1.5.1.2. Heart medications
13.7.1.5.2.
Therapy
13.7.1.5.2.1. Range-of-motion and stretching exercises
13.7.1.5.2.2. Braces
13.7.1.5.2.3. Mobility aids
13.7.1.5.2.4. Breathing assistance
13.7.1.5.2.5. Others
13.7.1.5.3.
Surgery
13.7.1.5.4.
Diet and
Nutritional supplements
13.7.1.5.5.
Others
13.7.2. Canada
13.7.2.1.
Canada Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Diagnosis
13.7.2.1.1.
Enzyme
Tests
13.7.2.1.2.
Blood
Tests
13.7.2.1.3.
Genetic
Testing
13.7.2.1.4.
Muscle biopsy
13.7.2.1.5.
CT Scan
13.7.2.1.6.
Electromyogram
(EMG)
13.7.2.1.7.
Electrocardiogram
(ECG or EKG)
13.7.2.1.8.
Others
13.7.2.2.
Canada Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Stages
13.7.2.2.1.
Early
Ambulatory
13.7.2.2.2.
Late
Ambulatory
13.7.2.2.3.
Early
Non-Ambulatory
13.7.2.2.4.
Late
Non-Ambulatory
13.7.2.3.
Canada Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Clinical
Phase
13.7.2.3.1.
Pre-clinical
13.7.2.3.2.
Phase 1
13.7.2.3.3.
Phase 2
13.7.2.3.4.
Phase 3
13.7.2.3.5.
Phase 4
13.7.2.3.6.
Approval
13.7.2.4.
Canada Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Age Group
13.7.2.4.1.
1 to 4
years
13.7.2.4.2.
5 to 7
years
13.7.2.4.3.
8 to 11
years
13.7.2.4.4.
12 to 19
years
13.7.2.4.5.
Adults
13.7.2.5.
Canada Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Treatment
13.7.2.5.1.
Medications
13.7.2.5.1.1. Corticosteroids
13.7.2.5.1.2. Heart medications
13.7.2.5.2.
Therapy
13.7.2.5.2.1. Range-of-motion and stretching exercises
13.7.2.5.2.2. Braces
13.7.2.5.2.3. Mobility aids
13.7.2.5.2.4. Breathing assistance
13.7.2.5.2.5. Others
13.7.2.5.3.
Surgery
13.7.2.5.4.
Diet and
Nutritional supplements
13.7.2.5.5.
Others
13.7.3. Mexico
13.7.3.1.
Mexico Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Diagnosis
13.7.3.1.1.
Enzyme
Tests
13.7.3.1.2.
Blood
Tests
13.7.3.1.3.
Genetic
Testing
13.7.3.1.4.
Muscle biopsy
13.7.3.1.5.
CT Scan
13.7.3.1.6.
Electromyogram
(EMG)
13.7.3.1.7.
Electrocardiogram
(ECG or EKG)
13.7.3.1.8.
Others
13.7.3.2.
Mexico Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Stages
13.7.3.2.1.
Early
Ambulatory
13.7.3.2.2.
Late
Ambulatory
13.7.3.2.3.
Early
Non-Ambulatory
13.7.3.2.4.
Late
Non-Ambulatory
13.7.3.3.
Mexico Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Clinical
Phase
13.7.3.3.1.
Pre-clinical
13.7.3.3.2.
Phase 1
13.7.3.3.3.
Phase 2
13.7.3.3.4.
Phase 3
13.7.3.3.5.
Phase 4
13.7.3.3.6.
Approval
13.7.3.4.
Mexico Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Age Group
13.7.3.4.1.
1 to 4
years
13.7.3.4.2.
5 to 7
years
13.7.3.4.3.
8 to 11
years
13.7.3.4.4.
12 to 19
years
13.7.3.4.5.
Adults
13.7.3.5.
Mexico Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Treatment
13.7.3.5.1.
Medications
13.7.3.5.1.1. Corticosteroids
13.7.3.5.1.2. Heart medications
13.7.3.5.2.
Therapy
13.7.3.5.2.1. Range-of-motion and stretching exercises
13.7.3.5.2.2. Braces
13.7.3.5.2.3. Mobility aids
13.7.3.5.2.4. Breathing assistance
13.7.3.5.2.5. Others
13.7.3.5.3.
Surgery
13.7.3.5.4.
Diet and
Nutritional supplements
13.7.3.5.5.
Others
13.7.4. Rest of North America
13.7.4.1.
Rest of
North America Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Diagnosis
13.7.4.1.1.
Enzyme
Tests
13.7.4.1.2.
Blood
Tests
13.7.4.1.3.
Genetic
Testing
13.7.4.1.4.
Muscle biopsy
13.7.4.1.5.
CT Scan
13.7.4.1.6.
Electromyogram
(EMG)
13.7.4.1.7.
Electrocardiogram
(ECG or EKG)
13.7.4.1.8.
Others
13.7.4.2.
Rest of
North America Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Stages
13.7.4.2.1.
Early
Ambulatory
13.7.4.2.2.
Late
Ambulatory
13.7.4.2.3.
Early
Non-Ambulatory
13.7.4.2.4.
Late
Non-Ambulatory
13.7.4.3.
Rest of
North America Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Clinical Phase
13.7.4.3.1.
Pre-clinical
13.7.4.3.2.
Phase 1
13.7.4.3.3.
Phase 2
13.7.4.3.4.
Phase 3
13.7.4.3.5.
Phase 4
13.7.4.3.6.
Approval
13.7.4.4.
Rest of
North America Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Age Group
13.7.4.4.1.
1 to 4
years
13.7.4.4.2.
5 to 7
years
13.7.4.4.3.
8 to 11
years
13.7.4.4.4.
12 to 19
years
13.7.4.4.5.
Adults
13.7.4.5.
Rest of
North America Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Treatment
13.7.4.5.1.
Medications
13.7.4.5.1.1. Corticosteroids
13.7.4.5.1.2. Heart medications
13.7.4.5.2.
Therapy
13.7.4.5.2.1. Range-of-motion and stretching exercises
13.7.4.5.2.2. Braces
13.7.4.5.2.3. Mobility aids
13.7.4.5.2.4. Breathing assistance
13.7.4.5.2.5. Others
13.7.4.5.3.
Surgery
13.7.4.5.4.
Diet and
Nutritional supplements
13.7.4.5.5.
Others
13.8. Key Segment for Channeling Investments
13.8.1. By Country
13.8.2. By Diagnosis
13.8.3. By Stages
13.8.4. By Clinical Phase
13.8.5. By Age Group
13.8.6. By Treatment
14. Europe Duchenne Muscular Dystrophy (DMD)
Market Analysis and Forecasts, 2022 - 2030
14.1. Overview
14.1.1. Europe Duchenne Muscular Dystrophy (DMD)
Market Revenue (US$ Mn)
14.2. Europe Duchenne Muscular Dystrophy (DMD)
Market Revenue (US$ Mn) and Forecasts, By Diagnosis
14.2.1. Enzyme Tests
14.2.2. Blood Tests
14.2.3. Genetic Testing
14.2.4. Muscle
biopsy
14.2.5. CT Scan
14.2.6. Electromyogram (EMG)
14.2.7. Electrocardiogram (ECG or EKG)
14.2.8. Others
14.3. Europe Duchenne Muscular Dystrophy (DMD)
Market Revenue (US$ Mn) and Forecasts, By Stages
14.3.1. Early Ambulatory
14.3.2. Late Ambulatory
14.3.3. Early Non-Ambulatory
14.3.4. Late Non-Ambulatory
14.4. Europe Duchenne Muscular Dystrophy (DMD)
Market Revenue (US$ Mn) and Forecasts, By Clinical Phase
14.4.1. Pre-clinical
14.4.2. Phase 1
14.4.3. Phase 2
14.4.4. Phase 3
14.4.5. Phase 4
14.4.6. Approval
14.5. Europe Duchenne Muscular Dystrophy (DMD)
Market Revenue (US$ Mn) and Forecasts, By Age Group
14.5.1. 1 to 4 years
14.5.2. 5 to 7 years
14.5.3. 8 to 11 years
14.5.4. 12 to 19 years
14.5.5. Adults
14.6. Europe Duchenne Muscular Dystrophy (DMD)
Market Revenue (US$ Mn) and Forecasts, By Treatment
14.6.1. Medications
14.6.1.1.
Corticosteroids
14.6.1.2.
Heart
medications
14.6.2. Therapy
14.6.2.1.
Range-of-motion
and stretching exercises
14.6.2.2.
Braces
14.6.2.3.
Mobility
aids
14.6.2.4.
Breathing
assistance
14.6.2.5.
Others
14.6.3. Surgery
14.6.4. Diet and Nutritional supplements
14.6.5. Others
14.7. Europe Duchenne Muscular Dystrophy (DMD)
Market Revenue (US$ Mn) and Forecasts, By Country
14.7.1. France
14.7.1.1.
France Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Diagnosis
14.7.1.1.1.
Enzyme
Tests
14.7.1.1.2.
Blood
Tests
14.7.1.1.3.
Genetic
Testing
14.7.1.1.4.
