Global Post-marketing Pharmacovigilance and Medical Information Market By Offering( Case Processing & Management, Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.), Audits and Inspection Support Services, Signal Detection & Risk Management, Global & Local Literature Screening, Consulting Services, Medical Writing & Publishing, Regulatory Publishing, Clinical Studies, Educational, Others) Others) By Organization Size (Small and Medium Organizations, Large Organizations) By End Users (Pharmaceutical Companies, Research Organizations, Others) By Region (North America (U.S., Canada, Mexico, Rest Of North America), Europe (France, The UK, Spain, Germany, Italy, Denmark, Finland, Iceland, Sweden, Norway, Belgium, The Netherlands, Luxembourg, Rest of Europe), Asia Pacific (China, Japan, India, New Zealand, Australia, South Korea, Southeast Asia, Indonesia, Thailand, Malaysia, Singapore, Rest of Southeast Asia, Rest of Asia Pacific, Middle East & Africa (Saudi Arabia, UAE, Egypt, Kuwait, South Africa, Rest of Middle East & Africa) Latin America (Brazil, Argentina, Rest of Latin America)) - Global Insights, Growth, Size, Comparative Analysis, Trends and Forecast, 2023 – 2031
Industry Trends
In terms of revenue, global post-marketing pharmacovigilance and medical information market was valued at US$ 5.3 Bn in 2022 growing at a CAGR of 16.2% over the forecast period (2023 – 2031).
Health care providers, producers, regulators, and the general public can all monitor a product's performance over the course of its lifespan in the open market through post-market surveillance, which is a passive, multivariate procedure based on performance. It is crucial that regulators and producers agree on shared safety surveillance objectives while keeping consumers in mind. In order to taking proactive measures to guarantee appropriate documentation and the filing of reports of adverse events to the authorities, manufacturers are abiding by the laws and regulations. Manufacturers can develop a corporate post-market surveillance programme to keep an eye on product safety while adhering to best practises and legal requirements. Thus, to guarantee that ethical procedures and impartial information is being utilised in promotional materials, enough resources and experience is also required. Pharmacovigilance firms oversee crucial tasks at every stage of clinical research, from the planning, execution, and follow-up of clinical trials through the completion of studies with data analysis, report writing, approval, and registration. Demands for drug safety and pharmacovigilance are increasingly developing in tandem with expanding medication usage along with the need for ongoing drug monitoring. Pharmaceutical businesses get data on medication safety profiles from pharmacovigilance analyses carried out during clinical trial phases, which may be utilised for either additional research and development or regulatory filings. Over a drug's entire history, the actual risk-benefit ratio is closely reviewed, enhanced, and updated as necessary. Pharmaceutical businesses get data on medication safety profiles from pharmacovigilance analyses carried out during clinical trial phases, which may be utilised for either additional research and development or regulatory filings. Over a drug's entire history, the actual risk-benefit ratio is closely reviewed, enhanced, and updated as necessary.
The global post-marketing pharmacovigilance and medical information market experienced tremendous demand during covid-19 pandemic. It was an amazing accomplishment that viable vaccinations were available in less than a year, thanks to the extraordinary degree of innovation across several partners. Faster vaccine research and licensing made it possible to concentrate a greater emphasis on the safety monitoring of vaccines sold. In contrary to other licenced therapies, one of the major obstacles to accelerating the rollout of vaccinations is the universal immunisation of people who are usually healthy. With their efforts supporting intricate pharmacovigilance procedures for some of the authorised COVID-19 vaccines and treatments, various firms have aided in the pandemic response.
Global Post-Marketing Pharmacovigilance and Medical Information Market Revenue & Forecast, (US$ Million), 2015 – 2031
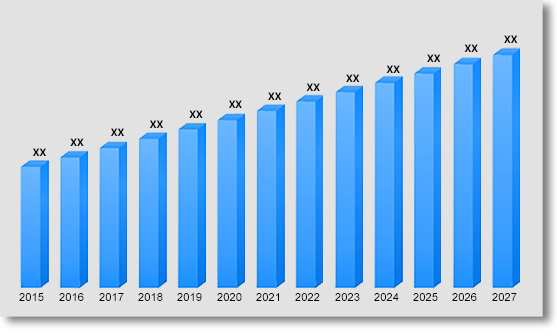
The importance of regulation in post-market clinical data has grown over the past few years. In order to offer early notice of quality issues and mandate input into the manufacturers' corrective and preventative action procedures, regulatory bodies have imposed the necessity for a feedback system. Drug use and production have greatly increased in recent years. Patients run the risk of having unintended and occasionally hazardous adverse effects while using medications for longer lengths of time than recommended. According to the WHO, drug errors result in one fatality for every million people. With a population of 447 million people in the EU, this would equate to 163000 fatalities each year. By studying the data of Spain, Germany and the U.S. in between 60,000 and 131,000 annual fatalities in Europe were recorded owing to drug related errors. As a result, it is necessary to alter the present procedures to enable prompt contact between medical professionals and the Food and Drug Administration. Thus, the global post-marketing pharmacovigilance and medical information market is projected to grow in the near future as it is crucial for drug evaluation and the avoidance of adverse effects.
Regional Outlook
North America had the highest share in the global post-marketing pharmacovigilance and medical information market in 2022. The market is primarily driven by rising demand for new medication development and instances of adverse drug reporting. The expansion of this local business is also anticipated to be fuelled by increasing healthcare spending, supportive infrastructure, and the implementation of Electronic Health Records (EHR). Additionally, North American countries are intensifying efforts towards monitoring the safety of medicines, while also striving towards faster development of generic medicines.
Competitive Landscape
The report provides both, qualitative and quantitative research of global post-marketing pharmacovigilance and medical information market, as well as provides comprehensive insights and development methods adopted by the key contenders. The report also offers extensive research on the key players in this market and details on the competitiveness of these players. Key business strategies such as mergers and acquisitions (M&A), affiliations, collaborations, and contracts adopted by these major market participants are also recognized and analysed in the report. For each company, the report studies their global presence, competitors, service offerings and specification amongst others.
Global Post-marketing Pharmacovigilance and Medical Information Market Competitors
- A2 Healthcare Corp.
- Accenture
- Clario
- Cognizant
- Genpact
- ICON plc
- IQVIA
- Labcorp Drug Development
- Parexel International Corporation
- PharSafer
- PPD Inc.
- Qvigilance
- Syneos Health
- Universal Medica Group
- Wipro Limited
- Other Market Participants
Global Post-marketing Pharmacovigilance and Medical Information Market:
By Offering
- Case Processing & Management
- Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
- Audits and Inspection Support Services
- Signal Detection & Risk Management
- Global & Local Literature Screening
- Consulting Services
- Medical Writing & Publishing
- Regulatory Publishing
- Clinical Studies
- Educational
- Others
- Others
By Organization Size
- Small and Medium Organizations
- Large Organizations
By End Users
- Pharmaceutical Companies
- Research Organizations
- Others
By Region
- North America (U.S., Canada, Mexico, Rest of North America)
- Europe (France, The UK, Spain, Germany, Italy, Nordic Countries (Denmark, Finland, Iceland, Sweden, Norway), Benelux Union (Belgium, The Netherlands, Luxembourg), Rest of Europe
- Asia Pacific (China, Japan, India, New Zealand, Australia, South Korea, Southeast Asia (Indonesia, Thailand, Malaysia, Singapore, Rest of Southeast Asia), Rest of Asia Pacific
- Middle East & Africa (Saudi Arabia, UAE, Egypt, Kuwait, South Africa, Rest of Middle East & Africa)
- Latin America (Brazil, Argentina, Rest of Latin America)
Table of Contents
1. Market Scope
1.1. Market
Segmentation
1.2. Years
Considered
1.2.1. Historic
Years: 2015 - 2021
1.2.2. Base
Year: 2022
1.2.3. Forecast
Years: 2023 – 2031
2. Key Target Audiences
3. Research Methodology
3.1. Primary
Research
3.1.1. Research
Questionnaire
3.1.2. Global
Percentage Breakdown
3.1.3. Primary
Interviews: Key Opinion Leaders (KOLs)
3.2. Secondary
Research
3.2.1. Paid
Databases
3.2.2. Secondary
Sources
3.3. Market
Size Estimates
3.3.1. Top-Down
Approach
3.3.2. Bottom-Up
Approach
3.4. Data
Triangulation Methodology
3.5. Research
Assumptions
4. Recommendations and Insights from AMI’s Perspective**
5. Holistic Overview of Post-marketing Pharmacovigilance and
Medical Information Market
6. Market Synopsis:
Post-marketing Pharmacovigilance and Medical Information Market
7. Post-marketing Pharmacovigilance and Medical Information
Market Analysis: Qualitative Perspective
7.1. Introduction
7.1.1. Product
Definition
7.1.2. Industry
Development
7.2. Market
Dynamics
7.2.1. Drivers
7.2.2. Restraints
7.2.3. Opportunities
7.3. Trends in
Post-marketing Pharmacovigilance and Medical Information Market
7.4. Market
Determinants Radar Chart
7.5. Macro-Economic
and Micro-Economic Indicators: Post-marketing Pharmacovigilance and Medical
Information Market
7.6. Porter’s
Five Force Analysis
7.7. Impact of
Covid-19 on Post-marketing Pharmacovigilance and Medical Information Market
8. Global Post-marketing Pharmacovigilance and Medical
Information Market Analysis and Forecasts, 2023 - 2031
8.1. Overview
8.1.1. Global
Post-marketing Pharmacovigilance and Medical Information Market Revenue (US$
Mn)
8.2. Global
Post-marketing Pharmacovigilance and Medical Information Market Revenue (US$
Mn) and Forecasts, By Offering
8.2.1. Case
Processing and Management
8.2.1.1. Definition
8.2.1.2. Market Estimation and Penetration, 2015 - 2022
8.2.1.3. Market Forecast, 2023 - 2031
8.2.1.4. Compound Annual Growth Rate (CAGR)
8.2.1.5. Regional Bifurcation
8.2.1.5.1. North America
8.2.1.5.1.1. Market
Estimation, 2015 - 2022
8.2.1.5.1.2. Market
Forecast, 2023 - 2031
8.2.1.5.2. Europe
8.2.1.5.2.1. Market
Estimation, 2015 - 2022
8.2.1.5.2.2. Market
Forecast, 2023 - 2031
8.2.1.5.3. Asia Pacific
8.2.1.5.3.1. Market
Estimation, 2015 - 2022
8.2.1.5.3.2. Market
Forecast, 2023 - 2031
8.2.1.5.4. Middle East and Africa
8.2.1.5.4.1. Market
Estimation, 2015 - 2022
8.2.1.5.4.2. Market
Forecast, 2023 - 2031
8.2.1.5.5. Latin America
8.2.1.5.5.1. Market
Estimation, 2015 - 2022
8.2.1.5.5.2. Market
Forecast, 2023 - 2031
8.2.2. Aggregate
Reporting (Including PSURs, PBRERs, DSURs, Etc.).
