Global RWE Oncology Market By Component (Real-world Datasets (EMR/EHR/Clinical Data, Administrative Claims & Billing Data, Pharmacy Data, Cancer Registries, Specialty Data Providers, Others) Real-world Consulting & Analytics Services) By Application (Clinical Trials, Drug Development and Approvals, Post marketing Safety Studies, Market Access and Reimbursement, Medical Device Development, Others) By End Users (Biotechnology Companies, Medical Device Companies, Healthcare Payers, Healthcare Providers, Others) By Region (North America (U.S., Canada, Mexico, Rest Of North America), Europe (France, The UK, Spain, Germany, Italy, Denmark, Finland, Iceland, Sweden, Norway, Belgium, The Netherlands, Luxembourg, Rest of Europe), Asia Pacific (China, Japan, India, New Zealand, Australia, South Korea, Southeast Asia, Indonesia, Thailand, Malaysia, Singapore, Rest of Southeast Asia, Rest of Asia Pacific, Middle East & Africa (Saudi Arabia, UAE, Egypt, Kuwait, South Africa, Rest of Middle East & Africa) Latin America (Brazil, Argentina, Rest of Latin America)) - Global Insights, Growth, Size, Comparative Analysis, Trends and Forecast, 2023 – 2031
Industry Trends
Research to assess the advantages and disadvantages of developing cancer therapies now heavily relies on real-world evidence (RWE). Finding alternatives for clinical development routes when clinical trials are not feasible is becoming increasingly crucial with the rise of uncommon and aggressive malignancies classified by molecular subtypes. RWE has become a significant additional resource that can offer knowledge beyond what is learnt through randomized clinical studies. RWE can offer comparative perspective for assessing findings about the security and effectiveness of pharmaceuticals. With observational, "lighter-touch" periods of follow-up that provide longer-term evaluation of patient outcomes, RWE is also enabling researchers to extend the follow-up of patients beyond the confines of a study.
Global RWE Oncology Market: Drivers
Randomized clinical trials have historically been used to provide light on novel medical remedies and are still the gold standard for approval. As patient level data has become more readily available in the real world, it is now feasible to produce evidence on the application and potential advantages or disadvantages of a medical therapy obtained from study of real-world data. RWE and randomized clinical trials are complimentary, and RWE is especially useful in oncology for guiding therapeutic decision-making along the patient journey. Key advantages include early cancer detection and diagnosis, the best available treatments (including personalized medication), and disease management options including dosage and side effect management.
Global RWE Oncology Market: Restraints
RWE's full potential is unrealized in emerging economies, because of structural and behavioral variables. Lack of regulatory involvement, accessibility, quality, and integrity of real-world data are examples of structural impediments. The slow understanding and acceptance of RWE are examples of behavioural obstacles among healthcare professionals. Healthcare stakeholders can work closely together to overcome these obstacles, but regulators should take the lead since they have the power to make it easier for RWE to be used in healthcare law and policy. Additionally, conducting RWE studies requires significant resources, including time, funding, data infrastructure, and expertise. The complexities involved in data collection, analysis, and interpretation can lead to delays and resource-intensive efforts. Collaboration among stakeholders and investment in infrastructure and analytical capabilities are necessary to overcome these constraints.
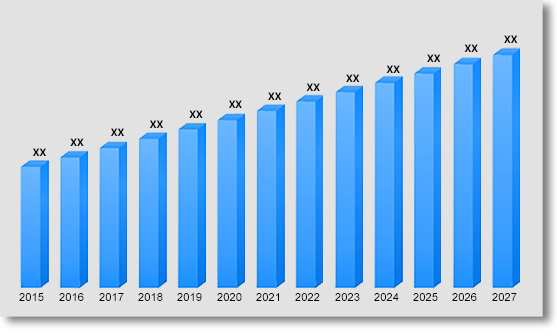
Global RWE Oncology Market Revenue & Forecast, (US$ Million), 2015 – 2031
Global RWE Oncology Market Component Analysis
The adoption of real-world evidence (RWE) in oncology can involve several components namely real-world datasets and real-world consulting & analytics services. The importance of real-world datasets lies in their ability to capture real-world clinical practice and patient experiences, allowing for a more comprehensive understanding of the effectiveness, safety, and cost-effectiveness of oncology treatments. These datasets provide the raw material for analysis and interpretation, enabling evidence-based decision-making. Robust and diverse real-world datasets are essential for generating reliable and generalizable RWE in oncology. Real-world consulting and analytics services complement the utilization of real-world datasets. These services involve strategic guidance, expertise, and analytical capabilities to extract meaningful insights from real-world data. For example, consulting services provide expertise in interpreting RWE findings and translating them into actionable recommendations for stakeholders, such as healthcare providers, payers, regulators, and pharmaceutical companies. They assist in understanding the implications of RWE for clinical decision-making, health policies, reimbursement strategies, and treatment guidelines.