Muscle biopsy
14.7.1.1.5.
CT Scan
14.7.1.1.6.
Electromyogram
(EMG)
14.7.1.1.7.
Electrocardiogram
(ECG or EKG)
14.7.1.1.8.
Others
14.7.1.2.
France Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Stages
14.7.1.2.1.
Early
Ambulatory
14.7.1.2.2.
Late
Ambulatory
14.7.1.2.3.
Early
Non-Ambulatory
14.7.1.2.4.
Late
Non-Ambulatory
14.7.1.3.
France Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Clinical
Phase
14.7.1.3.1.
Pre-clinical
14.7.1.3.2.
Phase 1
14.7.1.3.3.
Phase 2
14.7.1.3.4.
Phase 3
14.7.1.3.5.
Phase 4
14.7.1.3.6.
Approval
14.7.1.4.
France Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Age Group
14.7.1.4.1.
1 to 4
years
14.7.1.4.2.
5 to 7
years
14.7.1.4.3.
8 to 11
years
14.7.1.4.4.
12 to 19
years
14.7.1.4.5.
Adults
14.7.1.5.
France Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Treatment
14.7.1.5.1.
Medications
14.7.1.5.1.1. Corticosteroids
14.7.1.5.1.2. Heart medications
14.7.1.5.2.
Therapy
14.7.1.5.2.1. Range-of-motion and stretching exercises
14.7.1.5.2.2. Braces
14.7.1.5.2.3. Mobility aids
14.7.1.5.2.4. Breathing assistance
14.7.1.5.2.5. Others
14.7.1.5.3.
Surgery
14.7.1.5.4.
Diet and
Nutritional supplements
14.7.1.5.5.
Others
14.7.2. The UK
14.7.2.1.
The UK Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Diagnosis
14.7.2.1.1.
Enzyme
Tests
14.7.2.1.2.
Blood
Tests
14.7.2.1.3.
Genetic
Testing
14.7.2.1.4.
Muscle biopsy
14.7.2.1.5.
CT Scan
14.7.2.1.6.
Electromyogram
(EMG)
14.7.2.1.7.
Electrocardiogram
(ECG or EKG)
14.7.2.1.8.
Others
14.7.2.2.
The UK Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Stages
14.7.2.2.1.
Early
Ambulatory
14.7.2.2.2.
Late
Ambulatory
14.7.2.2.3.
Early
Non-Ambulatory
14.7.2.2.4.
Late
Non-Ambulatory
14.7.2.3.
The UK Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Clinical
Phase
14.7.2.3.1.
Pre-clinical
14.7.2.3.2.
Phase 1
14.7.2.3.3.
Phase 2
14.7.2.3.4.
Phase 3
14.7.2.3.5.
Phase 4
14.7.2.3.6.
Approval
14.7.2.4.
The UK Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Age Group
14.7.2.4.1.
1 to 4
years
14.7.2.4.2.
5 to 7
years
14.7.2.4.3.
8 to 11
years
14.7.2.4.4.
12 to 19
years
14.7.2.4.5.
Adults
14.7.2.5.
The UK Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Treatment
14.7.2.5.1.
Medications
14.7.2.5.1.1. Corticosteroids
14.7.2.5.1.2. Heart medications
14.7.2.5.2.
Therapy
14.7.2.5.2.1. Range-of-motion and stretching exercises
14.7.2.5.2.2. Braces
14.7.2.5.2.3. Mobility aids
14.7.2.5.2.4. Breathing assistance
14.7.2.5.2.5. Others
14.7.2.5.3.
Surgery
14.7.2.5.4.
Diet and
Nutritional supplements
14.7.2.5.5.
Others
14.7.3. Spain
14.7.3.1.
Spain Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Diagnosis
14.7.3.1.1.
Enzyme
Tests
14.7.3.1.2.
Blood
Tests
14.7.3.1.3.
Genetic
Testing
14.7.3.1.4.
Muscle biopsy
14.7.3.1.5.
CT Scan
14.7.3.1.6.
Electromyogram
(EMG)
14.7.3.1.7.
Electrocardiogram
(ECG or EKG)
14.7.3.1.8.
Others
14.7.3.2.
Spain Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Stages
14.7.3.2.1.
Early
Ambulatory
14.7.3.2.2.
Late
Ambulatory
14.7.3.2.3.
Early
Non-Ambulatory
14.7.3.2.4.
Late
Non-Ambulatory
14.7.3.3.
Spain Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Clinical
Phase
14.7.3.3.1.
Pre-clinical
14.7.3.3.2.
Phase 1
14.7.3.3.3.
Phase 2
14.7.3.3.4.
Phase 3
14.7.3.3.5.
Phase 4
14.7.3.3.6.
Approval
14.7.3.4.
Spain Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Age Group
14.7.3.4.1.
1 to 4
years
14.7.3.4.2.
5 to 7
years
14.7.3.4.3.
8 to 11
years
14.7.3.4.4.
12 to 19
years
14.7.3.4.5.
Adults
14.7.3.5.
Spain Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Treatment
14.7.3.5.1.
Medications
14.7.3.5.1.1. Corticosteroids
14.7.3.5.1.2. Heart medications
14.7.3.5.2.
Therapy
14.7.3.5.2.1. Range-of-motion and stretching exercises
14.7.3.5.2.2. Braces
14.7.3.5.2.3. Mobility aids
14.7.3.5.2.4. Breathing assistance
14.7.3.5.2.5. Others
14.7.3.5.3.
Surgery
14.7.3.5.4.
Diet and
Nutritional supplements
14.7.3.5.5.
Others
14.7.4. Germany
14.7.4.1.
Germany Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Diagnosis
14.7.4.1.1.
Enzyme
Tests
14.7.4.1.2.
Blood
Tests
14.7.4.1.3.
Genetic
Testing
14.7.4.1.4.
Muscle biopsy
14.7.4.1.5.
CT Scan
14.7.4.1.6.
Electromyogram
(EMG)
14.7.4.1.7.
Electrocardiogram
(ECG or EKG)
14.7.4.1.8.
Others
14.7.4.2.
Germany Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Stages
14.7.4.2.1.
Early
Ambulatory
14.7.4.2.2.
Late
Ambulatory
14.7.4.2.3.
Early Non-Ambulatory
14.7.4.2.4.
Late
Non-Ambulatory
14.7.4.3.
Germany Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Clinical
Phase
14.7.4.3.1.
Pre-clinical
14.7.4.3.2.
Phase 1
14.7.4.3.3.
Phase 2
14.7.4.3.4.
Phase 3
14.7.4.3.5.
Phase 4
14.7.4.3.6.
Approval
14.7.4.4.
Germany Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Age Group
14.7.4.4.1.
1 to 4
years
14.7.4.4.2.
5 to 7
years
14.7.4.4.3.
8 to 11
years
14.7.4.4.4.
12 to 19
years
14.7.4.4.5.
Adults
14.7.4.5.
Germany Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Treatment
14.7.4.5.1.
Medications
14.7.4.5.1.1. Corticosteroids
14.7.4.5.1.2. Heart medications
14.7.4.5.2.
Therapy
14.7.4.5.2.1. Range-of-motion and stretching exercises
14.7.4.5.2.2. Braces
14.7.4.5.2.3. Mobility aids
14.7.4.5.2.4. Breathing assistance
14.7.4.5.2.5. Others
14.7.4.5.3.
Surgery
14.7.4.5.4.
Diet and
Nutritional supplements
14.7.4.5.5.
Others
14.7.5. Italy
14.7.5.1.
Italy Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Diagnosis
14.7.5.1.1.
Enzyme
Tests
14.7.5.1.2.
Blood Tests
14.7.5.1.3.
Genetic
Testing
14.7.5.1.4.
Muscle biopsy
14.7.5.1.5.
CT Scan
14.7.5.1.6.
Electromyogram
(EMG)
14.7.5.1.7.
Electrocardiogram
(ECG or EKG)
14.7.5.1.8.
Others
14.7.5.2.
Italy Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Stages
14.7.5.2.1.
Early
Ambulatory
14.7.5.2.2.
Late
Ambulatory
14.7.5.2.3.
Early
Non-Ambulatory
14.7.5.2.4.
Late Non-Ambulatory
14.7.5.3.
Italy Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Clinical
Phase
14.7.5.3.1.
Pre-clinical
14.7.5.3.2.
Phase 1
14.7.5.3.3.
Phase 2
14.7.5.3.4.
Phase 3
14.7.5.3.5.
Phase 4
14.7.5.3.6.
Approval
14.7.5.4.
Italy Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Age Group
14.7.5.4.1.
1 to 4
years
14.7.5.4.2.
5 to 7
years
14.7.5.4.3.
8 to 11
years
14.7.5.4.4.
12 to 19
years
14.7.5.4.5.
Adults
14.7.5.5.
Italy Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Treatment
14.7.5.5.1.
Medications
14.7.5.5.1.1. Corticosteroids
14.7.5.5.1.2. Heart medications
14.7.5.5.2.
Therapy
14.7.5.5.2.1. Range-of-motion and stretching exercises
14.7.5.5.2.2. Braces
14.7.5.5.2.3. Mobility aids
14.7.5.5.2.4. Breathing assistance
14.7.5.5.2.5. Others
14.7.5.5.3.