8.2.2.1. Definition
8.2.2.2. Market Estimation and Penetration, 2015 - 2022
8.2.2.3. Market Forecast, 2023 - 2031
8.2.2.4. Compound Annual Growth Rate (CAGR)
8.2.2.5. Regional Bifurcation
8.2.2.5.1. North America
8.2.2.5.1.1. Market
Estimation, 2015 - 2022
8.2.2.5.1.2. Market
Forecast, 2023 - 2031
8.2.2.5.2. Europe
8.2.2.5.2.1. Market
Estimation, 2015 - 2022
8.2.2.5.2.2. Market
Forecast, 2023 - 2031
8.2.2.5.3. Asia Pacific
8.2.2.5.3.1. Market
Estimation, 2015 - 2022
8.2.2.5.3.2. Market
Forecast, 2023 - 2031
8.2.2.5.4. Middle East and Africa
8.2.2.5.4.1. Market
Estimation, 2015 - 2022
8.2.2.5.4.2. Market
Forecast, 2023 - 2031
8.2.2.5.5. Latin America
8.2.2.5.5.1. Market
Estimation, 2015 - 2022
8.2.2.5.5.2. Market
Forecast, 2023 - 2031
8.2.3. Audits
and Inspection Support Services
8.2.3.1. Definition
8.2.3.2. Market Estimation and Penetration, 2015 - 2022
8.2.3.3. Market Forecast, 2023 - 2031
8.2.3.4. Compound Annual Growth Rate (CAGR)
8.2.3.5. Regional Bifurcation
8.2.3.5.1. North America
8.2.3.5.1.1. Market
Estimation, 2015 - 2022
8.2.3.5.1.2. Market
Forecast, 2023 - 2031
8.2.3.5.2. Europe
8.2.3.5.2.1. Market
Estimation, 2015 - 2022
8.2.3.5.2.2. Market
Forecast, 2023 - 2031
8.2.3.5.3. Asia Pacific
8.2.3.5.3.1. Market
Estimation, 2015 - 2022
8.2.3.5.3.2. Market
Forecast, 2023 - 2031
8.2.3.5.4. Middle East and Africa
8.2.3.5.4.1. Market
Estimation, 2015 - 2022
8.2.3.5.4.2. Market
Forecast, 2023 - 2031
8.2.3.5.5. Latin America
8.2.3.5.5.1. Market
Estimation, 2015 - 2022
8.2.3.5.5.2. Market
Forecast, 2023 - 2031
8.2.4. Signal
Detection and Risk Management
8.2.4.1. Definition
8.2.4.2. Market Estimation and Penetration, 2015 - 2022
8.2.4.3. Market Forecast, 2023 - 2031
8.2.4.4. Compound Annual Growth Rate (CAGR)
8.2.4.5. Regional Bifurcation
8.2.4.5.1. North America
8.2.4.5.1.1. Market
Estimation, 2015 - 2022
8.2.4.5.1.2. Market
Forecast, 2023 - 2031
8.2.4.5.2. Europe
8.2.4.5.2.1. Market
Estimation, 2015 - 2022
8.2.4.5.2.2. Market
Forecast, 2023 - 2031
8.2.4.5.3. Asia Pacific
8.2.4.5.3.1. Market
Estimation, 2015 - 2022
8.2.4.5.3.2. Market
Forecast, 2023 - 2031
8.2.4.5.4. Middle East and Africa
8.2.4.5.4.1. Market
Estimation, 2015 - 2022
8.2.4.5.4.2. Market
Forecast, 2023 - 2031
8.2.4.5.5. Latin America
8.2.4.5.5.1. Market
Estimation, 2015 - 2022
8.2.4.5.5.2. Market
Forecast, 2023 - 2031
8.2.5. Global
and Local Literature Screening
8.2.5.1. Definition
8.2.5.2. Market Estimation and Penetration, 2015 - 2022
8.2.5.3. Market Forecast, 2023 - 2031
8.2.5.4. Compound Annual Growth Rate (CAGR)
8.2.5.5. Regional Bifurcation
8.2.5.5.1. North America
8.2.5.5.1.1. Market
Estimation, 2015 - 2022
8.2.5.5.1.2. Market
Forecast, 2023 - 2031
8.2.5.5.2. Europe
8.2.5.5.2.1. Market
Estimation, 2015 - 2022
8.2.5.5.2.2. Market
Forecast, 2023 - 2031
8.2.5.5.3. Asia Pacific
8.2.5.5.3.1. Market
Estimation, 2015 - 2022
8.2.5.5.3.2. Market
Forecast, 2023 - 2031
8.2.5.5.4. Middle East and Africa
8.2.5.5.4.1. Market
Estimation, 2015 - 2022
8.2.5.5.4.2. Market
Forecast, 2023 - 2031
8.2.5.5.5. Latin America
8.2.5.5.5.1. Market
Estimation, 2015 - 2022
8.2.5.5.5.2. Market
Forecast, 2023 - 2031
8.2.6. Consulting
Services
8.2.6.1. Definition
8.2.6.2. Market Estimation and Penetration, 2015 - 2022
8.2.6.3. Market Forecast, 2023 - 2031
8.2.6.4. Compound Annual Growth Rate (CAGR)
8.2.6.5. Regional Bifurcation
8.2.6.5.1. North America
8.2.6.5.1.1. Market
Estimation, 2015 - 2022
8.2.6.5.1.2. Market
Forecast, 2023 - 2031
8.2.6.5.2. Europe
8.2.6.5.2.1. Market
Estimation, 2015 - 2022
8.2.6.5.2.2. Market
Forecast, 2023 - 2031
8.2.6.5.3. Asia Pacific
8.2.6.5.3.1. Market
Estimation, 2015 - 2022
8.2.6.5.3.2. Market
Forecast, 2023 - 2031
8.2.6.5.4. Middle East and Africa
8.2.6.5.4.1. Market
Estimation, 2015 - 2022
8.2.6.5.4.2. Market
Forecast, 2023 - 2031
8.2.6.5.5. Latin America
8.2.6.5.5.1. Market
Estimation, 2015 - 2022
8.2.6.5.5.2. Market
Forecast, 2023 - 2031
8.2.7. Medical
Writing and Publishing (Definition, Market Estimation and Penetration, 2015 -
2022, Market Estimation (2015 - 2022), Market Forecast (2023 - 2031), Compound
Annual Growth Rate (CAGR), Regional Bifurcation (North America, Europe, Asia
Pacific, Middle East and Africa, Latin America) and Information on Regulatory
Publishing, Clinical Studies, Educational, Others)
8.2.7.1. Regulatory Publishing
8.2.7.2. Clinical Studies
8.2.7.3. Educational
8.2.7.4. Others
8.2.8. Others
8.2.8.1. Definition
8.2.8.2. Market Estimation and Penetration, 2015 - 2022
8.2.8.3. Market Forecast, 2023 - 2031
8.2.8.4. Compound Annual Growth Rate (CAGR)
8.2.8.5. Regional Bifurcation
8.2.8.5.1. North America
8.2.8.5.1.1. Market
Estimation, 2015 - 2022
8.2.8.5.1.2. Market
Forecast, 2023 - 2031
8.2.8.5.2. Europe
8.2.8.5.2.1. Market
Estimation, 2015 - 2022
8.2.8.5.2.2. Market
Forecast, 2023 - 2031
8.2.8.5.3. Asia Pacific
8.2.8.5.3.1. Market
Estimation, 2015 - 2022
8.2.8.5.3.2. Market
Forecast, 2023 - 2031
8.2.8.5.4. Middle East and Africa
8.2.8.5.4.1. Market
Estimation, 2015 - 2022
8.2.8.5.4.2. Market
Forecast, 2023 - 2031
8.2.8.5.5. Latin America
8.2.8.5.5.1. Market
Estimation, 2015 - 2022
8.2.8.5.5.2. Market
Forecast, 2023 - 2031