Global RWE Oncology Market Regional Analysis
RWE generation in oncology has been increasingly common in recent years in the USA and Western Europe. Oncology patients in developing economies may now have the chance to gain from clinical decision making guided by RWE due to extensive biopharmaceutical investments in infrastructure that capture patient-level data and more comprehensive local regulatory guidelines. While the USA and EU are thought to be leading the way in exploiting RWE, interest in RWE is also increasing in emerging economies. Compared to the regions of Africa, the Middle East, and Latin America (LATAM), nations in the Asia-Pacific (APAC) area are setting the pace for RWE generation and adoption among rising economies. This growth can be attributed to the fact that many APAC countries, such as China, India, South Korea, and Singapore, have been increasing their investments in healthcare R&D. These investments include research on RWE generation, data analytics, and health technology assessment, fostering a favorable environment for RWE adoption and utilization.
Competitive Landscape
The report provides both, qualitative and quantitative research of global RWE oncology market, as well as provides comprehensive insights and development methods adopted by the key contenders. The report also offers extensive research on the key players in this market and details on the competitiveness of these players. Key business strategies such as mergers and acquisitions (M&A), affiliations, collaborations, and contracts adopted by these major market participants are also recognized and analyzed in the report. For each company, the report studies their global presence, competitors, service offerings and specification amongst others.
Some of the players operating in the global RWE oncology market are:
- Aetion, Inc.
- AstraZeneca
- EVERSANA
- Flatiron Health
- ICON plc
- IQVIA Holdings Inc
- MCKESSON CORPORATION
- Medpace
- Oracle
- Parexel International Corporation.
- Syapse, Inc.
- Syneos Health.
- Tempus Labs, Inc.
- Thermo Fisher Scientific Inc.
- Other Market Participants
Global RWE Oncology Market
By Component
- Real-world Datasets
- EMR/EHR/Clinical Data
- Administrative Claims & Billing Data
- Pharmacy Data
- Cancer Registries
- Specialty Data Providers
- Others
- Real-world Consulting & Analytics Services
By Application
- Clinical Trials
- Drug Development and approvals
- Post marketing safety studies
- Market Access and Reimbursement
- Medical device development
- Others
By End Users
- Biotechnology companies
- Medical device companies
- Healthcare Payers
- Healthcare Providers
- Others
- North America (U.S., Canada, Mexico, Rest of North America)
- Europe (France, The UK, Spain, Germany, Italy, Nordic Countries (Denmark, Finland, Iceland, Sweden, Norway), Benelux Union (Belgium, The Netherlands, Luxembourg), Rest of Europe)
- Asia Pacific (China, Japan, India, New Zealand, Australia, South Korea, Southeast Asia (Indonesia, Thailand, Malaysia, Singapore, Rest of Southeast Asia), Rest of Asia Pacific)
- Middle East & Africa (Saudi Arabia, UAE, Egypt, Kuwait, South Africa, Rest of Middle East & Africa)
- Latin America (Brazil, Argentina, Rest of Latin America)
Table of Contents
1. Market Scope
1.1. Market
Segmentation
1.2. Years
Considered
1.2.1. Historic
Years: 2015 - 2021
1.2.2. Base
Year: 2022
1.2.3. Forecast
Years: 2023 – 2031
2. Key Target Audiences
3. Research Methodology
3.1. Primary
Research
3.1.1. Research
Questionnaire
3.1.2. Global
Percentage Breakdown
3.1.3. Primary
Interviews: Key Opinion Leaders (KOLs)
3.2. Secondary
Research
3.2.1. Paid
Databases
3.2.2. Secondary
Sources
3.3. Market
Size Estimates
3.3.1. Top-Down
Approach
3.3.2. Bottom-Up
Approach
3.4. Data
Triangulation Methodology
3.5. Research
Assumptions
4. Recommendations and Insights from AMI’s Perspective**