Surgery
14.7.5.5.4.
Diet and
Nutritional supplements
14.7.5.5.5.
Others
14.7.6. Nordic Countries
14.7.6.1.
Nordic
Countries Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Diagnosis
14.7.6.1.1.
Enzyme
Tests
14.7.6.1.2.
Blood
Tests
14.7.6.1.3.
Genetic
Testing
14.7.6.1.4.
Muscle biopsy
14.7.6.1.5.
CT Scan
14.7.6.1.6.
Electromyogram
(EMG)
14.7.6.1.7.
Electrocardiogram
(ECG or EKG)
14.7.6.1.8.
Others
14.7.6.2.
Nordic
Countries Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Stages
14.7.6.2.1.
Early
Ambulatory
14.7.6.2.2.
Late
Ambulatory
14.7.6.2.3.
Early
Non-Ambulatory
14.7.6.2.4.
Late Non-Ambulatory
14.7.6.3.
Nordic
Countries Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Clinical Phase
14.7.6.3.1.
Pre-clinical
14.7.6.3.2.
Phase 1
14.7.6.3.3.
Phase 2
14.7.6.3.4.
Phase 3
14.7.6.3.5.
Phase 4
14.7.6.3.6.
Approval
14.7.6.4.
Nordic
Countries Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Age Group
14.7.6.4.1.
1 to 4
years
14.7.6.4.2.
5 to 7
years
14.7.6.4.3.
8 to 11
years
14.7.6.4.4.
12 to 19
years
14.7.6.4.5.
Adults
14.7.6.5.
Nordic
Countries Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Treatment
14.7.6.5.1.
Medications
14.7.6.5.1.1. Corticosteroids
14.7.6.5.1.2. Heart medications
14.7.6.5.2.
Therapy
14.7.6.5.2.1. Range-of-motion and stretching exercises
14.7.6.5.2.2. Braces
14.7.6.5.2.3. Mobility aids
14.7.6.5.2.4. Breathing assistance
14.7.6.5.2.5. Others
14.7.6.5.3.
Surgery
14.7.6.5.4.
Diet and
Nutritional supplements
14.7.6.5.5.
Others
14.7.6.6.
Nordic
Countries Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Country
14.7.6.6.1.
Denmark
14.7.6.6.2.
Finland
14.7.6.6.3.
Iceland
14.7.6.6.4.
Sweden
14.7.6.6.5.
Norway
14.7.7. Benelux Union
14.7.7.1.
Benelux
Union Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Diagnosis
14.7.7.1.1.
Enzyme
Tests
14.7.7.1.2.
Blood
Tests
14.7.7.1.3.
Genetic
Testing
14.7.7.1.4.
Muscle biopsy
14.7.7.1.5.
CT Scan
14.7.7.1.6.
Electromyogram
(EMG)
14.7.7.1.7.
Electrocardiogram
(ECG or EKG)
14.7.7.1.8.
Others
14.7.7.2.
Benelux
Union Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Stages
14.7.7.2.1.
Early
Ambulatory
14.7.7.2.2.
Late
Ambulatory
14.7.7.2.3.
Early
Non-Ambulatory
14.7.7.2.4.
Late
Non-Ambulatory
14.7.7.3.
Benelux
Union Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Clinical Phase
14.7.7.3.1.
Pre-clinical
14.7.7.3.2.
Phase 1
14.7.7.3.3.
Phase 2
14.7.7.3.4.
Phase 3
14.7.7.3.5.
Phase 4
14.7.7.3.6.
Approval
14.7.7.4.
Benelux
Union Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Age Group
14.7.7.4.1.
1 to 4
years
14.7.7.4.2.
5 to 7
years
14.7.7.4.3.
8 to 11
years
14.7.7.4.4.
12 to 19
years
14.7.7.4.5.
Adults
14.7.7.5.
Benelux
Union Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Treatment
14.7.7.5.1.
Medications
14.7.7.5.1.1. Corticosteroids
14.7.7.5.1.2. Heart medications
14.7.7.5.2.
Therapy
14.7.7.5.2.1. Range-of-motion and stretching exercises
14.7.7.5.2.2. Braces
14.7.7.5.2.3. Mobility aids
14.7.7.5.2.4. Breathing assistance
14.7.7.5.2.5. Others
14.7.7.5.3.
Surgery
14.7.7.5.4.
Diet and
Nutritional supplements
14.7.7.5.5.
Others
14.7.7.6.
Benelux
Union Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Country
14.7.7.6.1.
Belgium
14.7.7.6.2.
The
Netherlands
14.7.7.6.3.
Luxembourg
14.7.8. Rest of Europe
14.7.8.1.
Rest of
Europe Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Diagnosis
14.7.8.1.1.
Enzyme
Tests
14.7.8.1.2.
Blood
Tests
14.7.8.1.3.
Genetic
Testing
14.7.8.1.4.
Muscle biopsy
14.7.8.1.5.
CT Scan
14.7.8.1.6.
Electromyogram
(EMG)
14.7.8.1.7.
Electrocardiogram
(ECG or EKG)
14.7.8.1.8.
Others
14.7.8.2.
Rest of
Europe Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Stages
14.7.8.2.1.
Early
Ambulatory
14.7.8.2.2.
Late
Ambulatory
14.7.8.2.3.
Early
Non-Ambulatory
14.7.8.2.4.
Late
Non-Ambulatory
14.7.8.3.
Rest of
Europe Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Clinical Phase
14.7.8.3.1.
Pre-clinical
14.7.8.3.2.
Phase 1
14.7.8.3.3.
Phase 2
14.7.8.3.4.
Phase 3
14.7.8.3.5.
Phase 4
14.7.8.3.6.
Approval
14.7.8.4.
Rest of
Europe Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Age Group
14.7.8.4.1.
1 to 4
years
14.7.8.4.2.
5 to 7
years
14.7.8.4.3.
8 to 11
years
14.7.8.4.4.
12 to 19
years
14.7.8.4.5.
Adults
14.7.8.5.
Rest of
Europe Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Treatment
14.7.8.5.1.
Medications
14.7.8.5.1.1. Corticosteroids
14.7.8.5.1.2. Heart medications
14.7.8.5.2.
Therapy
14.7.8.5.2.1. Range-of-motion and stretching exercises
14.7.8.5.2.2. Braces
14.7.8.5.2.3. Mobility aids
14.7.8.5.2.4. Breathing assistance
14.7.8.5.2.5. Others
14.7.8.5.3.
Surgery
14.7.8.5.4.
Diet and
Nutritional supplements
14.7.8.5.5.
Others
14.8. Key Segment for Channeling Investments
14.8.1. By Country
14.8.2. By Diagnosis
14.8.3. By Stages
14.8.4. By Clinical Phase
14.8.5. By Age Group
14.8.6. By Treatment
15. Asia Pacific Duchenne Muscular Dystrophy
(DMD) Market Analysis and Forecasts, 2022 - 2030
15.1. Overview
15.1.1. Asia Pacific Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn)
15.2. Asia Pacific Duchenne Muscular Dystrophy (DMD)
Market Revenue (US$ Mn) and Forecasts, By Diagnosis
15.2.1. Enzyme Tests
15.2.2. Blood Tests
15.2.3. Genetic Testing
15.2.4. Muscle
biopsy
15.2.5. CT Scan
15.2.6. Electromyogram (EMG)
15.2.7. Electrocardiogram (ECG or EKG)
15.2.8. Others
15.3. Asia Pacific Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn) and Forecasts, By Stages
15.3.1. Early Ambulatory
15.3.2. Late Ambulatory
15.3.3. Early Non-Ambulatory
15.3.4. Late Non-Ambulatory
15.4. Asia Pacific Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn) and Forecasts, By Clinical Phase
15.4.1. Pre-clinical
15.4.2. Phase 1
15.4.3. Phase 2
15.4.4. Phase 3
15.4.5. Phase 4
15.4.6. Approval
15.5. Asia Pacific Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn) and Forecasts, By Age Group
15.5.1. 1 to 4 years
15.5.2. 5 to 7 years
15.5.3. 8 to 11 years
15.5.4. 12 to 19 years
15.5.5. Adults
15.6. Asia Pacific Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn) and Forecasts, By Treatment
15.6.1. Medications
15.6.1.1.
Corticosteroids
15.6.1.2.
Heart
medications
15.6.2. Therapy
15.6.2.1.
Range-of-motion
and stretching exercises
15.6.2.2.
Braces
15.6.2.3.
Mobility
aids
15.6.2.4.
Breathing
assistance
15.6.2.5.
Others
15.6.3. Surgery
15.6.4. Diet and Nutritional supplements
15.6.5. Others
15.7. Asia Pacific Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn) and Forecasts, By Country
15.7.1. China
15.7.1.1.
China Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Diagnosis
15.7.1.1.1.
Enzyme
Tests
15.7.1.1.2.
Blood
Tests
15.7.1.1.3.
Genetic
Testing
15.7.1.1.4.
Muscle biopsy
15.7.1.1.5.