8.3. Key
Segment for Channeling Investments
8.3.1. By Offering
9. Global Post-marketing Pharmacovigilance and Medical
Information Market Analysis and Forecasts, 2023 - 2031
9.1. Overview
9.2. Global
Post-marketing Pharmacovigilance and Medical Information Market Revenue (US$
Mn) and Forecasts, By Organization Size
9.2.1. Small
and Medium Organizations
9.2.1.1. Definition
9.2.1.2. Market Estimation and Penetration, 2015 - 2022
9.2.1.3. Market Forecast, 2023 - 2031
9.2.1.4. Compound Annual Growth Rate (CAGR)
9.2.1.5. Regional Bifurcation
9.2.1.5.1. North America
9.2.1.5.1.1. Market
Estimation, 2015 - 2022
9.2.1.5.1.2. Market
Forecast, 2023 - 2031
9.2.1.5.2. Europe
9.2.1.5.2.1. Market
Estimation, 2015 - 2022
9.2.1.5.2.2. Market
Forecast, 2023 - 2031
9.2.1.5.3. Asia Pacific
9.2.1.5.3.1. Market
Estimation, 2015 - 2022
9.2.1.5.3.2. Market
Forecast, 2023 - 2031
9.2.1.5.4. Middle East and Africa
9.2.1.5.4.1. Market
Estimation, 2015 - 2022
9.2.1.5.4.2. Market
Forecast, 2023 - 2031
9.2.1.5.5. Latin America
9.2.1.5.5.1. Market
Estimation, 2015 - 2022
9.2.1.5.5.2. Market
Forecast, 2023 - 2031
9.2.2. Large
Organizations
9.2.2.1. Definition
9.2.2.2. Market Estimation and Penetration, 2015 - 2022
9.2.2.3. Market Forecast, 2023 - 2031
9.2.2.4. Compound Annual Growth Rate (CAGR)
9.2.2.5. Regional Bifurcation
9.2.2.5.1. North America
9.2.2.5.1.1. Market
Estimation, 2015 - 2022
9.2.2.5.1.2. Market
Forecast, 2023 - 2031
9.2.2.5.2. Europe
9.2.2.5.2.1. Market
Estimation, 2015 - 2022
9.2.2.5.2.2. Market
Forecast, 2023 - 2031
9.2.2.5.3. Asia Pacific
9.2.2.5.3.1. Market
Estimation, 2015 - 2022
9.2.2.5.3.2. Market
Forecast, 2023 - 2031
9.2.2.5.4. Middle East and Africa
9.2.2.5.4.1. Market
Estimation, 2015 - 2022
9.2.2.5.4.2. Market
Forecast, 2023 - 2031
9.2.2.5.5. Latin America
9.2.2.5.5.1. Market
Estimation, 2015 - 2022
9.2.2.5.5.2. Market
Forecast, 2023 - 2031
9.3. Key
Segment for Channeling Investments
9.3.1. By
Organization Size
10. Global Post-marketing Pharmacovigilance and Medical
Information Market Analysis and Forecasts, 2023 - 2031
10.1. Overview
10.2. Global
Post-marketing Pharmacovigilance and Medical Information Market Revenue (US$
Mn) and Forecasts, By End Users
10.2.1. Pharmaceutical
Companies
10.2.1.1. Definition
10.2.1.2. Market Estimation and Penetration, 2015 - 2022
10.2.1.3. Market Forecast, 2023 - 2031
10.2.1.4. Compound Annual Growth Rate (CAGR)
10.2.1.5. Regional Bifurcation
10.2.1.5.1. North America
10.2.1.5.1.1. Market
Estimation, 2015 - 2022
10.2.1.5.1.2. Market
Forecast, 2023 - 2031
10.2.1.5.2. Europe
10.2.1.5.2.1. Market
Estimation, 2015 - 2022
10.2.1.5.2.2. Market
Forecast, 2023 - 2031
10.2.1.5.3. Asia Pacific
10.2.1.5.3.1. Market
Estimation, 2015 - 2022
10.2.1.5.3.2. Market
Forecast, 2023 - 2031
10.2.1.5.4. Middle East and Africa
10.2.1.5.4.1. Market
Estimation, 2015 - 2022
10.2.1.5.4.2. Market
Forecast, 2023 - 2031
10.2.1.5.5. Latin America
10.2.1.5.5.1. Market
Estimation, 2015 - 2022
10.2.1.5.5.2. Market
Forecast, 2023 - 2031
10.2.2. Research
Organizations
10.2.2.1. Definition
10.2.2.2. Market Estimation and Penetration, 2015 - 2022
10.2.2.3. Market Forecast, 2023 - 2031
10.2.2.4. Compound Annual Growth Rate (CAGR)
10.2.2.5. Regional Bifurcation
10.2.2.5.1. North America
10.2.2.5.1.1. Market
Estimation, 2015 - 2022
10.2.2.5.1.2. Market
Forecast, 2023 - 2031
10.2.2.5.2. Europe
10.2.2.5.2.1. Market
Estimation, 2015 - 2022
10.2.2.5.2.2. Market
Forecast, 2023 - 2031
10.2.2.5.3. Asia Pacific
10.2.2.5.3.1. Market
Estimation, 2015 - 2022
10.2.2.5.3.2. Market
Forecast, 2023 - 2031
10.2.2.5.4. Middle East and Africa
10.2.2.5.4.1. Market
Estimation, 2015 - 2022
10.2.2.5.4.2. Market
Forecast, 2023 - 2031
10.2.2.5.5. Latin America
10.2.2.5.5.1. Market
Estimation, 2015 - 2022
10.2.2.5.5.2. Market
Forecast, 2023 - 2031
10.2.3. Others
10.2.3.1. Definition
10.2.3.2. Market Estimation and Penetration, 2015 - 2022
10.2.3.3. Market Forecast, 2023 - 2031
10.2.3.4. Compound Annual Growth Rate (CAGR)
10.2.3.5. Regional Bifurcation
10.2.3.5.1. North America
10.2.3.5.1.1. Market
Estimation, 2015 - 2022
10.2.3.5.1.2. Market
Forecast, 2023 - 2031
10.2.3.5.2. Europe
10.2.3.5.2.1. Market
Estimation, 2015 - 2022
10.2.3.5.2.2. Market
Forecast, 2023 - 2031
10.2.3.5.3. Asia Pacific
10.2.3.5.3.1. Market
Estimation, 2015 - 2022
10.2.3.5.3.2. Market
Forecast, 2023 - 2031
10.2.3.5.4. Middle East and Africa
10.2.3.5.4.1. Market
Estimation, 2015 - 2022
10.2.3.5.4.2. Market
Forecast, 2023 - 2031
10.2.3.5.5. Latin America
10.2.3.5.5.1. Market
Estimation, 2015 - 2022
10.2.3.5.5.2. Market
Forecast, 2023 - 2031
10.3. Key
Segment for Channeling Investments
10.3.1. By End
Users
11. North America Post-marketing Pharmacovigilance and Medical
Information Market Analysis and Forecasts, 2023 - 2031
11.1. Overview
11.1.1. North
America Post-marketing Pharmacovigilance and Medical Information Market Revenue
(US$ Mn)
11.2. North
America Post-marketing Pharmacovigilance and Medical Information Market Revenue
(US$ Mn) and Forecasts, By Offering
11.2.1. Case
Processing and Management
11.2.2. Aggregate
Reporting (Including PSURs, PBRERs, DSURs, Etc.).