5. Holistic Overview of RWE Oncology Market
6. Market Synopsis: RWE
Oncology Market
7. RWE Oncology Market Analysis: Qualitative Perspective
7.1. Introduction
7.1.1. Product
Definition
7.1.2. Industry
Development
7.2. Market
Dynamics
7.2.1. Drivers
7.2.2. Restraints
7.2.3. Opportunities
7.3. Trends in RWE
Oncology Market
7.4. Market
Determinants Radar Chart
7.5. Macro-Economic
and Micro-Economic Indicators: RWE Oncology Market
7.6. Porter’s
Five Force Analysis
7.7. Impact of
Covid-19 on RWE Oncology Market
8. Global RWE Oncology Market Analysis and Forecasts, 2023 –
2031
8.1. Overview
8.1.1. Global RWE
Oncology Market Revenue (US$ Mn)
8.2. Global RWE
Oncology Market Revenue (US$ Mn) and Forecasts, By Component
8.2.1. Real-world
Datasets (Definition, Market Estimation and Penetration, 2015 - 2022, Market
Estimation (2015 - 2022), Market Forecast (2023 - 2031), Compound Annual Growth
Rate (CAGR), Regional Bifurcation (North America, Europe, Asia Pacific, Middle
East and Africa, Latin America) and Information on EMR/EHR/Clinical Data,
Administrative Claims and Billing Data, Pharmacy Data, Cancer Registries,
Specialty Data Providers, Others)
8.2.1.1. EMR/EHR/Clinical Data
8.2.1.2. Administrative Claims and Billing Data
8.2.1.3. Pharmacy Data
8.2.1.4. Cancer Registries
8.2.1.5. Specialty Data Providers
8.2.1.6. Others
8.2.2. Real-world
Consulting and Analytics Services
8.2.2.1. Definition
8.2.2.2. Market Estimation and Penetration, 2015 - 2022
8.2.2.3. Market Forecast, 2023 - 2031
8.2.2.4. Compound Annual Growth Rate (CAGR)
8.2.2.5. Regional Bifurcation
8.2.2.5.1. North America
8.2.2.5.1.1. Market
Estimation, 2015 - 2022
8.2.2.5.1.2. Market
Forecast, 2023 - 2031
8.2.2.5.2. Europe
8.2.2.5.2.1. Market
Estimation, 2015 - 2022
8.2.2.5.2.2. Market
Forecast, 2023 - 2031
8.2.2.5.3. Asia Pacific
8.2.2.5.3.1. Market
Estimation, 2015 - 2022
8.2.2.5.3.2. Market
Forecast, 2023 - 2031
8.2.2.5.4. Middle East and Africa
8.2.2.5.4.1. Market
Estimation, 2015 - 2022
8.2.2.5.4.2. Market
Forecast, 2023 - 2031
8.2.2.5.5. Latin America
8.2.2.5.5.1. Market
Estimation, 2015 - 2022
8.2.2.5.5.2. Market
Forecast, 2023 - 2031
8.3. Key
Segment for Channeling Investments
8.3.1. By
Component
9. Global RWE Oncology Market Analysis and Forecasts, 2023 –
2031
9.1. Overview
9.2. Global RWE
Oncology Market Revenue (US$ Mn) and Forecasts, By Application
9.2.1. Clinical
Trials
9.2.1.1. Definition
9.2.1.2. Market Estimation and Penetration, 2015 - 2022
9.2.1.3. Market Forecast, 2023 - 2031
9.2.1.4. Compound Annual Growth Rate (CAGR)
9.2.1.5. Regional Bifurcation
9.2.1.5.1. North America
9.2.1.5.1.1. Market
Estimation, 2015 - 2022
9.2.1.5.1.2. Market
Forecast, 2023 - 2031
9.2.1.5.2. Europe
9.2.1.5.2.1. Market
Estimation, 2015 - 2022
9.2.1.5.2.2. Market
Forecast, 2023 - 2031
9.2.1.5.3. Asia Pacific
9.2.1.5.3.1. Market
Estimation, 2015 - 2022
9.2.1.5.3.2. Market
Forecast, 2023 - 2031
9.2.1.5.4. Middle East and Africa
9.2.1.5.4.1. Market
Estimation, 2015 - 2022
9.2.1.5.4.2. Market
Forecast, 2023 - 2031
9.2.1.5.5. Latin America
9.2.1.5.5.1. Market
Estimation, 2015 - 2022
9.2.1.5.5.2. Market
Forecast, 2023 - 2031
9.2.2. Drug
Development and approvals
9.2.2.1. Definition
9.2.2.2. Market Estimation and Penetration, 2015 - 2022
9.2.2.3. Market Forecast, 2023 - 2031
9.2.2.4. Compound Annual Growth Rate (CAGR)
9.2.2.5. Regional Bifurcation
9.2.2.5.1. North America
9.2.2.5.1.1. Market
Estimation, 2015 - 2022
9.2.2.5.1.2. Market
Forecast, 2023 - 2031
9.2.2.5.2. Europe
9.2.2.5.2.1. Market
Estimation, 2015 - 2022
9.2.2.5.2.2. Market
Forecast, 2023 - 2031
9.2.2.5.3. Asia Pacific
9.2.2.5.3.1. Market
Estimation, 2015 - 2022
9.2.2.5.3.2. Market
Forecast, 2023 - 2031
9.2.2.5.4. Middle East and Africa
9.2.2.5.4.1. Market
Estimation, 2015 - 2022
9.2.2.5.4.2. Market
Forecast, 2023 - 2031
9.2.2.5.5. Latin America
9.2.2.5.5.1. Market
Estimation, 2015 - 2022
9.2.2.5.5.2. Market