CT Scan
15.7.1.1.6.
Electromyogram
(EMG)
15.7.1.1.7.
Electrocardiogram
(ECG or EKG)
15.7.1.1.8.
Others
15.7.1.2.
China Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Stages
15.7.1.2.1.
Early
Ambulatory
15.7.1.2.2.
Late
Ambulatory
15.7.1.2.3.
Early
Non-Ambulatory
15.7.1.2.4.
Late
Non-Ambulatory
15.7.1.3.
China Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Clinical
Phase
15.7.1.3.1.
Pre-clinical
15.7.1.3.2.
Phase 1
15.7.1.3.3.
Phase 2
15.7.1.3.4.
Phase 3
15.7.1.3.5.
Phase 4
15.7.1.3.6.
Approval
15.7.1.4.
China Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Age Group
15.7.1.4.1.
1 to 4
years
15.7.1.4.2.
5 to 7
years
15.7.1.4.3.
8 to 11
years
15.7.1.4.4.
12 to 19
years
15.7.1.4.5.
Adults
15.7.1.5.
China Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Treatment
15.7.1.5.1.
Medications
15.7.1.5.1.1. Corticosteroids
15.7.1.5.1.2. Heart medications
15.7.1.5.2.
Therapy
15.7.1.5.2.1. Range-of-motion and stretching exercises
15.7.1.5.2.2. Braces
15.7.1.5.2.3. Mobility aids
15.7.1.5.2.4. Breathing assistance
15.7.1.5.2.5. Others
15.7.1.5.3.
Surgery
15.7.1.5.4.
Diet and
Nutritional supplements
15.7.1.5.5.
Others
15.7.2. Japan
15.7.2.1.
Japan Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Diagnosis
15.7.2.1.1.
Enzyme
Tests
15.7.2.1.2.
Blood
Tests
15.7.2.1.3.
Genetic
Testing
15.7.2.1.4.
Muscle biopsy
15.7.2.1.5.
CT Scan
15.7.2.1.6.
Electromyogram
(EMG)
15.7.2.1.7.
Electrocardiogram
(ECG or EKG)
15.7.2.1.8.
Others
15.7.2.2.
Japan Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Stages
15.7.2.2.1.
Early
Ambulatory
15.7.2.2.2.
Late
Ambulatory
15.7.2.2.3.
Early
Non-Ambulatory
15.7.2.2.4.
Late
Non-Ambulatory
15.7.2.3.
Japan Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Clinical
Phase
15.7.2.3.1.
Pre-clinical
15.7.2.3.2.
Phase 1
15.7.2.3.3.
Phase 2
15.7.2.3.4.
Phase 3
15.7.2.3.5.
Phase 4
15.7.2.3.6.
Approval
15.7.2.4.
Japan Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Age Group
15.7.2.4.1.
1 to 4
years
15.7.2.4.2.
5 to 7
years
15.7.2.4.3.
8 to 11
years
15.7.2.4.4.
12 to 19
years
15.7.2.4.5.
Adults
15.7.2.5.
Japan Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Treatment
15.7.2.5.1.
Medications
15.7.2.5.1.1. Corticosteroids
15.7.2.5.1.2. Heart medications
15.7.2.5.2.
Therapy
15.7.2.5.2.1. Range-of-motion and stretching exercises
15.7.2.5.2.2. Braces
15.7.2.5.2.3. Mobility aids
15.7.2.5.2.4. Breathing assistance
15.7.2.5.2.5. Others
15.7.2.5.3.
Surgery
15.7.2.5.4.
Diet and
Nutritional supplements
15.7.2.5.5.
Others
15.7.3. India
15.7.3.1.
India Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Diagnosis
15.7.3.1.1.
Enzyme
Tests
15.7.3.1.2.
Blood
Tests
15.7.3.1.3.
Genetic
Testing
15.7.3.1.4.
Muscle biopsy
15.7.3.1.5.
CT Scan
15.7.3.1.6.
Electromyogram
(EMG)
15.7.3.1.7.
Electrocardiogram
(ECG or EKG)
15.7.3.1.8.
Others
15.7.3.2.
India Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Stages
15.7.3.2.1.
Early
Ambulatory
15.7.3.2.2.
Late
Ambulatory
15.7.3.2.3.
Early
Non-Ambulatory
15.7.3.2.4.
Late
Non-Ambulatory
15.7.3.3.
India Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Clinical
Phase
15.7.3.3.1.
Pre-clinical
15.7.3.3.2.
Phase 1
15.7.3.3.3.
Phase 2
15.7.3.3.4.
Phase 3
15.7.3.3.5.
Phase 4
15.7.3.3.6.
Approval
15.7.3.4.
India Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Age Group
15.7.3.4.1.
1 to 4
years
15.7.3.4.2.
5 to 7
years
15.7.3.4.3.
8 to 11
years
15.7.3.4.4.
12 to 19
years
15.7.3.4.5.
Adults
15.7.3.5.
India Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Treatment
15.7.3.5.1.
Medications
15.7.3.5.1.1. Corticosteroids
15.7.3.5.1.2. Heart medications
15.7.3.5.2.
Therapy
15.7.3.5.2.1. Range-of-motion and stretching exercises
15.7.3.5.2.2. Braces
15.7.3.5.2.3. Mobility aids
15.7.3.5.2.4. Breathing assistance
15.7.3.5.2.5. Others
15.7.3.5.3.
Surgery
15.7.3.5.4.
Diet and
Nutritional supplements
15.7.3.5.5.
Others
15.7.4. New Zealand
15.7.4.1.
New
Zealand Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Diagnosis
15.7.4.1.1.
Enzyme
Tests
15.7.4.1.2.
Blood
Tests
15.7.4.1.3.
Genetic
Testing
15.7.4.1.4.
Muscle biopsy
15.7.4.1.5.
CT Scan
15.7.4.1.6.
Electromyogram
(EMG)
15.7.4.1.7.
Electrocardiogram
(ECG or EKG)
15.7.4.1.8.
Others
15.7.4.2.
New
Zealand Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Stages
15.7.4.2.1.
Early
Ambulatory
15.7.4.2.2.
Late
Ambulatory
15.7.4.2.3.
Early
Non-Ambulatory
15.7.4.2.4.
Late
Non-Ambulatory
15.7.4.3.
New
Zealand Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Clinical Phase
15.7.4.3.1.
Pre-clinical
15.7.4.3.2.
Phase 1
15.7.4.3.3.
Phase 2
15.7.4.3.4.
Phase 3
15.7.4.3.5.
Phase 4
15.7.4.3.6.
Approval
15.7.4.4.
New
Zealand Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Age Group
15.7.4.4.1.
1 to 4
years
15.7.4.4.2.
5 to 7
years
15.7.4.4.3.
8 to 11
years
15.7.4.4.4.
12 to 19
years
15.7.4.4.5.
Adults
15.7.4.5.
New
Zealand Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Treatment
15.7.4.5.1.
Medications
15.7.4.5.1.1. Corticosteroids
15.7.4.5.1.2. Heart medications
15.7.4.5.2.
Therapy
15.7.4.5.2.1. Range-of-motion and stretching exercises
15.7.4.5.2.2. Braces
15.7.4.5.2.3. Mobility aids
15.7.4.5.2.4. Breathing assistance
15.7.4.5.2.5. Others
15.7.4.5.3.
Surgery
15.7.4.5.4.
Diet and
Nutritional supplements
15.7.4.5.5.
Others
15.7.5. Australia
15.7.5.1.
Australia
Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By
Diagnosis
15.7.5.1.1.
Enzyme
Tests
15.7.5.1.2.
Blood
Tests
15.7.5.1.3.
Genetic
Testing
15.7.5.1.4.
Muscle biopsy
15.7.5.1.5.
CT Scan
15.7.5.1.6.
Electromyogram
(EMG)
15.7.5.1.7.
Electrocardiogram
(ECG or EKG)
15.7.5.1.8.
Others
15.7.5.2.
Australia
Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Stages
15.7.5.2.1.
Early
Ambulatory
15.7.5.2.2.
Late
Ambulatory
15.7.5.2.3.
Early
Non-Ambulatory
15.7.5.2.4.
Late
Non-Ambulatory
15.7.5.3.
Australia
Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By
Clinical Phase
15.7.5.3.1.
Pre-clinical
15.7.5.3.2.
Phase 1
15.7.5.3.3.
Phase 2
15.7.5.3.4.
Phase 3
15.7.5.3.5.
Phase 4
15.7.5.3.6.
Approval
15.7.5.4.
Australia
Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Age
Group
15.7.5.4.1.
1 to 4
years
15.7.5.4.2.
5 to 7
years
15.7.5.4.3.
8 to 11
years
15.7.5.4.4.
12 to 19
years
15.7.5.4.5.
Adults
15.7.5.5.
Australia
Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By
Treatment
15.7.5.5.1.
Medications
15.7.5.5.1.1. Corticosteroids
15.7.5.5.1.2. Heart medications
15.7.5.5.2.
Therapy
15.7.5.5.2.1. Range-of-motion and stretching exercises
15.7.5.5.2.2. Braces
15.7.5.5.2.3. Mobility aids
15.7.5.5.2.4. Breathing assistance
15.7.5.5.2.5. Others
15.7.5.5.3.