11.2.3. Audits
and Inspection Support Services
11.2.4. Signal
Detection and Risk Management
11.2.5. Global
and Local Literature Screening
11.2.6. Consulting
Services
11.2.7. Medical
Writing and Publishing
11.2.7.1. Regulatory Publishing
11.2.7.2. Clinical Studies
11.2.7.3. Educational
11.2.7.4. Others
11.2.8. Others
11.3. North
America Post-marketing Pharmacovigilance and Medical Information Market Revenue
(US$ Mn) and Forecasts, By Organization Size
11.3.1. Small
and Medium Organizations
11.3.2. Large
Organizations
11.4. North
America Post-marketing Pharmacovigilance and Medical Information Market Revenue
(US$ Mn) and Forecasts, By End Users
11.4.1. Pharmaceutical
Companies
11.4.2. Research
Organizations
11.4.3. Others
11.5. North
America Post-marketing Pharmacovigilance and Medical Information Market Revenue
(US$ Mn) and Forecasts, By Country
11.5.1. U.S
11.5.1.1. U.S Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By
Offering
11.5.1.1.1. Case Processing and Management
11.5.1.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
11.5.1.1.3. Audits and Inspection Support Services
11.5.1.1.4. Signal Detection and Risk Management
11.5.1.1.5. Global and Local Literature Screening
11.5.1.1.6. Consulting Services
11.5.1.1.7. Medical Writing and Publishing
11.5.1.1.7.1. Regulatory
Publishing
11.5.1.1.7.2. Clinical
Studies
11.5.1.1.7.3. Educational
11.5.1.1.7.4. Others
11.5.1.1.8. Others
11.5.1.2. U.S Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By Organization Size
11.5.1.2.1. Small and Medium Organizations
11.5.1.2.2. Large Organizations
11.5.1.3. U.S Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By End Users
11.5.1.3.1. Pharmaceutical Companies
11.5.1.3.2. Research Organizations
11.5.1.3.3. Others
11.5.2. Canada
11.5.2.1. Canada Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Offering
11.5.2.1.1. Case Processing and Management
11.5.2.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
11.5.2.1.3. Audits and Inspection Support Services
11.5.2.1.4. Signal Detection and Risk Management
11.5.2.1.5. Global and Local Literature Screening
11.5.2.1.6. Consulting Services
11.5.2.1.7. Medical Writing and Publishing
11.5.2.1.7.1. Regulatory
Publishing
11.5.2.1.7.2. Clinical
Studies
11.5.2.1.7.3. Educational
11.5.2.1.7.4. Others
11.5.2.1.8. Others
11.5.2.2. Canada Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Organization Size
11.5.2.2.1. Small and Medium Organizations
11.5.2.2.2. Large Organizations
11.5.2.3. Canada Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By End Users
11.5.2.3.1. Pharmaceutical Companies
11.5.2.3.2. Research Organizations
11.5.2.3.3. Others
11.5.3. Mexico
11.5.3.1. Mexico Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Offering
11.5.3.1.1. Case Processing and Management
11.5.3.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
11.5.3.1.3. Audits and Inspection Support Services
11.5.3.1.4. Signal Detection and Risk Management
11.5.3.1.5. Global and Local Literature Screening
11.5.3.1.6. Consulting Services
11.5.3.1.7. Medical Writing and Publishing
11.5.3.1.7.1. Regulatory
Publishing
11.5.3.1.7.2. Clinical
Studies
11.5.3.1.7.3. Educational
11.5.3.1.7.4. Others
11.5.3.1.8. Others
11.5.3.2. Mexico Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Organization Size
11.5.3.2.1. Small and Medium Organizations
11.5.3.2.2. Large Organizations
11.5.3.3. Mexico Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By End Users
11.5.3.3.1. Pharmaceutical Companies
11.5.3.3.2. Research Organizations
11.5.3.3.3. Others
11.5.4. Rest of
North America
11.5.4.1. Rest of North America Post-marketing Pharmacovigilance and
Medical Information Market Revenue (US$ Mn) and Forecasts, By Offering
11.5.4.1.1. Case Processing and Management
11.5.4.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
11.5.4.1.3. Audits and Inspection Support Services
11.5.4.1.4. Signal Detection and Risk Management
11.5.4.1.5. Global and Local Literature Screening
11.5.4.1.6. Consulting Services
11.5.4.1.7. Medical Writing and Publishing
11.5.4.1.7.1. Regulatory
Publishing
11.5.4.1.7.2. Clinical
Studies
11.5.4.1.7.3. Educational
11.5.4.1.7.4. Others
11.5.4.1.8. Others
11.5.4.2. Rest of North America Post-marketing Pharmacovigilance and
Medical Information Market Revenue (US$ Mn) and Forecasts, By Organization Size
11.5.4.2.1. Small and Medium Organizations
11.5.4.2.2. Large Organizations
11.5.4.3. Rest of North America Post-marketing Pharmacovigilance and
Medical Information Market Revenue (US$ Mn) and Forecasts, By End Users
11.5.4.3.1. Pharmaceutical Companies
11.5.4.3.2. Research Organizations
11.5.4.3.3. Others
11.6. Key
Segment for Channeling Investments
11.6.1. By
Country
11.6.2. By Offering
11.6.3. By
Organization Size
11.6.4. By End
Users
12. Europe Post-marketing Pharmacovigilance and Medical
Information Market Analysis and Forecasts, 2023 - 2031
12.1. Overview
12.1.1. Europe
Post-marketing Pharmacovigilance and Medical Information Market Revenue (US$
Mn)
12.2. Europe
Post-marketing Pharmacovigilance and Medical Information Market Revenue (US$
Mn) and Forecasts, By Offering
12.2.1. Case
Processing and Management
12.2.2. Aggregate
Reporting (Including PSURs, PBRERs, DSURs, Etc.).
12.2.3. Audits
and Inspection Support Services
12.2.4. Signal
Detection and Risk Management
12.2.5. Global
and Local Literature Screening
12.2.6. Consulting
Services
12.2.7. Medical
Writing and Publishing
12.2.7.1. Regulatory Publishing
12.2.7.2. Clinical Studies
12.2.7.3. Educational
12.2.7.4. Others
12.2.8. Others
12.3. Europe
Post-marketing Pharmacovigilance and Medical Information Market Revenue (US$
Mn) and Forecasts, By Organization Size
12.3.1. Small
and Medium Organizations
12.3.2. Large
Organizations
12.4. Europe
Post-marketing Pharmacovigilance and Medical Information Market Revenue (US$
Mn) and Forecasts, By End Users
12.4.1. Pharmaceutical
Companies
12.4.2. Research
Organizations
12.4.3. Others
12.5. Europe
Post-marketing Pharmacovigilance and Medical Information Market Revenue (US$
Mn) and Forecasts, By Country
12.5.1. France
12.5.1.1. France Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Offering
12.5.1.1.1. Case Processing and Management
12.5.1.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
12.5.1.1.3. Audits and Inspection Support Services
12.5.1.1.4. Signal Detection and Risk Management
12.5.1.1.5. Global and Local Literature Screening
12.5.1.1.6. Consulting Services
12.5.1.1.7. Medical Writing and Publishing
12.5.1.1.7.1. Regulatory
Publishing
12.5.1.1.7.2. Clinical
Studies
12.5.1.1.7.3. Educational
12.5.1.1.7.4. Others
12.5.1.1.8. Others
12.5.1.2. France Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Organization Size
12.5.1.2.1. Small and Medium Organizations
12.5.1.2.2. Large Organizations
12.5.1.3. France Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By End Users
12.5.1.3.1. Pharmaceutical Companies
12.5.1.3.2. Research Organizations
12.5.1.3.3. Others
12.5.2. The UK
12.5.2.1. The UK Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Offering
12.5.2.1.1. Case Processing and Management
12.5.2.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