Forecast, 2023 - 2031
9.2.3. Post
marketing safety studies
9.2.3.1. Definition
9.2.3.2. Market Estimation and Penetration, 2015 - 2022
9.2.3.3. Market Forecast, 2023 - 2031
9.2.3.4. Compound Annual Growth Rate (CAGR)
9.2.3.5. Regional Bifurcation
9.2.3.5.1. North America
9.2.3.5.1.1. Market
Estimation, 2015 - 2022
9.2.3.5.1.2. Market
Forecast, 2023 - 2031
9.2.3.5.2. Europe
9.2.3.5.2.1. Market
Estimation, 2015 - 2022
9.2.3.5.2.2. Market
Forecast, 2023 - 2031
9.2.3.5.3. Asia Pacific
9.2.3.5.3.1. Market
Estimation, 2015 - 2022
9.2.3.5.3.2. Market
Forecast, 2023 - 2031
9.2.3.5.4. Middle East and Africa
9.2.3.5.4.1. Market
Estimation, 2015 - 2022
9.2.3.5.4.2. Market
Forecast, 2023 - 2031
9.2.3.5.5. Latin America
9.2.3.5.5.1. Market
Estimation, 2015 - 2022
9.2.3.5.5.2. Market
Forecast, 2023 - 2031
9.2.4. Market
Access and Reimbursement
9.2.4.1. Definition
9.2.4.2. Market Estimation and Penetration, 2015 - 2022
9.2.4.3. Market Forecast, 2023 - 2031
9.2.4.4. Compound Annual Growth Rate (CAGR)
9.2.4.5. Regional Bifurcation
9.2.4.5.1. North America
9.2.4.5.1.1. Market
Estimation, 2015 - 2022
9.2.4.5.1.2. Market
Forecast, 2023 - 2031
9.2.4.5.2. Europe
9.2.4.5.2.1. Market
Estimation, 2015 - 2022
9.2.4.5.2.2. Market
Forecast, 2023 - 2031
9.2.4.5.3. Asia Pacific
9.2.4.5.3.1. Market
Estimation, 2015 - 2022
9.2.4.5.3.2. Market
Forecast, 2023 - 2031
9.2.4.5.4. Middle East and Africa
9.2.4.5.4.1. Market
Estimation, 2015 - 2022
9.2.4.5.4.2. Market
Forecast, 2023 - 2031
9.2.4.5.5. Latin America
9.2.4.5.5.1. Market
Estimation, 2015 - 2022
9.2.4.5.5.2. Market
Forecast, 2023 - 2031
9.2.5. Medical
device development
9.2.5.1. Definition
9.2.5.2. Market Estimation and Penetration, 2015 - 2022
9.2.5.3. Market Forecast, 2023 - 2031
9.2.5.4. Compound Annual Growth Rate (CAGR)
9.2.5.5. Regional Bifurcation
9.2.5.5.1. North America
9.2.5.5.1.1. Market
Estimation, 2015 - 2022
9.2.5.5.1.2. Market
Forecast, 2023 - 2031
9.2.5.5.2. Europe
9.2.5.5.2.1. Market
Estimation, 2015 - 2022
9.2.5.5.2.2. Market
Forecast, 2023 - 2031
9.2.5.5.3. Asia Pacific
9.2.5.5.3.1. Market
Estimation, 2015 - 2022
9.2.5.5.3.2. Market
Forecast, 2023 - 2031
9.2.5.5.4. Middle East and Africa
9.2.5.5.4.1. Market
Estimation, 2015 - 2022
9.2.5.5.4.2. Market
Forecast, 2023 - 2031
9.2.5.5.5. Latin America
9.2.5.5.5.1. Market
Estimation, 2015 - 2022
9.2.5.5.5.2. Market
Forecast, 2023 - 2031
9.2.6. Others
9.2.6.1. Definition
9.2.6.2. Market Estimation and Penetration, 2015 - 2022
9.2.6.3. Market Forecast, 2023 - 2031
9.2.6.4. Compound Annual Growth Rate (CAGR)
9.2.6.5. Regional Bifurcation
9.2.6.5.1. North America
9.2.6.5.1.1. Market
Estimation, 2015 - 2022
9.2.6.5.1.2. Market
Forecast, 2023 - 2031
9.2.6.5.2. Europe
9.2.6.5.2.1. Market
Estimation, 2015 - 2022
9.2.6.5.2.2. Market
Forecast, 2023 - 2031
9.2.6.5.3. Asia Pacific
9.2.6.5.3.1. Market
Estimation, 2015 - 2022
9.2.6.5.3.2. Market
Forecast, 2023 - 2031
9.2.6.5.4. Middle East and Africa
9.2.6.5.4.1. Market
Estimation, 2015 - 2022
9.2.6.5.4.2. Market
Forecast, 2023 - 2031
9.2.6.5.5. Latin America
9.2.6.5.5.1. Market
Estimation, 2015 - 2022
9.2.6.5.5.2. Market
Forecast, 2023 - 2031
9.3. Key
Segment for Channeling Investments
9.3.1. By
Application
10. Global RWE Oncology Market Analysis and Forecasts, 2023 –
2031
10.1. Overview
10.2. Global RWE
Oncology Market Revenue (US$ Mn) and Forecasts, By End Users
10.2.1. Biotechnology
companies
10.2.1.1. Definition
10.2.1.2. Market Estimation and Penetration, 2015 - 2022
10.2.1.3. Market Forecast, 2023 - 2031
10.2.1.4. Compound Annual Growth Rate (CAGR)
10.2.1.5. Regional Bifurcation
10.2.1.5.1. North America
10.2.1.5.1.1. Market
Estimation, 2015 - 2022
10.2.1.5.1.2. Market
Forecast, 2023 - 2031
10.2.1.5.2. Europe
10.2.1.5.2.1. Market
Estimation, 2015 - 2022
10.2.1.5.2.2. Market
Forecast, 2023 - 2031
10.2.1.5.3. Asia Pacific
10.2.1.5.3.1. Market
Estimation, 2015 - 2022