Surgery
15.7.5.5.4.
Diet and
Nutritional supplements
15.7.5.5.5.
Others
15.7.6. South Korea
15.7.6.1.
South
Korea Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Diagnosis
15.7.6.1.1.
Enzyme
Tests
15.7.6.1.2.
Blood
Tests
15.7.6.1.3.
Genetic
Testing
15.7.6.1.4.
Muscle biopsy
15.7.6.1.5.
CT Scan
15.7.6.1.6.
Electromyogram
(EMG)
15.7.6.1.7.
Electrocardiogram
(ECG or EKG)
15.7.6.1.8.
Others
15.7.6.2.
South
Korea Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Stages
15.7.6.2.1.
Early
Ambulatory
15.7.6.2.2.
Late
Ambulatory
15.7.6.2.3.
Early
Non-Ambulatory
15.7.6.2.4.
Late
Non-Ambulatory
15.7.6.3.
South
Korea Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Clinical Phase
15.7.6.3.1.
Pre-clinical
15.7.6.3.2.
Phase 1
15.7.6.3.3.
Phase 2
15.7.6.3.4.
Phase 3
15.7.6.3.5.
Phase 4
15.7.6.3.6.
Approval
15.7.6.4.
South
Korea Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Age Group
15.7.6.4.1.
1 to 4
years
15.7.6.4.2.
5 to 7
years
15.7.6.4.3.
8 to 11
years
15.7.6.4.4.
12 to 19
years
15.7.6.4.5.
Adults
15.7.6.5.
South
Korea Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Treatment
15.7.6.5.1.
Medications
15.7.6.5.1.1. Corticosteroids
15.7.6.5.1.2. Heart medications
15.7.6.5.2.
Therapy
15.7.6.5.2.1. Range-of-motion and stretching exercises
15.7.6.5.2.2. Braces
15.7.6.5.2.3. Mobility aids
15.7.6.5.2.4. Breathing assistance
15.7.6.5.2.5. Others
15.7.6.5.3.
Surgery
15.7.6.5.4.
Diet and
Nutritional supplements
15.7.6.5.5.
Others
15.7.7. Southeast Asia
15.7.7.1.
Southeast
Asia Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Diagnosis
15.7.7.1.1.
Enzyme
Tests
15.7.7.1.2.
Blood
Tests
15.7.7.1.3.
Genetic
Testing
15.7.7.1.4.
Muscle biopsy
15.7.7.1.5.
CT Scan
15.7.7.1.6.
Electromyogram
(EMG)
15.7.7.1.7.
Electrocardiogram
(ECG or EKG)
15.7.7.1.8.
Others
15.7.7.2.
Southeast
Asia Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Stages
15.7.7.2.1.
Early
Ambulatory
15.7.7.2.2.
Late
Ambulatory
15.7.7.2.3.
Early
Non-Ambulatory
15.7.7.2.4.
Late
Non-Ambulatory
15.7.7.3.
Southeast
Asia Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Clinical Phase
15.7.7.3.1.
Pre-clinical
15.7.7.3.2.
Phase 1
15.7.7.3.3.
Phase 2
15.7.7.3.4.
Phase 3
15.7.7.3.5.
Phase 4
15.7.7.3.6.
Approval
15.7.7.4.
Southeast
Asia Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Age Group
15.7.7.4.1.
1 to 4
years
15.7.7.4.2.
5 to 7
years
15.7.7.4.3.
8 to 11
years
15.7.7.4.4.
12 to 19
years
15.7.7.4.5.
Adults
15.7.7.5.
Southeast
Asia Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Treatment
15.7.7.5.1.
Medications
15.7.7.5.1.1. Corticosteroids
15.7.7.5.1.2. Heart medications
15.7.7.5.2.
Therapy
15.7.7.5.2.1. Range-of-motion and stretching exercises
15.7.7.5.2.2. Braces
15.7.7.5.2.3. Mobility aids
15.7.7.5.2.4. Breathing assistance
15.7.7.5.2.5. Others
15.7.7.5.3.
Surgery
15.7.7.5.4.
Diet and
Nutritional supplements
15.7.7.5.5.
Others
15.7.7.6.
Southeast
Asia Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Country
15.7.7.6.1.
Indonesia
15.7.7.6.2.
Thailand
15.7.7.6.3.
Malaysia
15.7.7.6.4.
Singapore
15.7.7.6.5.
Rest of
Southeast Asia
15.7.8. Rest of Asia Pacific
15.7.8.1.
Rest of
Asia Pacific Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Diagnosis
15.7.8.1.1.
Enzyme
Tests
15.7.8.1.2.
Blood
Tests
15.7.8.1.3.
Genetic
Testing
15.7.8.1.4.
Muscle biopsy
15.7.8.1.5.
CT Scan
15.7.8.1.6.
Electromyogram
(EMG)
15.7.8.1.7.
Electrocardiogram
(ECG or EKG)
15.7.8.1.8.
Others
15.7.8.2.
Rest of
Asia Pacific Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Stages
15.7.8.2.1.
Early
Ambulatory
15.7.8.2.2.
Late
Ambulatory
15.7.8.2.3.
Early
Non-Ambulatory
15.7.8.2.4.
Late
Non-Ambulatory
15.7.8.3.
Rest of
Asia Pacific Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Clinical Phase
15.7.8.3.1.
Pre-clinical
15.7.8.3.2.
Phase 1
15.7.8.3.3.
Phase 2
15.7.8.3.4.
Phase 3
15.7.8.3.5.
Phase 4
15.7.8.3.6.
Approval
15.7.8.4.
Rest of
Asia Pacific Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Age Group
15.7.8.4.1.
1 to 4
years
15.7.8.4.2.
5 to 7
years
15.7.8.4.3.
8 to 11
years
15.7.8.4.4.
12 to 19
years
15.7.8.4.5.
Adults
15.7.8.5.
Rest of
Asia Pacific Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Treatment
15.7.8.5.1.
Medications
15.7.8.5.1.1. Corticosteroids
15.7.8.5.1.2. Heart medications
15.7.8.5.2.
Therapy
15.7.8.5.2.1. Range-of-motion and stretching exercises
15.7.8.5.2.2. Braces
15.7.8.5.2.3. Mobility aids
15.7.8.5.2.4. Breathing assistance
15.7.8.5.2.5. Others
15.7.8.5.3.
Surgery
15.7.8.5.4.
Diet and
Nutritional supplements
15.7.8.5.5.
Others
15.8. Key Segment for Channeling Investments
15.8.1. By Country
15.8.2. By Diagnosis
15.8.3. By Stages
15.8.4. By Clinical Phase
15.8.5. By Age Group
15.8.6. By Treatment
16. Middle East and Africa Duchenne Muscular
Dystrophy (DMD) Market Analysis and Forecasts, 2022 - 2030
16.1. Overview
16.1.1. Middle East and Africa Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn)
16.2. Middle East and Africa Duchenne Muscular
Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Diagnosis
16.2.1. Enzyme Tests
16.2.2. Blood Tests
16.2.3. Genetic Testing
16.2.4. Muscle
biopsy
16.2.5. CT Scan
16.2.6. Electromyogram (EMG)
16.2.7. Electrocardiogram (ECG or EKG)
16.2.8. Others
16.3. Middle East and Africa Duchenne Muscular
Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Stages
16.3.1. Early Ambulatory
16.3.2. Late Ambulatory
16.3.3. Early Non-Ambulatory
16.3.4. Late Non-Ambulatory
16.4. Middle East and Africa Duchenne Muscular
Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Clinical Phase
16.4.1. Pre-clinical
16.4.2. Phase 1
16.4.3. Phase 2
16.4.4. Phase 3
16.4.5. Phase 4
16.4.6. Approval
16.5. Middle East and Africa Duchenne Muscular
Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Age Group
16.5.1. 1 to 4 years
16.5.2. 5 to 7 years
16.5.3. 8 to 11 years
16.5.4. 12 to 19 years
16.5.5. Adults
16.6. Middle East and Africa Duchenne Muscular
Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Treatment
16.6.1. Medications
16.6.1.1.
Corticosteroids
16.6.1.2.
Heart
medications
16.6.2. Therapy
16.6.2.1.
Range-of-motion
and stretching exercises
16.6.2.2.
Braces
16.6.2.3.
Mobility
aids
16.6.2.4.
Breathing
assistance
16.6.2.5.
Others
16.6.3. Surgery
16.6.4. Diet and Nutritional supplements
16.6.5. Others
16.7. Middle East and Africa Duchenne Muscular
Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Country
16.7.1. Saudi Arabia
16.7.1.1.
Saudi
Arabia Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Diagnosis
16.7.1.1.1.
Enzyme
Tests
16.7.1.1.2.
Blood
Tests
16.7.1.1.3.
Genetic
Testing
16.7.1.1.4.
Muscle biopsy
16.7.1.1.5.
CT Scan
16.7.1.1.6.
Electromyogram
(EMG)
16.7.1.1.7.
Electrocardiogram
(ECG or EKG)
16.7.1.1.8.