12.5.2.1.3. Audits and Inspection Support Services
12.5.2.1.4. Signal Detection and Risk Management
12.5.2.1.5. Global and Local Literature Screening
12.5.2.1.6. Consulting Services
12.5.2.1.7. Medical Writing and Publishing
12.5.2.1.7.1. Regulatory
Publishing
12.5.2.1.7.2. Clinical
Studies
12.5.2.1.7.3. Educational
12.5.2.1.7.4. Others
12.5.2.1.8. Others
12.5.2.2. The UK Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Organization Size
12.5.2.2.1. Small and Medium Organizations
12.5.2.2.2. Large Organizations
12.5.2.3. The UK Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By End Users
12.5.2.3.1. Pharmaceutical Companies
12.5.2.3.2. Research Organizations
12.5.2.3.3. Others
12.5.3. Spain
12.5.3.1. Spain Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By
Offering
12.5.3.1.1. Case Processing and Management
12.5.3.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
12.5.3.1.3. Audits and Inspection Support Services
12.5.3.1.4. Signal Detection and Risk Management
12.5.3.1.5. Global and Local Literature Screening
12.5.3.1.6. Consulting Services
12.5.3.1.7. Medical Writing and Publishing
12.5.3.1.7.1. Regulatory
Publishing
12.5.3.1.7.2. Clinical
Studies
12.5.3.1.7.3. Educational
12.5.3.1.7.4. Others
12.5.3.1.8. Others
12.5.3.2. Spain Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By Organization Size
12.5.3.2.1. Small and Medium Organizations
12.5.3.2.2. Large Organizations
12.5.3.3. Spain Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By End Users
12.5.3.3.1. Pharmaceutical Companies
12.5.3.3.2. Research Organizations
12.5.3.3.3. Others
12.5.4. Germany
12.5.4.1. Germany Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Offering
12.5.4.1.1. Case Processing and Management
12.5.4.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
12.5.4.1.3. Audits and Inspection Support Services
12.5.4.1.4. Signal Detection and Risk Management
12.5.4.1.5. Global and Local Literature Screening
12.5.4.1.6. Consulting Services
12.5.4.1.7. Medical Writing and Publishing
12.5.4.1.7.1. Regulatory
Publishing
12.5.4.1.7.2. Clinical
Studies
12.5.4.1.7.3. Educational
12.5.4.1.7.4. Others
12.5.4.1.8. Others
12.5.4.2. Germany Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Organization Size
12.5.4.2.1. Small and Medium Organizations
12.5.4.2.2. Large Organizations
12.5.4.3. Germany Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By End Users
12.5.4.3.1. Pharmaceutical Companies
12.5.4.3.2. Research Organizations
12.5.4.3.3. Others
12.5.5. Italy
12.5.5.1. Italy Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By
Offering
12.5.5.1.1. Case Processing and Management
12.5.5.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
12.5.5.1.3. Audits and Inspection Support Services
12.5.5.1.4. Signal Detection and Risk Management
12.5.5.1.5. Global and Local Literature Screening
12.5.5.1.6. Consulting Services
12.5.5.1.7. Medical Writing and Publishing
12.5.5.1.7.1. Regulatory
Publishing
12.5.5.1.7.2. Clinical
Studies
12.5.5.1.7.3. Educational
12.5.5.1.7.4. Others
12.5.5.1.8. Others
12.5.5.2. Italy Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By Organization Size
12.5.5.2.1. Small and Medium Organizations
12.5.5.2.2. Large Organizations
12.5.5.3. Italy Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By End Users
12.5.5.3.1. Pharmaceutical Companies
12.5.5.3.2. Research Organizations
12.5.5.3.3. Others
12.5.6. Nordic
Countries
12.5.6.1. Nordic Countries Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Offering
12.5.6.1.1. Case Processing and Management
12.5.6.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
12.5.6.1.3. Audits and Inspection Support Services
12.5.6.1.4. Signal Detection and Risk Management
12.5.6.1.5. Global and Local Literature Screening
12.5.6.1.6. Consulting Services
12.5.6.1.7. Medical Writing and Publishing
12.5.6.1.7.1. Regulatory
Publishing
12.5.6.1.7.2. Clinical
Studies
12.5.6.1.7.3. Educational
12.5.6.1.7.4. Others
12.5.6.1.8. Others
12.5.6.2. Nordic Countries Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Organization Size
12.5.6.2.1. Small and Medium Organizations
12.5.6.2.2. Large Organizations
12.5.6.3. Nordic Countries Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By End Users
12.5.6.3.1. Pharmaceutical Companies
12.5.6.3.2. Research Organizations
12.5.6.3.3. Others
12.5.6.4. Nordic Countries Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Country
12.5.6.4.1. Denmark
12.5.6.4.2. Finland
12.5.6.4.3. Iceland
12.5.6.4.4. Sweden
12.5.6.4.5. Norway
12.5.7. Benelux
Union
12.5.7.1. Benelux Union Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Offering
12.5.7.1.1. Case Processing and Management
12.5.7.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
12.5.7.1.3. Audits and Inspection Support Services
12.5.7.1.4. Signal Detection and Risk Management
12.5.7.1.5. Global and Local Literature Screening
12.5.7.1.6. Consulting Services
12.5.7.1.7. Medical Writing and Publishing
12.5.7.1.7.1. Regulatory
Publishing
12.5.7.1.7.2. Clinical
Studies
12.5.7.1.7.3. Educational
12.5.7.1.7.4. Others
12.5.7.1.8. Others
12.5.7.2. Benelux Union Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Organization Size
12.5.7.2.1. Small and Medium Organizations
12.5.7.2.2. Large Organizations
12.5.7.3. Benelux Union Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By End Users
12.5.7.3.1. Pharmaceutical Companies
12.5.7.3.2. Research Organizations
12.5.7.3.3. Others
12.5.7.4. Benelux Union Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Country
12.5.7.4.1. Belgium
12.5.7.4.2. The Netherlands
12.5.7.4.3. Luxembourg
12.5.8. Rest of
Europe
12.5.8.1. Rest of Europe Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Offering
12.5.8.1.1. Case Processing and Management
12.5.8.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
12.5.8.1.3. Audits and Inspection Support Services
12.5.8.1.4. Signal Detection and Risk Management
12.5.8.1.5. Global and Local Literature Screening
12.5.8.1.6. Consulting Services
12.5.8.1.7. Medical Writing and Publishing
12.5.8.1.7.1. Regulatory
Publishing
12.5.8.1.7.2. Clinical
Studies
12.5.8.1.7.3. Educational
12.5.8.1.7.4. Others
12.5.8.1.8. Others
12.5.8.2. Rest of Europe Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Organization Size
12.5.8.2.1. Small and Medium Organizations
12.5.8.2.2. Large Organizations
12.5.8.3. Rest of Europe Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By End Users
12.5.8.3.1. Pharmaceutical Companies
12.5.8.3.2. Research Organizations
12.5.8.3.3. Others
12.6. Key
Segment for Channeling Investments
12.6.1. By
Country
12.6.2. By Offering
12.6.3. By
Organization Size
12.6.4. By End
Users
13. Asia Pacific Post-marketing Pharmacovigilance and Medical
Information Market Analysis and Forecasts, 2023 - 2031
13.1. Overview
13.1.1. Asia
Pacific Post-marketing Pharmacovigilance and Medical Information Market Revenue
(US$ Mn)
13.2. Asia
Pacific Post-marketing Pharmacovigilance and Medical Information Market Revenue
(US$ Mn) and Forecasts, By Offering
13.2.1. Case
Processing and Management
13.2.2. Aggregate
Reporting (Including PSURs, PBRERs, DSURs, Etc.).