10.2.1.5.3.2. Market
Forecast, 2023 - 2031
10.2.1.5.4. Middle East and Africa
10.2.1.5.4.1. Market
Estimation, 2015 - 2022
10.2.1.5.4.2. Market
Forecast, 2023 - 2031
10.2.1.5.5. Latin America
10.2.1.5.5.1. Market
Estimation, 2015 - 2022
10.2.1.5.5.2. Market
Forecast, 2023 - 2031
10.2.2. Medical
device companies
10.2.2.1. Definition
10.2.2.2. Market Estimation and Penetration, 2015 - 2022
10.2.2.3. Market Forecast, 2023 - 2031
10.2.2.4. Compound Annual Growth Rate (CAGR)
10.2.2.5. Regional Bifurcation
10.2.2.5.1. North America
10.2.2.5.1.1. Market
Estimation, 2015 - 2022
10.2.2.5.1.2. Market
Forecast, 2023 - 2031
10.2.2.5.2. Europe
10.2.2.5.2.1. Market
Estimation, 2015 - 2022
10.2.2.5.2.2. Market
Forecast, 2023 - 2031
10.2.2.5.3. Asia Pacific
10.2.2.5.3.1. Market
Estimation, 2015 - 2022
10.2.2.5.3.2. Market
Forecast, 2023 - 2031
10.2.2.5.4. Middle East and Africa
10.2.2.5.4.1. Market
Estimation, 2015 - 2022
10.2.2.5.4.2. Market
Forecast, 2023 - 2031
10.2.2.5.5. Latin America
10.2.2.5.5.1. Market
Estimation, 2015 - 2022
10.2.2.5.5.2. Market
Forecast, 2023 - 2031
10.2.3. Healthcare
Payers
10.2.3.1. Definition
10.2.3.2. Market Estimation and Penetration, 2015 - 2022
10.2.3.3. Market Forecast, 2023 - 2031
10.2.3.4. Compound Annual Growth Rate (CAGR)
10.2.3.5. Regional Bifurcation
10.2.3.5.1. North America
10.2.3.5.1.1. Market
Estimation, 2015 - 2022
10.2.3.5.1.2. Market
Forecast, 2023 - 2031
10.2.3.5.2. Europe
10.2.3.5.2.1. Market
Estimation, 2015 - 2022
10.2.3.5.2.2. Market
Forecast, 2023 - 2031
10.2.3.5.3. Asia Pacific
10.2.3.5.3.1. Market
Estimation, 2015 - 2022
10.2.3.5.3.2. Market
Forecast, 2023 - 2031
10.2.3.5.4. Middle East and Africa
10.2.3.5.4.1. Market
Estimation, 2015 - 2022
10.2.3.5.4.2. Market
Forecast, 2023 - 2031
10.2.3.5.5. Latin America
10.2.3.5.5.1. Market
Estimation, 2015 - 2022
10.2.3.5.5.2. Market
Forecast, 2023 - 2031
10.2.4. Healthcare
Providers
10.2.4.1. Definition
10.2.4.2. Market Estimation and Penetration, 2015 - 2022
10.2.4.3. Market Forecast, 2023 - 2031
10.2.4.4. Compound Annual Growth Rate (CAGR)
10.2.4.5. Regional Bifurcation
10.2.4.5.1. North America
10.2.4.5.1.1. Market
Estimation, 2015 - 2022
10.2.4.5.1.2. Market
Forecast, 2023 - 2031
10.2.4.5.2. Europe
10.2.4.5.2.1. Market
Estimation, 2015 - 2022
10.2.4.5.2.2. Market
Forecast, 2023 - 2031
10.2.4.5.3. Asia Pacific
10.2.4.5.3.1. Market
Estimation, 2015 - 2022
10.2.4.5.3.2. Market
Forecast, 2023 - 2031
10.2.4.5.4. Middle East and Africa
10.2.4.5.4.1. Market
Estimation, 2015 - 2022
10.2.4.5.4.2. Market
Forecast, 2023 - 2031
10.2.4.5.5. Latin America
10.2.4.5.5.1. Market
Estimation, 2015 - 2022
10.2.4.5.5.2. Market
Forecast, 2023 - 2031
10.2.5. Others
10.2.5.1. Definition
10.2.5.2. Market Estimation and Penetration, 2015 - 2022
10.2.5.3. Market Forecast, 2023 - 2031
10.2.5.4. Compound Annual Growth Rate (CAGR)
10.2.5.5. Regional Bifurcation
10.2.5.5.1. North America
10.2.5.5.1.1. Market
Estimation, 2015 - 2022
10.2.5.5.1.2. Market
Forecast, 2023 - 2031
10.2.5.5.2. Europe
10.2.5.5.2.1. Market
Estimation, 2015 - 2022
10.2.5.5.2.2. Market
Forecast, 2023 - 2031
10.2.5.5.3. Asia Pacific
10.2.5.5.3.1. Market
Estimation, 2015 - 2022
10.2.5.5.3.2. Market
Forecast, 2023 - 2031
10.2.5.5.4. Middle East and Africa
10.2.5.5.4.1. Market
Estimation, 2015 - 2022
10.2.5.5.4.2. Market
Forecast, 2023 - 2031
10.2.5.5.5. Latin America
10.2.5.5.5.1. Market
Estimation, 2015 - 2022
10.2.5.5.5.2. Market
Forecast, 2023 - 2031
10.3. Key
Segment for Channeling Investments
10.3.1. By End Users
11. North America RWE Oncology Market Analysis and Forecasts,
2023 - 2031
11.1. Overview
11.1.1. North
America RWE Oncology Market Revenue (US$ Mn)
11.2. North
America RWE Oncology Market Revenue (US$ Mn) and Forecasts, By Component
11.2.1. Real-world
Datasets
11.2.1.1. EMR/EHR/Clinical Data
11.2.1.2. Administrative Claims and Billing Data
11.2.1.3. Pharmacy Data