Others
16.7.1.2.
Saudi
Arabia Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Stages
16.7.1.2.1.
Early
Ambulatory
16.7.1.2.2.
Late
Ambulatory
16.7.1.2.3.
Early
Non-Ambulatory
16.7.1.2.4.
Late
Non-Ambulatory
16.7.1.3.
Saudi
Arabia Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Clinical Phase
16.7.1.3.1.
Pre-clinical
16.7.1.3.2.
Phase 1
16.7.1.3.3.
Phase 2
16.7.1.3.4.
Phase 3
16.7.1.3.5.
Phase 4
16.7.1.3.6.
Approval
16.7.1.4.
Saudi
Arabia Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Age Group
16.7.1.4.1.
1 to 4
years
16.7.1.4.2.
5 to 7
years
16.7.1.4.3.
8 to 11
years
16.7.1.4.4.
12 to 19
years
16.7.1.4.5.
Adults
16.7.1.5.
Saudi
Arabia Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Treatment
16.7.1.5.1.
Medications
16.7.1.5.1.1. Corticosteroids
16.7.1.5.1.2. Heart medications
16.7.1.5.2.
Therapy
16.7.1.5.2.1. Range-of-motion and stretching exercises
16.7.1.5.2.2. Braces
16.7.1.5.2.3. Mobility aids
16.7.1.5.2.4. Breathing assistance
16.7.1.5.2.5. Others
16.7.1.5.3.
Surgery
16.7.1.5.4.
Diet and
Nutritional supplements
16.7.1.5.5.
Others
16.7.2. UAE
16.7.2.1.
UAE Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Diagnosis
16.7.2.1.1.
Enzyme
Tests
16.7.2.1.2.
Blood
Tests
16.7.2.1.3.
Genetic
Testing
16.7.2.1.4.
Muscle biopsy
16.7.2.1.5.
CT Scan
16.7.2.1.6.
Electromyogram
(EMG)
16.7.2.1.7.
Electrocardiogram
(ECG or EKG)
16.7.2.1.8.
Others
16.7.2.2.
UAE Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Stages
16.7.2.2.1.
Early
Ambulatory
16.7.2.2.2.
Late
Ambulatory
16.7.2.2.3.
Early
Non-Ambulatory
16.7.2.2.4.
Late
Non-Ambulatory
16.7.2.3.
UAE Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Clinical
Phase
16.7.2.3.1.
Pre-clinical
16.7.2.3.2.
Phase 1
16.7.2.3.3.
Phase 2
16.7.2.3.4.
Phase 3
16.7.2.3.5.
Phase 4
16.7.2.3.6.
Approval
16.7.2.4.
UAE Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Age Group
16.7.2.4.1.
1 to 4
years
16.7.2.4.2.
5 to 7
years
16.7.2.4.3.
8 to 11
years
16.7.2.4.4.
12 to 19
years
16.7.2.4.5.
Adults
16.7.2.5.
UAE Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Treatment
16.7.2.5.1.
Medications
16.7.2.5.1.1. Corticosteroids
16.7.2.5.1.2. Heart medications
16.7.2.5.2.
Therapy
16.7.2.5.2.1. Range-of-motion and stretching exercises
16.7.2.5.2.2. Braces
16.7.2.5.2.3. Mobility aids
16.7.2.5.2.4. Breathing assistance
16.7.2.5.2.5. Others
16.7.2.5.3.
Surgery
16.7.2.5.4.
Diet and
Nutritional supplements
16.7.2.5.5.
Others
16.7.3. Egypt
16.7.3.1.
Egypt Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Diagnosis
16.7.3.1.1.
Enzyme Tests
16.7.3.1.2.
Blood
Tests
16.7.3.1.3.
Genetic
Testing
16.7.3.1.4.
Muscle biopsy
16.7.3.1.5.
CT Scan
16.7.3.1.6.
Electromyogram
(EMG)
16.7.3.1.7.
Electrocardiogram
(ECG or EKG)
16.7.3.1.8.
Others
16.7.3.2.
Egypt Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Stages
16.7.3.2.1.
Early
Ambulatory
16.7.3.2.2.
Late
Ambulatory
16.7.3.2.3.
Early
Non-Ambulatory
16.7.3.2.4.
Late
Non-Ambulatory
16.7.3.3.
Egypt Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Clinical
Phase
16.7.3.3.1.
Pre-clinical
16.7.3.3.2.
Phase 1
16.7.3.3.3.
Phase 2
16.7.3.3.4.
Phase 3
16.7.3.3.5.
Phase 4
16.7.3.3.6.
Approval
16.7.3.4.
Egypt Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Age Group
16.7.3.4.1.
1 to 4
years
16.7.3.4.2.
5 to 7
years
16.7.3.4.3.
8 to 11
years
16.7.3.4.4.
12 to 19
years
16.7.3.4.5.
Adults
16.7.3.5.
Egypt Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Treatment
16.7.3.5.1.
Medications
16.7.3.5.1.1. Corticosteroids
16.7.3.5.1.2. Heart medications
16.7.3.5.2.
Therapy
16.7.3.5.2.1. Range-of-motion and stretching exercises
16.7.3.5.2.2. Braces
16.7.3.5.2.3. Mobility aids
16.7.3.5.2.4. Breathing assistance
16.7.3.5.2.5. Others
16.7.3.5.3.
Surgery
16.7.3.5.4.
Diet and
Nutritional supplements
16.7.3.5.5.
Others
16.7.4. Kuwait
16.7.4.1.
Kuwait Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Diagnosis
16.7.4.1.1.
Enzyme
Tests
16.7.4.1.2.
Blood
Tests
16.7.4.1.3.
Genetic
Testing
16.7.4.1.4.
Muscle biopsy
16.7.4.1.5.
CT Scan
16.7.4.1.6.
Electromyogram
(EMG)
16.7.4.1.7.
Electrocardiogram
(ECG or EKG)
16.7.4.1.8.
Others
16.7.4.2.
Kuwait Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Stages
16.7.4.2.1.
Early
Ambulatory
16.7.4.2.2.
Late
Ambulatory
16.7.4.2.3.
Early
Non-Ambulatory
16.7.4.2.4.
Late
Non-Ambulatory
16.7.4.3.
Kuwait Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Clinical
Phase
16.7.4.3.1.
Pre-clinical
16.7.4.3.2.
Phase 1
16.7.4.3.3.
Phase 2
16.7.4.3.4.
Phase 3
16.7.4.3.5.
Phase 4
16.7.4.3.6.
Approval
16.7.4.4.
Kuwait Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Age Group
16.7.4.4.1.
1 to 4
years
16.7.4.4.2.
5 to 7
years
16.7.4.4.3.
8 to 11
years
16.7.4.4.4.
12 to 19
years
16.7.4.4.5.
Adults
16.7.4.5.
Kuwait Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Treatment
16.7.4.5.1.
Medications
16.7.4.5.1.1. Corticosteroids
16.7.4.5.1.2. Heart medications
16.7.4.5.2.
Therapy
16.7.4.5.2.1. Range-of-motion and stretching exercises
16.7.4.5.2.2. Braces
16.7.4.5.2.3. Mobility aids
16.7.4.5.2.4. Breathing assistance
16.7.4.5.2.5. Others
16.7.4.5.3.
Surgery
16.7.4.5.4.
Diet and
Nutritional supplements
16.7.4.5.5.
Others
16.7.5. South Africa
16.7.5.1.
South
Africa Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Diagnosis
16.7.5.1.1.
Enzyme
Tests
16.7.5.1.2.
Blood
Tests
16.7.5.1.3.
Genetic
Testing
16.7.5.1.4.
Muscle biopsy
16.7.5.1.5.
CT Scan
16.7.5.1.6.
Electromyogram
(EMG)
16.7.5.1.7.
Electrocardiogram
(ECG or EKG)
16.7.5.1.8.
Others
16.7.5.2.
South
Africa Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Stages
16.7.5.2.1.
Early
Ambulatory
16.7.5.2.2.
Late
Ambulatory
16.7.5.2.3.
Early
Non-Ambulatory
16.7.5.2.4.
Late
Non-Ambulatory
16.7.5.3.
South
Africa Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Clinical Phase
16.7.5.3.1.
Pre-clinical
16.7.5.3.2.
Phase 1
16.7.5.3.3.
Phase 2
16.7.5.3.4.
Phase 3
16.7.5.3.5.
Phase 4
16.7.5.3.6.
Approval
16.7.5.4.
South
Africa Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Age Group
16.7.5.4.1.
1 to 4
years
16.7.5.4.2.
5 to 7
years
16.7.5.4.3.
8 to 11
years
16.7.5.4.4.
12 to 19
years
16.7.5.4.5.
Adults
16.7.5.5.
South
Africa Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts,
By Treatment
16.7.5.5.1.
Medications
16.7.5.5.1.1. Corticosteroids
16.7.5.5.1.2. Heart medications
16.7.5.5.2.
Therapy
16.7.5.5.2.1. Range-of-motion and stretching exercises
16.7.5.5.2.2. Braces
16.7.5.5.2.3. Mobility aids
16.7.5.5.2.4. Breathing assistance
16.7.5.5.2.5. Others
16.7.5.5.3.