13.2.3. Audits
and Inspection Support Services
13.2.4. Signal
Detection and Risk Management
13.2.5. Global
and Local Literature Screening
13.2.6. Consulting
Services
13.2.7. Medical
Writing and Publishing
13.2.7.1. Regulatory Publishing
13.2.7.2. Clinical Studies
13.2.7.3. Educational
13.2.7.4. Others
13.2.8. Others
13.3. Asia
Pacific Post-marketing Pharmacovigilance and Medical Information Market Revenue
(US$ Mn) and Forecasts, By Organization Size
13.3.1. Small
and Medium Organizations
13.3.2. Large
Organizations
13.4. Asia
Pacific Post-marketing Pharmacovigilance and Medical Information Market Revenue
(US$ Mn) and Forecasts, By End Users
13.4.1. Pharmaceutical
Companies
13.4.2. Research
Organizations
13.4.3. Others
13.5. Asia
Pacific Post-marketing Pharmacovigilance and Medical Information Market Revenue
(US$ Mn) and Forecasts, By Country
13.5.1. China
13.5.1.1. China Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By
Offering
13.5.1.1.1. Case Processing and Management
13.5.1.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
13.5.1.1.3. Audits and Inspection Support Services
13.5.1.1.4. Signal Detection and Risk Management
13.5.1.1.5. Global and Local Literature Screening
13.5.1.1.6. Consulting Services
13.5.1.1.7. Medical Writing and Publishing
13.5.1.1.7.1. Regulatory
Publishing
13.5.1.1.7.2. Clinical
Studies
13.5.1.1.7.3. Educational
13.5.1.1.7.4. Others
13.5.1.1.8. Others
13.5.1.2. China Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By Organization Size
13.5.1.2.1. Small and Medium Organizations
13.5.1.2.2. Large Organizations
13.5.1.3. China Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By End Users
13.5.1.3.1. Pharmaceutical Companies
13.5.1.3.2. Research Organizations
13.5.1.3.3. Others
13.5.2. Japan
13.5.2.1. Japan Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By
Offering
13.5.2.1.1. Case Processing and Management
13.5.2.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
13.5.2.1.3. Audits and Inspection Support Services
13.5.2.1.4. Signal Detection and Risk Management
13.5.2.1.5. Global and Local Literature Screening
13.5.2.1.6. Consulting Services
13.5.2.1.7. Medical Writing and Publishing
13.5.2.1.7.1. Regulatory
Publishing
13.5.2.1.7.2. Clinical
Studies
13.5.2.1.7.3. Educational
13.5.2.1.7.4. Others
13.5.2.1.8. Others
13.5.2.2. Japan Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By Organization Size
13.5.2.2.1. Small and Medium Organizations
13.5.2.2.2. Large Organizations
13.5.2.3. Japan Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By End Users
13.5.2.3.1. Pharmaceutical Companies
13.5.2.3.2. Research Organizations
13.5.2.3.3. Others
13.5.3. India
13.5.3.1. India Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By
Offering
13.5.3.1.1. Case Processing and Management
13.5.3.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
13.5.3.1.3. Audits and Inspection Support Services
13.5.3.1.4. Signal Detection and Risk Management
13.5.3.1.5. Global and Local Literature Screening
13.5.3.1.6. Consulting Services
13.5.3.1.7. Medical Writing and Publishing
13.5.3.1.7.1. Regulatory
Publishing
13.5.3.1.7.2. Clinical
Studies
13.5.3.1.7.3. Educational
13.5.3.1.7.4. Others
13.5.3.1.8. Others
13.5.3.2. India Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By Organization Size
13.5.3.2.1. Small and Medium Organizations
13.5.3.2.2. Large Organizations
13.5.3.3. India Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By End Users
13.5.3.3.1. Pharmaceutical Companies
13.5.3.3.2. Research Organizations
13.5.3.3.3. Others
13.5.4. New
Zealand
13.5.4.1. New Zealand Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Offering
13.5.4.1.1. Case Processing and Management
13.5.4.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
13.5.4.1.3. Audits and Inspection Support Services
13.5.4.1.4. Signal Detection and Risk Management
13.5.4.1.5. Global and Local Literature Screening
13.5.4.1.6. Consulting Services
13.5.4.1.7. Medical Writing and Publishing
13.5.4.1.7.1. Regulatory
Publishing
13.5.4.1.7.2. Clinical
Studies
13.5.4.1.7.3. Educational
13.5.4.1.7.4. Others
13.5.4.1.8. Others
13.5.4.2. New Zealand Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Organization Size
13.5.4.2.1. Small and Medium Organizations
13.5.4.2.2. Large Organizations
13.5.4.3. New Zealand Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By End Users
13.5.4.3.1. Pharmaceutical Companies
13.5.4.3.2. Research Organizations
13.5.4.3.3. Others
13.5.5. Australia
13.5.5.1. Australia Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Offering
13.5.5.1.1. Case Processing and Management
13.5.5.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
13.5.5.1.3. Audits and Inspection Support Services
13.5.5.1.4. Signal Detection and Risk Management
13.5.5.1.5. Global and Local Literature Screening
13.5.5.1.6. Consulting Services
13.5.5.1.7. Medical Writing and Publishing
13.5.5.1.7.1. Regulatory
Publishing
13.5.5.1.7.2. Clinical
Studies
13.5.5.1.7.3. Educational
13.5.5.1.7.4. Others
13.5.5.1.8. Others
13.5.5.2. Australia Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Organization Size
13.5.5.2.1. Small and Medium Organizations
13.5.5.2.2. Large Organizations
13.5.5.3. Australia Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By End Users
13.5.5.3.1. Pharmaceutical Companies
13.5.5.3.2. Research Organizations
13.5.5.3.3. Others
13.5.6. South
Korea
13.5.6.1. South Korea Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Offering
13.5.6.1.1. Case Processing and Management
13.5.6.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
13.5.6.1.3. Audits and Inspection Support Services
13.5.6.1.4. Signal Detection and Risk Management
13.5.6.1.5. Global and Local Literature Screening
13.5.6.1.6. Consulting Services
13.5.6.1.7. Medical Writing and Publishing
13.5.6.1.7.1. Regulatory
Publishing
13.5.6.1.7.2. Clinical
Studies
13.5.6.1.7.3. Educational
13.5.6.1.7.4. Others
13.5.6.1.8. Others
13.5.6.2. South Korea Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Organization Size
13.5.6.2.1. Small and Medium Organizations
13.5.6.2.2. Large Organizations
13.5.6.3. South Korea Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By End Users
13.5.6.3.1. Pharmaceutical Companies
13.5.6.3.2. Research Organizations
13.5.6.3.3. Others
13.5.7. Southeast
Asia
13.5.7.1. Southeast Asia Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Offering
13.5.7.1.1. Case Processing and Management
13.5.7.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
13.5.7.1.3. Audits and Inspection Support Services
13.5.7.1.4. Signal Detection and Risk Management
13.5.7.1.5. Global and Local Literature Screening
13.5.7.1.6. Consulting Services
13.5.7.1.7. Medical Writing and Publishing
13.5.7.1.7.1. Regulatory
Publishing
13.5.7.1.7.2. Clinical
Studies
13.5.7.1.7.3. Educational
13.5.7.1.7.4. Others
13.5.7.1.8. Others
13.5.7.2. Southeast Asia Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Organization Size
13.5.7.2.1. Small and Medium Organizations
13.5.7.2.2. Large Organizations
13.5.7.3. Southeast Asia Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By End Users
13.5.7.3.1. Pharmaceutical Companies
13.5.7.3.2. Research Organizations
13.5.7.3.3. Others
13.5.7.4. Southeast Asia Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Country
13.5.7.4.1. Indonesia
13.5.7.4.2. Thailand
13.5.7.4.3. Malaysia
13.5.7.4.4. Singapore
13.5.7.4.5. Rest of Southeast Asia
13.5.8. Rest of
Asia Pacific
13.5.8.1. Rest of Asia Pacific Post-marketing Pharmacovigilance and
Medical Information Market Revenue (US$ Mn) and Forecasts, By Offering
13.5.8.1.1. Case Processing and Management
13.5.8.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
13.5.8.1.3. Audits and Inspection Support Services
13.5.8.1.4. Signal Detection and Risk Management
13.5.8.1.5. Global and Local Literature Screening
13.5.8.1.6. Consulting Services
13.5.8.1.7. Medical Writing and Publishing
13.5.8.1.7.1. Regulatory
Publishing
13.5.8.1.7.2. Clinical
Studies
13.5.8.1.7.3. Educational
13.5.8.1.7.4. Others
13.5.8.1.8. Others
13.5.8.2. Rest of Asia Pacific Post-marketing Pharmacovigilance and
Medical Information Market Revenue (US$ Mn) and Forecasts, By Organization Size
13.5.8.2.1. Small and Medium Organizations
13.5.8.2.2. Large Organizations
13.5.8.3. Rest of Asia Pacific Post-marketing Pharmacovigilance and
Medical Information Market Revenue (US$ Mn) and Forecasts, By End Users
13.5.8.3.1. Pharmaceutical Companies
13.5.8.3.2. Research Organizations
13.5.8.3.3. Others
13.6. Key
Segment for Channeling Investments
13.6.1. By
Country
13.6.2. By Offering
13.6.3. By
Organization Size
13.6.4. By End
Users
14. Middle East and Africa Post-marketing Pharmacovigilance and
Medical Information Market Analysis and Forecasts, 2023 - 2031
14.1. Overview
14.1.1. Middle
East and Africa Post-marketing Pharmacovigilance and Medical Information Market
Revenue (US$ Mn)
14.2. Middle
East and Africa Post-marketing Pharmacovigilance and Medical Information Market
Revenue (US$ Mn) and Forecasts, By
Offering
14.2.1. Case
Processing and Management
14.2.2. Aggregate
Reporting (Including PSURs, PBRERs, DSURs, Etc.).