11.2.1.4. Cancer Registries
11.2.1.5. Specialty Data Providers
11.2.1.6. Others
11.2.2. Real-world
Consulting and Analytics Services
11.3. North
America RWE Oncology Market Revenue (US$ Mn) and Forecasts, By Application
11.3.1. Clinical
Trials
11.3.2. Drug
Development and approvals
11.3.3. Post
marketing safety studies
11.3.4. Market
Access and Reimbursement
11.3.5. Medical
device development
11.3.6. Others
11.4. North
America RWE Oncology Market Revenue (US$ Mn) and Forecasts, By End Users
11.4.1. Biotechnology
companies
11.4.2. Medical
device companies
11.4.3. Healthcare
Payers
11.4.4. Healthcare
Providers
11.4.5. Others
11.5. North
America RWE Oncology Market Revenue (US$ Mn) and Forecasts, By Country
11.5.1. U.S
11.5.1.1. U.S RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Component
11.5.1.1.1. Real-world Datasets
11.5.1.1.1.1. EMR/EHR/Clinical
Data
11.5.1.1.1.2. Administrative
Claims and Billing Data
11.5.1.1.1.3. Pharmacy
Data
11.5.1.1.1.4. Cancer
Registries
11.5.1.1.1.5. Specialty
Data Providers
11.5.1.1.1.6. Others
11.5.1.1.2. Real-world Consulting and Analytics Services
11.5.1.2. U.S RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Application
11.5.1.2.1. Clinical Trials
11.5.1.2.2. Drug Development and approvals
11.5.1.2.3. Post marketing safety studies
11.5.1.2.4. Market Access and Reimbursement
11.5.1.2.5. Medical device development
11.5.1.2.6. Others
11.5.1.3. U.S RWE Oncology Market Revenue (US$ Mn) and Forecasts,
By End Users
11.5.1.3.1. Biotechnology companies
11.5.1.3.2. Medical device companies
11.5.1.3.3. Healthcare Payers
11.5.1.3.4. Healthcare Providers
11.5.1.3.5. Others
11.5.2. Canada
11.5.2.1. Canada RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Component
11.5.2.1.1. Real-world Datasets
11.5.2.1.1.1. EMR/EHR/Clinical
Data
11.5.2.1.1.2. Administrative
Claims and Billing Data
11.5.2.1.1.3. Pharmacy
Data
11.5.2.1.1.4. Cancer
Registries
11.5.2.1.1.5. Specialty
Data Providers
11.5.2.1.1.6. Others
11.5.2.1.2. Real-world Consulting and Analytics Services
11.5.2.2. Canada RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Application
11.5.2.2.1. Clinical Trials
11.5.2.2.2. Drug Development and approvals
11.5.2.2.3. Post marketing safety studies
11.5.2.2.4. Market Access and Reimbursement
11.5.2.2.5. Medical device development
11.5.2.2.6. Others
11.5.2.3. Canada RWE Oncology Market Revenue (US$ Mn) and Forecasts,
By End Users
11.5.2.3.1. Biotechnology companies
11.5.2.3.2. Medical device companies
11.5.2.3.3. Healthcare Payers
11.5.2.3.4. Healthcare Providers
11.5.2.3.5. Others
11.5.3. Mexico
11.5.3.1. Mexico RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Component
11.5.3.1.1. Real-world Datasets
11.5.3.1.1.1. EMR/EHR/Clinical
Data
11.5.3.1.1.2. Administrative
Claims and Billing Data
11.5.3.1.1.3. Pharmacy
Data
11.5.3.1.1.4. Cancer
Registries
11.5.3.1.1.5. Specialty
Data Providers
11.5.3.1.1.6. Others
11.5.3.1.2. Real-world Consulting and Analytics Services
11.5.3.2. Mexico RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Application
11.5.3.2.1. Clinical Trials
11.5.3.2.2. Drug Development and approvals
11.5.3.2.3. Post marketing safety studies
11.5.3.2.4. Market Access and Reimbursement
11.5.3.2.5. Medical device development
11.5.3.2.6. Others
11.5.3.3. Mexico RWE Oncology Market Revenue (US$ Mn) and Forecasts,
By End Users
11.5.3.3.1. Biotechnology companies
11.5.3.3.2. Medical device companies
11.5.3.3.3. Healthcare Payers
11.5.3.3.4. Healthcare Providers
11.5.3.3.5. Others
11.5.4. Rest of
North America
11.5.4.1. Rest of North America RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Component
11.5.4.1.1. Real-world Datasets
11.5.4.1.1.1. EMR/EHR/Clinical
Data
11.5.4.1.1.2. Administrative
Claims and Billing Data
11.5.4.1.1.3. Pharmacy
Data
11.5.4.1.1.4. Cancer
Registries
11.5.4.1.1.5. Specialty
Data Providers
11.5.4.1.1.6. Others