Surgery
16.7.5.5.4.
Diet and
Nutritional supplements
16.7.5.5.5.
Others
16.7.6. Rest of Middle East & Africa
16.7.6.1.
Rest of
Middle East & Africa Duchenne Muscular Dystrophy (DMD) Market Revenue (US$
Mn) and Forecasts, By Diagnosis
16.7.6.1.1.
Enzyme
Tests
16.7.6.1.2.
Blood
Tests
16.7.6.1.3.
Genetic Testing
16.7.6.1.4.
Muscle biopsy
16.7.6.1.5.
CT Scan
16.7.6.1.6.
Electromyogram
(EMG)
16.7.6.1.7.
Electrocardiogram
(ECG or EKG)
16.7.6.1.8.
Others
16.7.6.2.
Rest of
Middle East & Africa Duchenne Muscular Dystrophy (DMD) Market Revenue (US$
Mn) and Forecasts, By Stages
16.7.6.2.1.
Early
Ambulatory
16.7.6.2.2.
Late
Ambulatory
16.7.6.2.3.
Early
Non-Ambulatory
16.7.6.2.4.
Late
Non-Ambulatory
16.7.6.3.
Rest of
Middle East & Africa Duchenne Muscular Dystrophy (DMD) Market Revenue (US$
Mn) and Forecasts, By Clinical Phase
16.7.6.3.1.
Pre-clinical
16.7.6.3.2.
Phase 1
16.7.6.3.3.
Phase 2
16.7.6.3.4.
Phase 3
16.7.6.3.5.
Phase 4
16.7.6.3.6.
Approval
16.7.6.4.
Rest of
Middle East & Africa Duchenne Muscular Dystrophy (DMD) Market Revenue (US$
Mn) and Forecasts, By Age Group
16.7.6.4.1.
1 to 4
years
16.7.6.4.2.
5 to 7
years
16.7.6.4.3.
8 to 11
years
16.7.6.4.4.
12 to 19
years
16.7.6.4.5.
Adults
16.7.6.5.
Rest of
Middle East & Africa Duchenne Muscular Dystrophy (DMD) Market Revenue (US$
Mn) and Forecasts, By Treatment
16.7.6.5.1.
Medications
16.7.6.5.1.1. Corticosteroids
16.7.6.5.1.2. Heart medications
16.7.6.5.2.
Therapy
16.7.6.5.2.1. Range-of-motion and stretching exercises
16.7.6.5.2.2. Braces
16.7.6.5.2.3. Mobility aids
16.7.6.5.2.4. Breathing assistance
16.7.6.5.2.5. Others
16.7.6.5.3.
Surgery
16.7.6.5.4.
Diet and
Nutritional supplements
16.7.6.5.5.
Others
16.8. Key Segment for Channeling Investments
16.8.1. By Country
16.8.2. By Diagnosis
16.8.3. By Stages
16.8.4. By Clinical Phase
16.8.5. By Age Group
16.8.6. By Treatment
17. Latin America Duchenne Muscular Dystrophy
(DMD) Market Analysis and Forecasts, 2022 - 2030
17.1. Overview
17.1.1. Latin America Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn)
17.2. Latin America Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn) and Forecasts, By Diagnosis
17.2.1. Enzyme Tests
17.2.2. Blood Tests
17.2.3. Genetic Testing
17.2.4. Muscle
biopsy
17.2.5. CT Scan
17.2.6. Electromyogram (EMG)
17.2.7. Electrocardiogram (ECG or EKG)
17.2.8. Others
17.3. Latin America Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn) and Forecasts, By Stages
17.3.1. Early Ambulatory
17.3.2. Late Ambulatory
17.3.3. Early Non-Ambulatory
17.3.4. Late Non-Ambulatory
17.4. Latin America Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn) and Forecasts, By Clinical Phase
17.4.1. Pre-clinical
17.4.2. Phase 1
17.4.3. Phase 2
17.4.4. Phase 3
17.4.5. Phase 4
17.4.6. Approval
17.5. Latin America Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn) and Forecasts, By Age Group
17.5.1. 1 to 4 years
17.5.2. 5 to 7 years
17.5.3. 8 to 11 years
17.5.4. 12 to 19 years
17.5.5. Adults
17.6. Latin America Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn) and Forecasts, By Treatment
17.6.1. Medications
17.6.1.1.
Corticosteroids
17.6.1.2.
Heart
medications
17.6.2. Therapy
17.6.2.1.
Range-of-motion
and stretching exercises
17.6.2.2.
Braces
17.6.2.3.
Mobility
aids
17.6.2.4.
Breathing
assistance
17.6.2.5.
Others
17.6.3. Surgery
17.6.4. Diet and Nutritional supplements
17.6.5. Others
17.7. Latin America Duchenne Muscular Dystrophy
(DMD) Market Revenue (US$ Mn) and Forecasts, By Country
17.7.1. Brazil
17.7.1.1.
Brazil Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Diagnosis
17.7.1.1.1.
Enzyme
Tests
17.7.1.1.2.
Blood
Tests
17.7.1.1.3.
Genetic
Testing
17.7.1.1.4.
Muscle biopsy
17.7.1.1.5.
CT Scan
17.7.1.1.6.
Electromyogram
(EMG)
17.7.1.1.7.
Electrocardiogram
(ECG or EKG)
17.7.1.1.8.
Others
17.7.1.2.
Brazil Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Stages
17.7.1.2.1.
Early
Ambulatory
17.7.1.2.2.
Late
Ambulatory
17.7.1.2.3.
Early
Non-Ambulatory
17.7.1.2.4.
Late
Non-Ambulatory
17.7.1.3.
Brazil Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Clinical
Phase
17.7.1.3.1.
Pre-clinical
17.7.1.3.2.
Phase 1
17.7.1.3.3.
Phase 2
17.7.1.3.4.
Phase 3
17.7.1.3.5.
Phase 4
17.7.1.3.6.
Approval
17.7.1.4.
Brazil Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Age Group
17.7.1.4.1.
1 to 4
years
17.7.1.4.2.
5 to 7
years
17.7.1.4.3.
8 to 11
years
17.7.1.4.4.
12 to 19
years
17.7.1.4.5.
Adults
17.7.1.5.
Brazil Duchenne
Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Treatment
17.7.1.5.1.
Medications
17.7.1.5.1.1. Corticosteroids
17.7.1.5.1.2. Heart medications
17.7.1.5.2.
Therapy
17.7.1.5.2.1. Range-of-motion and stretching exercises
17.7.1.5.2.2. Braces
17.7.1.5.2.3. Mobility aids
17.7.1.5.2.4. Breathing assistance
17.7.1.5.2.5. Others
17.7.1.5.3.
Surgery
17.7.1.5.4.
Diet and
Nutritional supplements
17.7.1.5.5.
Others
17.7.2. Argentina
17.7.2.1.
Argentina
Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By
Diagnosis
17.7.2.1.1.
Enzyme
Tests
17.7.2.1.2.
Blood
Tests
17.7.2.1.3.
Genetic
Testing
17.7.2.1.4.
Muscle biopsy
17.7.2.1.5.
CT Scan
17.7.2.1.6.
Electromyogram
(EMG)
17.7.2.1.7.
Electrocardiogram
(ECG or EKG)
17.7.2.1.8.
Others
17.7.2.2.
Argentina
Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By
Stages
17.7.2.2.1.
Early
Ambulatory
17.7.2.2.2.
Late
Ambulatory
17.7.2.2.3.
Early
Non-Ambulatory
17.7.2.2.4.
Late
Non-Ambulatory
17.7.2.3.
Argentina
Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By
Clinical Phase
17.7.2.3.1.
Pre-clinical
17.7.2.3.2.
Phase 1
17.7.2.3.3.
Phase 2
17.7.2.3.4.
Phase 3
17.7.2.3.5.
Phase 4
17.7.2.3.6.
Approval
17.7.2.4.
Argentina
Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By Age
Group
17.7.2.4.1.
1 to 4
years
17.7.2.4.2.
5 to 7
years
17.7.2.4.3.
8 to 11
years
17.7.2.4.4.
12 to 19
years
17.7.2.4.5.
Adults
17.7.2.5.
Argentina
Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and Forecasts, By
Treatment
17.7.2.5.1.
Medications
17.7.2.5.1.1. Corticosteroids
17.7.2.5.1.2. Heart medications
17.7.2.5.2.
Therapy
17.7.2.5.2.1. Range-of-motion and stretching exercises
17.7.2.5.2.2. Braces
17.7.2.5.2.3. Mobility aids
17.7.2.5.2.4. Breathing assistance
17.7.2.5.2.5. Others
17.7.2.5.3.
Surgery
17.7.2.5.4.
Diet and
Nutritional supplements
17.7.2.5.5.
Others
17.7.3. Rest of Latin America
17.7.3.1.
Rest of
Latin America Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Diagnosis
17.7.3.1.1.
Enzyme
Tests
17.7.3.1.2.
Blood
Tests
17.7.3.1.3.
Genetic
Testing
17.7.3.1.4.