14.2.3. Audits
and Inspection Support Services
14.2.4. Signal
Detection and Risk Management
14.2.5. Global
and Local Literature Screening
14.2.6. Consulting
Services
14.2.7. Medical
Writing and Publishing
14.2.7.1. Regulatory Publishing
14.2.7.2. Clinical Studies
14.2.7.3. Educational
14.2.7.4. Others
14.2.8. Others
14.3. Middle
East and Africa Post-marketing Pharmacovigilance and Medical Information Market
Revenue (US$ Mn) and Forecasts, By Organization Size
14.3.1. Small
and Medium Organizations
14.3.2. Large
Organizations
14.4. Middle
East and Africa Post-marketing Pharmacovigilance and Medical Information Market
Revenue (US$ Mn) and Forecasts, By End Users
14.4.1. Pharmaceutical
Companies
14.4.2. Research
Organizations
14.4.3. Others
14.5. Middle
East and Africa Post-marketing Pharmacovigilance and Medical Information Market
Revenue (US$ Mn) and Forecasts, By Country
14.5.1. Saudi
Arabia
14.5.1.1. Saudi Arabia Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Offering
14.5.1.1.1. Case Processing and Management
14.5.1.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
14.5.1.1.3. Audits and Inspection Support Services
14.5.1.1.4. Signal Detection and Risk Management
14.5.1.1.5. Global and Local Literature Screening
14.5.1.1.6. Consulting Services
14.5.1.1.7. Medical Writing and Publishing
14.5.1.1.7.1. Regulatory
Publishing
14.5.1.1.7.2. Clinical
Studies
14.5.1.1.7.3. Educational
14.5.1.1.7.4. Others
14.5.1.1.8. Others
14.5.1.2. Saudi Arabia Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Organization Size
14.5.1.2.1. Small and Medium Organizations
14.5.1.2.2. Large Organizations
14.5.1.3. Saudi Arabia Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By End Users
14.5.1.3.1. Pharmaceutical Companies
14.5.1.3.2. Research Organizations
14.5.1.3.3. Others
14.5.2. UAE
14.5.2.1. UAE Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By
Offering
14.5.2.1.1. Case Processing and Management
14.5.2.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
14.5.2.1.3. Audits and Inspection Support Services
14.5.2.1.4. Signal Detection and Risk Management
14.5.2.1.5. Global and Local Literature Screening
14.5.2.1.6. Consulting Services
14.5.2.1.7. Medical Writing and Publishing
14.5.2.1.7.1. Regulatory
Publishing
14.5.2.1.7.2. Clinical
Studies
14.5.2.1.7.3. Educational
14.5.2.1.7.4. Others
14.5.2.1.8. Others
14.5.2.2. UAE Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By Organization Size
14.5.2.2.1. Small and Medium Organizations
14.5.2.2.2. Large Organizations
14.5.2.3. UAE Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By End Users
14.5.2.3.1. Pharmaceutical Companies
14.5.2.3.2. Research Organizations
14.5.2.3.3. Others
14.5.3. Egypt
14.5.3.1. Egypt Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By
Offering
14.5.3.1.1. Case Processing and Management
14.5.3.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
14.5.3.1.3. Audits and Inspection Support Services
14.5.3.1.4. Signal Detection and Risk Management
14.5.3.1.5. Global and Local Literature Screening
14.5.3.1.6. Consulting Services
14.5.3.1.7. Medical Writing and Publishing
14.5.3.1.7.1. Regulatory
Publishing
14.5.3.1.7.2. Clinical
Studies
14.5.3.1.7.3. Educational
14.5.3.1.7.4. Others
14.5.3.1.8. Others
14.5.3.2. Egypt Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By Organization Size
14.5.3.2.1. Small and Medium Organizations
14.5.3.2.2. Large Organizations
14.5.3.3. Egypt Post-marketing Pharmacovigilance and Medical Information
Market Revenue (US$ Mn) and Forecasts, By End Users
14.5.3.3.1. Pharmaceutical Companies
14.5.3.3.2. Research Organizations
14.5.3.3.3. Others
14.5.4. Kuwait
14.5.4.1. Kuwait Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Offering
14.5.4.1.1. Case Processing and Management
14.5.4.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
14.5.4.1.3. Audits and Inspection Support Services
14.5.4.1.4. Signal Detection and Risk Management
14.5.4.1.5. Global and Local Literature Screening
14.5.4.1.6. Consulting Services
14.5.4.1.7. Medical Writing and Publishing
14.5.4.1.7.1. Regulatory
Publishing
14.5.4.1.7.2. Clinical
Studies
14.5.4.1.7.3. Educational
14.5.4.1.7.4. Others
14.5.4.1.8. Others
14.5.4.2. Kuwait Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Organization Size
14.5.4.2.1. Small and Medium Organizations
14.5.4.2.2. Large Organizations
14.5.4.3. Kuwait Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By End Users
14.5.4.3.1. Pharmaceutical Companies
14.5.4.3.2. Research Organizations
14.5.4.3.3. Others
14.5.5. South
Africa
14.5.5.1. South Africa Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Offering
14.5.5.1.1. Case Processing and Management
14.5.5.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
14.5.5.1.3. Audits and Inspection Support Services
14.5.5.1.4. Signal Detection and Risk Management
14.5.5.1.5. Global and Local Literature Screening
14.5.5.1.6. Consulting Services
14.5.5.1.7. Medical Writing and Publishing
14.5.5.1.7.1. Regulatory
Publishing
14.5.5.1.7.2. Clinical
Studies
14.5.5.1.7.3. Educational
14.5.5.1.7.4. Others
14.5.5.1.8. Others
14.5.5.2. South Africa Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Organization Size
14.5.5.2.1. Small and Medium Organizations
14.5.5.2.2. Large Organizations
14.5.5.3. South Africa Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By End Users
14.5.5.3.1. Pharmaceutical Companies
14.5.5.3.2. Research Organizations
14.5.5.3.3. Others
14.5.6. Rest of
Middle East & Africa
14.5.6.1. Rest of Middle East & Africa Post-marketing
Pharmacovigilance and Medical Information Market Revenue (US$ Mn) and
Forecasts, By Offering
14.5.6.1.1. Case Processing and Management
14.5.6.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
14.5.6.1.3. Audits and Inspection Support Services
14.5.6.1.4. Signal Detection and Risk Management
14.5.6.1.5. Global and Local Literature Screening
14.5.6.1.6. Consulting Services
14.5.6.1.7. Medical Writing and Publishing
14.5.6.1.7.1. Regulatory
Publishing
14.5.6.1.7.2. Clinical
Studies
14.5.6.1.7.3. Educational
14.5.6.1.7.4. Others
14.5.6.1.8. Others
14.5.6.2. Rest of Middle East & Africa Post-marketing
Pharmacovigilance and Medical Information Market Revenue (US$ Mn) and
Forecasts, By Organization Size
14.5.6.2.1. Small and Medium Organizations
14.5.6.2.2. Large Organizations
14.5.6.3. Rest of Middle East & Africa Post-marketing
Pharmacovigilance and Medical Information Market Revenue (US$ Mn) and
Forecasts, By End Users
14.5.6.3.1. Pharmaceutical Companies
14.5.6.3.2. Research Organizations
14.5.6.3.3. Others
14.6. Key
Segment for Channeling Investments
14.6.1. By
Country
14.6.2. By Offering
14.6.3. By
Organization Size
14.6.4. By End
Users
15. Latin America Post-marketing Pharmacovigilance and Medical
Information Market Analysis and Forecasts, 2023 - 2031
15.1. Overview
15.1.1. Latin
America Post-marketing Pharmacovigilance and Medical Information Market Revenue
(US$ Mn)
15.2. Latin
America Post-marketing Pharmacovigilance and Medical Information Market Revenue
(US$ Mn) and Forecasts, By Offering
15.2.1. Case
Processing and Management
15.2.2. Aggregate
Reporting (Including PSURs, PBRERs, DSURs, Etc.).