11.5.4.1.2. Real-world Consulting and Analytics Services
11.5.4.2. Rest of North America RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Application
11.5.4.2.1. Clinical Trials
11.5.4.2.2. Drug Development and approvals
11.5.4.2.3. Post marketing safety studies
11.5.4.2.4. Market Access and Reimbursement
11.5.4.2.5. Medical device development
11.5.4.2.6. Others
11.5.4.3. Rest of North America RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By End Users
11.5.4.3.1. Biotechnology companies
11.5.4.3.2. Medical device companies
11.5.4.3.3. Healthcare Payers
11.5.4.3.4. Healthcare Providers
11.5.4.3.5. Others
11.6. Key
Segment for Channeling Investments
11.6.1. By
Country
11.6.2. By
Component
11.6.3. By
Application
11.6.4. By End Users
12. Europe RWE Oncology Market Analysis and Forecasts, 2023 -
2031
12.1. Overview
12.1.1. Europe RWE
Oncology Market Revenue (US$ Mn)
12.2. Europe RWE
Oncology Market Revenue (US$ Mn) and Forecasts, By Component
12.2.1. Real-world
Datasets
12.2.1.1. EMR/EHR/Clinical Data
12.2.1.2. Administrative Claims and Billing Data
12.2.1.3. Pharmacy Data
12.2.1.4. Cancer Registries
12.2.1.5. Specialty Data Providers
12.2.1.6. Others
12.2.2. Real-world
Consulting and Analytics Services
12.3. Europe RWE
Oncology Market Revenue (US$ Mn) and Forecasts, By Application
12.3.1. Clinical
Trials
12.3.2. Drug
Development and approvals
12.3.3. Post
marketing safety studies
12.3.4. Market
Access and Reimbursement
12.3.5. Medical
device development
12.3.6. Others
12.4. Europe RWE
Oncology Market Revenue (US$ Mn) and Forecasts, By End Users
12.4.1. Biotechnology
companies
12.4.2. Medical
device companies
12.4.3. Healthcare
Payers
12.4.4. Healthcare
Providers
12.4.5. Others
12.5. Europe RWE
Oncology Market Revenue (US$ Mn) and Forecasts, By Country
12.5.1. France
12.5.1.1. France RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Component
12.5.1.1.1. Real-world Datasets
12.5.1.1.1.1. EMR/EHR/Clinical
Data
12.5.1.1.1.2. Administrative
Claims and Billing Data
12.5.1.1.1.3. Pharmacy
Data
12.5.1.1.1.4. Cancer
Registries
12.5.1.1.1.5. Specialty
Data Providers
12.5.1.1.1.6. Others
12.5.1.1.2. Real-world Consulting and Analytics Services
12.5.1.2. France RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Application
12.5.1.2.1. Clinical Trials
12.5.1.2.2. Drug Development and approvals
12.5.1.2.3. Post marketing safety studies
12.5.1.2.4. Market Access and Reimbursement
12.5.1.2.5. Medical device development
12.5.1.2.6. Others
12.5.1.3. France RWE Oncology Market Revenue (US$ Mn) and Forecasts,
By End Users
12.5.1.3.1. Biotechnology companies
12.5.1.3.2. Medical device companies
12.5.1.3.3. Healthcare Payers
12.5.1.3.4. Healthcare Providers
12.5.1.3.5. Others
12.5.2. The UK
12.5.2.1. The UK RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Component
12.5.2.1.1. Real-world Datasets
12.5.2.1.1.1. EMR/EHR/Clinical
Data
12.5.2.1.1.2. Administrative
Claims and Billing Data
12.5.2.1.1.3. Pharmacy
Data
12.5.2.1.1.4. Cancer
Registries
12.5.2.1.1.5. Specialty
Data Providers
12.5.2.1.1.6. Others
12.5.2.1.2. Real-world Consulting and Analytics Services
12.5.2.2. The UK RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Application
12.5.2.2.1. Clinical Trials
12.5.2.2.2. Drug Development and approvals
12.5.2.2.3. Post marketing safety studies
12.5.2.2.4. Market Access and Reimbursement
12.5.2.2.5. Medical device development
12.5.2.2.6. Others
12.5.2.3. The UK RWE Oncology Market Revenue (US$ Mn) and Forecasts,
By End Users
12.5.2.3.1. Biotechnology companies
12.5.2.3.2. Medical device companies
12.5.2.3.3. Healthcare Payers
12.5.2.3.4. Healthcare Providers
12.5.2.3.5. Others
12.5.3. Spain
12.5.3.1. Spain RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Component
12.5.3.1.1. Real-world Datasets
12.5.3.1.1.1. EMR/EHR/Clinical
Data
12.5.3.1.1.2. Administrative
Claims and Billing Data