Muscle biopsy
17.7.3.1.5.
CT Scan
17.7.3.1.6.
Electromyogram
(EMG)
17.7.3.1.7.
Electrocardiogram
(ECG or EKG)
17.7.3.1.8.
Others
17.7.3.2.
Rest of
Latin America Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Stages
17.7.3.2.1.
Early
Ambulatory
17.7.3.2.2.
Late
Ambulatory
17.7.3.2.3.
Early
Non-Ambulatory
17.7.3.2.4.
Late
Non-Ambulatory
17.7.3.3.
Rest of
Latin America Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Clinical Phase
17.7.3.3.1.
Pre-clinical
17.7.3.3.2.
Phase 1
17.7.3.3.3.
Phase 2
17.7.3.3.4.
Phase 3
17.7.3.3.5.
Phase 4
17.7.3.3.6.
Approval
17.7.3.4.
Rest of
Latin America Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Age Group
17.7.3.4.1.
1 to 4
years
17.7.3.4.2.
5 to 7
years
17.7.3.4.3.
8 to 11
years
17.7.3.4.4.
12 to 19
years
17.7.3.4.5.
Adults
17.7.3.5.
Rest of
Latin America Duchenne Muscular Dystrophy (DMD) Market Revenue (US$ Mn) and
Forecasts, By Treatment
17.7.3.5.1.
Medications
17.7.3.5.1.1. Corticosteroids
17.7.3.5.1.2. Heart medications
17.7.3.5.2.
Therapy
17.7.3.5.2.1. Range-of-motion and stretching exercises
17.7.3.5.2.2. Braces
17.7.3.5.2.3. Mobility aids
17.7.3.5.2.4. Breathing assistance
17.7.3.5.2.5. Others
17.7.3.5.3.
Surgery
17.7.3.5.4.
Diet and
Nutritional supplements
17.7.3.5.5.
Others
17.8. Key Segment for Channeling Investments
17.8.1. By Country
17.8.2. By Diagnosis
17.8.3. By Stages
17.8.4. By Clinical Phase
17.8.5. By Age Group
17.8.6. By Treatment
18. Competitive Benchmarking
18.1. Brand Benchmarking
18.2. Market Share Analysis, 2021
18.3. Global Presence and Growth Strategies
18.3.1. Mergers and Acquisitions
18.3.2. Product Launches
18.3.3. Investments Trends
18.3.4. R&D Initiatives
19. Player Profiles
19.1. Antisense Therapeutics Limited
19.1.1. Company Details
19.1.2. Company Overview
19.1.3. Product Offerings
19.1.4. Key Developments
19.1.5. Financial Analysis
19.1.6. SWOT Analysis
19.1.7. Business Strategies
19.2. Capricor Therapeutics
19.2.1. Company Details
19.2.2. Company Overview
19.2.3. Product Offerings
19.2.4. Key Developments
19.2.5. Financial Analysis
19.2.6. SWOT Analysis
19.2.7. Business Strategies
19.3. DAIICHI SANKYO COMPANY, LIMITED
19.3.1. Company Details
19.3.2. Company Overview
19.3.3. Product Offerings
19.3.4. Key Developments
19.3.5. Financial Analysis
19.3.6. SWOT Analysis
19.3.7. Business Strategies
19.4. Dyne Therapeutics
19.4.1. Company Details
19.4.2. Company Overview
19.4.3. Product Offerings
19.4.4. Key Developments
19.4.5. Financial Analysis
19.4.6. SWOT Analysis
19.4.7. Business Strategies
19.5. Edgewise Therapeutics
19.5.1. Company Details
19.5.2. Company Overview
19.5.3. Product Offerings
19.5.4. Key Developments
19.5.5. Financial Analysis
19.5.6. SWOT Analysis
19.5.7. Business Strategies
19.6. ENCELL Co, Ltd
19.6.1. Company Details
19.6.2. Company Overview
19.6.3. Product Offerings
19.6.4. Key Developments
19.6.5. Financial Analysis
19.6.6. SWOT Analysis
19.6.7. Business Strategies
19.7. Entrada Therapeutics, Inc
19.7.1. Company Details
19.7.2. Company Overview
19.7.3. Product Offerings
19.7.4. Key Developments
19.7.5. Financial Analysis
19.7.6. SWOT Analysis
19.7.7. Business Strategies
19.8. FibroGen, Inc.
19.8.1. Company Details
19.8.2. Company Overview
19.8.3. Product Offerings
19.8.4. Key Developments
19.8.5. Financial Analysis
19.8.6. SWOT Analysis
19.8.7. Business Strategies
19.9. ITALFARMACO S.p.A
19.9.1. Company Details
19.9.2. Company Overview
19.9.3. Product Offerings
19.9.4. Key Developments
19.9.5. Financial Analysis
19.9.6. SWOT Analysis
19.9.7. Business Strategies
19.10. NIPPON SHINYAKU CO. LTD
19.10.1. Company Details
19.10.2. Company Overview
19.10.3. Product Offerings
19.10.4. Key Developments
19.10.5. Financial Analysis
19.10.6. SWOT Analysis
19.10.7. Business Strategies
19.11. PepGen Inc.
19.11.1. Company Details
19.11.2. Company Overview
19.11.3. Product Offerings
19.11.4. Key Developments
19.11.5. Financial Analysis
19.11.6. SWOT Analysis
19.11.7. Business Strategies
19.12. Pfizer Inc.
19.12.1. Company Details
19.12.2. Company Overview
19.12.3. Product Offerings
19.12.4. Key Developments
19.12.5. Financial Analysis
19.12.6. SWOT Analysis
19.12.7. Business Strategies
19.13. SANTHERA PHARMACEUTICALS
19.13.1. Company Details
19.13.2. Company Overview
19.13.3. Product Offerings
19.13.4. Key Developments
19.13.5. Financial Analysis
19.13.6. SWOT Analysis
19.13.7. Business Strategies
19.14. Sarepta Therapeutics, Inc.
19.14.1. Company Details
19.14.2. Company Overview
19.14.3. Product Offerings
19.14.4. Key Developments
19.14.5. Financial Analysis
19.14.6. SWOT Analysis
19.14.7. Business Strategies
19.15. Solid Biosciences Inc
19.15.1. Company Details
19.15.2. Company Overview
19.15.3. Product Offerings
19.15.4. Key Developments
19.15.5. Financial Analysis
19.15.6. SWOT Analysis
19.15.7. Business Strategies
19.16. Taiho Pharmaceuticals Co. Ltd
19.16.1. Company Details
19.16.2. Company Overview
19.16.3. Product Offerings
19.16.4. Key Developments
19.16.5. Financial Analysis
19.16.6. SWOT Analysis
19.16.7. Business Strategies
19.17. Wave Life Sciences
19.17.1. Company Details
19.17.2. Company Overview
19.17.3. Product Offerings
19.17.4. Key Developments
19.17.5. Financial Analysis
19.17.6. SWOT Analysis
19.17.7. Business Strategies
19.18. Other Market Participants
20. Key Findings
Note: This ToC is tentative
and can be changed according to the research study conducted during the course
of report completion.
**Exclusive for Multi-User
and Enterprise User.
At Absolute Markets Insights, we are engaged in building both global as well as country specific reports. As a result, the approach taken for deriving the estimation and forecast for a specific country is a bit unique and different in comparison to the global research studies. In this case, we not only study the concerned market factors & trends prevailing in a particular country (from secondary research) but we also tend to calculate the actual market size & forecast from the revenue generated from the market participants involved in manufacturing or distributing the any concerned product. These companies can also be service providers. For analyzing any country specifically, we do consider the growth factors prevailing under the states/cities/county for the same. For instance, if we are analyzing an industry specific to United States, we primarily need to study about the states present under the same(where the product/service has the highest growth). Similar analysis will be followed by other countries. Our scope of the report changes with different markets.
Our research study is mainly implement through a mix of both secondary and primary research. Various sources such as industry magazines, trade journals, and government websites and trade associations are reviewed for gathering precise data. Primary interviews are conducted to validate the market size derived from secondary research. Industry experts, major manufacturers and distributors are contacted for further validation purpose on the current market penetration and growth trends.
Prominent participants in our primary research process include:
- Key Opinion Leaders namely the CEOs, CSOs, VPs, purchasing managers, amongst others
- Research and development participants, distributors/suppliers and subject matter experts
Secondary Research includes data extracted from paid data sources:
- Reuters
- Factiva
- Bloomberg
- One Source
- Hoovers
Research Methodology
Key Inclusions

Why Absolute Markets Insights?
An effective strategy is the entity that influences a business to stand out of the crowd. An organization with a phenomenal strategy for success dependably has the edge over the rivals in the market. It offers the organizations a head start in planning their strategy. Absolute Market Insights is the new initiation in the industry that will furnish you with the lead your business needs. Absolute Market Insights is the best destination for your business intelligence and analytical solutions; essentially because our qualitative and quantitative sources of information are competent to give one-stop solutions. We inventively combine qualitative and quantitative research in accurate proportions to have the best report, which not only gives the most recent insights but also assists you to grow.