15.2.3. Audits
and Inspection Support Services
15.2.4. Signal
Detection and Risk Management
15.2.5. Global
and Local Literature Screening
15.2.6. Consulting
Services
15.2.7. Medical
Writing and Publishing
15.2.7.1. Regulatory Publishing
15.2.7.2. Clinical Studies
15.2.7.3. Educational
15.2.7.4. Others
15.2.8. Others
15.3. Latin
America Post-marketing Pharmacovigilance and Medical Information Market Revenue
(US$ Mn) and Forecasts, By Organization Size
15.3.1. Small
and Medium Organizations
15.3.2. Large
Organizations
15.4. Latin
America Post-marketing Pharmacovigilance and Medical Information Market Revenue
(US$ Mn) and Forecasts, By End Users
15.4.1. Pharmaceutical
Companies
15.4.2. Research
Organizations
15.4.3. Others
15.5. Latin
America Post-marketing Pharmacovigilance and Medical Information Market Revenue
(US$ Mn) and Forecasts, By Country
15.5.1. Brazil
15.5.1.1. Brazil Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Offering
15.5.1.1.1. Case Processing and Management
15.5.1.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
15.5.1.1.3. Audits and Inspection Support Services
15.5.1.1.4. Signal Detection and Risk Management
15.5.1.1.5. Global and Local Literature Screening
15.5.1.1.6. Consulting Services
15.5.1.1.7. Medical Writing and Publishing
15.5.1.1.7.1. Regulatory
Publishing
15.5.1.1.7.2. Clinical
Studies
15.5.1.1.7.3. Educational
15.5.1.1.7.4. Others
15.5.1.1.8. Others
15.5.1.2. Brazil Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Organization Size
15.5.1.2.1. Small and Medium Organizations
15.5.1.2.2. Large Organizations
15.5.1.3. Brazil Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By End Users
15.5.1.3.1. Pharmaceutical Companies
15.5.1.3.2. Research Organizations
15.5.1.3.3. Others
15.5.2. Argentina
15.5.2.1. Argentina Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Offering
15.5.2.1.1. Case Processing and Management
15.5.2.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
15.5.2.1.3. Audits and Inspection Support Services
15.5.2.1.4. Signal Detection and Risk Management
15.5.2.1.5. Global and Local Literature Screening
15.5.2.1.6. Consulting Services
15.5.2.1.7. Medical Writing and Publishing
15.5.2.1.7.1. Regulatory
Publishing
15.5.2.1.7.2. Clinical
Studies
15.5.2.1.7.3. Educational
15.5.2.1.7.4. Others
15.5.2.1.8. Others
15.5.2.2. Argentina Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By Organization Size
15.5.2.2.1. Small and Medium Organizations
15.5.2.2.2. Large Organizations
15.5.2.3. Argentina Post-marketing Pharmacovigilance and Medical
Information Market Revenue (US$ Mn) and Forecasts, By End Users
15.5.2.3.1. Pharmaceutical Companies
15.5.2.3.2. Research Organizations
15.5.2.3.3. Others
15.5.3. Rest of
Latin America
15.5.3.1. Rest of Latin America Post-marketing Pharmacovigilance and
Medical Information Market Revenue (US$ Mn) and Forecasts, By Offering
15.5.3.1.1. Case Processing and Management
15.5.3.1.2. Aggregate Reporting (Including PSURs, PBRERs, DSURs, Etc.).
15.5.3.1.3. Audits and Inspection Support Services
15.5.3.1.4. Signal Detection and Risk Management
15.5.3.1.5. Global and Local Literature Screening
15.5.3.1.6. Consulting Services
15.5.3.1.7. Medical Writing and Publishing
15.5.3.1.7.1. Regulatory
Publishing
15.5.3.1.7.2. Clinical
Studies
15.5.3.1.7.3. Educational
15.5.3.1.7.4. Others
15.5.3.1.8. Others
15.5.3.2. Rest of Latin America Post-marketing Pharmacovigilance and
Medical Information Market Revenue (US$ Mn) and Forecasts, By Organization Size
15.5.3.2.1. Small and Medium Organizations
15.5.3.2.2. Large Organizations
15.5.3.3. Rest of Latin America Post-marketing Pharmacovigilance and
Medical Information Market Revenue (US$ Mn) and Forecasts, By End Users
15.5.3.3.1. Pharmaceutical Companies
15.5.3.3.2. Research Organizations
15.5.3.3.3. Others
15.6. Key
Segment for Channeling Investments
15.6.1. By
Country
15.6.2. By Offering
15.6.3. By
Organization Size
15.6.4. By End
Users
16. Competitive Benchmarking
16.1. Market
Share Analysis, 2022
16.2. Global
Presence and Growth Strategies
16.2.1. Mergers
and Acquisitions
16.2.2. Product
Launches
16.2.3. Investments
Trends
16.2.4. R&D
Initiatives
17. Player Profiles
17.1. A2
Healthcare Corp.
17.1.1. Company
Details
17.1.2. Company
Overview
17.1.3. Product
Offerings
17.1.4. Key
Developments
17.1.5. Financial
Analysis
17.1.6. SWOT
Analysis
17.1.7. Business
Strategies
17.2. Accenture
17.2.1. Company
Details
17.2.2. Company
Overview
17.2.3. Product
Offerings
17.2.4. Key
Developments
17.2.5. Financial
Analysis
17.2.6. SWOT
Analysis
17.2.7. Business
Strategies
17.3. Clario
17.3.1. Company
Details
17.3.2. Company
Overview
17.3.3. Product
Offerings
17.3.4. Key
Developments
17.3.5. Financial
Analysis
17.3.6. SWOT
Analysis
17.3.7. Business
Strategies
17.4. Cognizant
17.4.1. Company
Details
17.4.2. Company
Overview
17.4.3. Product
Offerings
17.4.4. Key
Developments
17.4.5. Financial
Analysis
17.4.6. SWOT
Analysis
17.4.7. Business
Strategies
17.5. Genpact
17.5.1. Company
Details
17.5.2. Company
Overview
17.5.3. Product
Offerings
17.5.4. Key
Developments
17.5.5. Financial
Analysis
17.5.6. SWOT
Analysis
17.5.7. Business
Strategies
17.6. ICON plc
17.6.1. Company
Details
17.6.2. Company
Overview
17.6.3. Product
Offerings
17.6.4. Key
Developments
17.6.5. Financial
Analysis
17.6.6. SWOT
Analysis
17.6.7. Business
Strategies
17.7. IQVIA
17.7.1. Company
Details
17.7.2. Company
Overview
17.7.3. Product
Offerings
17.7.4. Key
Developments
17.7.5. Financial
Analysis
17.7.6. SWOT
Analysis
17.7.7. Business
Strategies
17.8. Labcorp
Drug Development
17.8.1. Company
Details
17.8.2. Company
Overview
17.8.3. Product
Offerings
17.8.4. Key
Developments
17.8.5. Financial
Analysis
17.8.6. SWOT
Analysis
17.8.7. Business
Strategies
17.9. Parexel
International Corporation
17.9.1. Company
Details
17.9.2. Company
Overview
17.9.3. Product
Offerings
17.9.4. Key
Developments
17.9.5. Financial
Analysis
17.9.6. SWOT
Analysis
17.9.7. Business
Strategies
17.10. PharSafer
17.10.1. Company
Details
17.10.2. Company
Overview
17.10.3. Product
Offerings
17.10.4. Key
Developments
17.10.5. Financial
Analysis
17.10.6. SWOT
Analysis
17.10.7. Business
Strategies
17.11. PPD Inc.
17.11.1. Company
Details
17.11.2. Company
Overview
17.11.3. Product
Offerings
17.11.4. Key
Developments
17.11.5. Financial
Analysis
17.11.6. SWOT
Analysis
17.11.7. Business
Strategies
17.12. Qvigilance
17.12.1. Company
Details
17.12.2. Company
Overview
17.12.3. Product
Offerings
17.12.4. Key
Developments
17.12.5. Financial
Analysis
17.12.6. SWOT
Analysis
17.12.7. Business
Strategies
17.13. Syneos
Health.
17.13.1. Company
Details
17.13.2. Company
Overview
17.13.3. Product
Offerings
17.13.4. Key
Developments
17.13.5. Financial
Analysis
17.13.6. SWOT
Analysis
17.13.7. Business
Strategies
17.14. Universal
Medica Group
17.14.1. Company
Details
17.14.2. Company
Overview
17.14.3. Product
Offerings
17.14.4. Key
Developments
17.14.5. Financial
Analysis
17.14.6. SWOT
Analysis
17.14.7. Business
Strategies
17.15. Wipro
Limited
17.15.1. Company
Details
17.15.2. Company
Overview
17.15.3. Product
Offerings
17.15.4. Key
Developments
17.15.5. Financial
Analysis
17.15.6. SWOT
Analysis
17.15.7. Business Strategies
17.16. Other
market participants
18. Key Findings
Note: This ToC is tentative
and can be changed according to the research study conducted during the course
of report completion.
**Exclusive for Multi-User
and Enterprise User.
At Absolute Markets Insights, we are engaged in building both global as well as country specific reports. As a result, the approach taken for deriving the estimation and forecast for a specific country is a bit unique and different in comparison to the global research studies. In this case, we not only study the concerned market factors & trends prevailing in a particular country (from secondary research) but we also tend to calculate the actual market size & forecast from the revenue generated from the market participants involved in manufacturing or distributing the any concerned product. These companies can also be service providers. For analyzing any country specifically, we do consider the growth factors prevailing under the states/cities/county for the same. For instance, if we are analyzing an industry specific to United States, we primarily need to study about the states present under the same(where the product/service has the highest growth). Similar analysis will be followed by other countries. Our scope of the report changes with different markets.
Our research study is mainly implement through a mix of both secondary and primary research. Various sources such as industry magazines, trade journals, and government websites and trade associations are reviewed for gathering precise data. Primary interviews are conducted to validate the market size derived from secondary research. Industry experts, major manufacturers and distributors are contacted for further validation purpose on the current market penetration and growth trends.
Prominent participants in our primary research process include:
- Key Opinion Leaders namely the CEOs, CSOs, VPs, purchasing managers, amongst others
- Research and development participants, distributors/suppliers and subject matter experts
Secondary Research includes data extracted from paid data sources:
- Reuters
- Factiva
- Bloomberg
- One Source
- Hoovers
Research Methodology
Key Inclusions

Reach to us
Call us on
+91-74002-42424
Drop us an email at
sales@absolutemarketsinsights.com
Why Absolute Markets Insights?
An effective strategy is the entity that influences a business to stand out of the crowd. An organization with a phenomenal strategy for success dependably has the edge over the rivals in the market. It offers the organizations a head start in planning their strategy. Absolute Market Insights is the new initiation in the industry that will furnish you with the lead your business needs. Absolute Market Insights is the best destination for your business intelligence and analytical solutions; essentially because our qualitative and quantitative sources of information are competent to give one-stop solutions. We inventively combine qualitative and quantitative research in accurate proportions to have the best report, which not only gives the most recent insights but also assists you to grow.