12.5.3.1.1.3. Pharmacy
Data
12.5.3.1.1.4. Cancer
Registries
12.5.3.1.1.5. Specialty
Data Providers
12.5.3.1.1.6. Others
12.5.3.1.2. Real-world Consulting and Analytics Services
12.5.3.2. Spain RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Application
12.5.3.2.1. Clinical Trials
12.5.3.2.2. Drug Development and approvals
12.5.3.2.3. Post marketing safety studies
12.5.3.2.4. Market Access and Reimbursement
12.5.3.2.5. Medical device development
12.5.3.2.6. Others
12.5.3.3. Spain RWE Oncology Market Revenue (US$ Mn) and Forecasts,
By End Users
12.5.3.3.1. Biotechnology companies
12.5.3.3.2. Medical device companies
12.5.3.3.3. Healthcare Payers
12.5.3.3.4. Healthcare Providers
12.5.3.3.5. Others
12.5.4. Germany
12.5.4.1. Germany RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Component
12.5.4.1.1. Real-world Datasets
12.5.4.1.1.1. EMR/EHR/Clinical
Data
12.5.4.1.1.2. Administrative
Claims and Billing Data
12.5.4.1.1.3. Pharmacy
Data
12.5.4.1.1.4. Cancer
Registries
12.5.4.1.1.5. Specialty
Data Providers
12.5.4.1.1.6. Others
12.5.4.1.2. Real-world Consulting and Analytics Services
12.5.4.2. Germany RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Application
12.5.4.2.1. Clinical Trials
12.5.4.2.2. Drug Development and approvals
12.5.4.2.3. Post marketing safety studies
12.5.4.2.4. Market Access and Reimbursement
12.5.4.2.5. Medical device development
12.5.4.2.6. Others
12.5.4.3. Germany RWE Oncology Market Revenue (US$ Mn) and Forecasts, By End Users
12.5.4.3.1. Biotechnology companies
12.5.4.3.2. Medical device companies
12.5.4.3.3. Healthcare Payers
12.5.4.3.4. Healthcare Providers
12.5.4.3.5. Others
12.5.5. Italy
12.5.5.1. Italy RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Component
12.5.5.1.1. Real-world Datasets
12.5.5.1.1.1. EMR/EHR/Clinical
Data
12.5.5.1.1.2. Administrative
Claims and Billing Data
12.5.5.1.1.3. Pharmacy
Data
12.5.5.1.1.4. Cancer
Registries
12.5.5.1.1.5. Specialty
Data Providers
12.5.5.1.1.6. Others
12.5.5.1.2. Real-world Consulting and Analytics Services
12.5.5.2. Italy RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Application
12.5.5.2.1. Clinical Trials
12.5.5.2.2. Drug Development and approvals
12.5.5.2.3. Post marketing safety studies
12.5.5.2.4. Market Access and Reimbursement
12.5.5.2.5. Medical device development
12.5.5.2.6. Others
12.5.5.3. Italy RWE Oncology Market Revenue (US$ Mn) and Forecasts,
By End Users
12.5.5.3.1. Biotechnology companies
12.5.5.3.2. Medical device companies
12.5.5.3.3. Healthcare Payers
12.5.5.3.4. Healthcare Providers
12.5.5.3.5. Others
12.5.6. Nordic
Countries
12.5.6.1. Nordic Countries RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Component
12.5.6.1.1. Real-world Datasets
12.5.6.1.1.1. EMR/EHR/Clinical
Data
12.5.6.1.1.2. Administrative
Claims and Billing Data
12.5.6.1.1.3. Pharmacy
Data
12.5.6.1.1.4. Cancer
Registries
12.5.6.1.1.5. Specialty
Data Providers
12.5.6.1.1.6. Others
12.5.6.1.2. Real-world Consulting and Analytics Services
12.5.6.2. Nordic Countries RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Application
12.5.6.2.1. Clinical Trials
12.5.6.2.2. Drug Development and approvals
12.5.6.2.3. Post marketing safety studies
12.5.6.2.4. Market Access and Reimbursement
12.5.6.2.5. Medical device development
12.5.6.2.6. Others
12.5.6.3. Nordic Countries RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By End Users
12.5.6.3.1. Biotechnology companies
12.5.6.3.2. Medical device companies
12.5.6.3.3. Healthcare Payers
12.5.6.3.4. Healthcare Providers
12.5.6.3.5. Others
12.5.6.4. Nordic Countries RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Country
12.5.6.4.1. Denmark
12.5.6.4.2. Finland
12.5.6.4.3. Iceland
12.5.6.4.4. Sweden
12.5.6.4.5. Norway
12.5.7. Benelux
Union
12.5.7.1. Benelux Union RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Component
12.5.7.1.1. Real-world Datasets
12.5.7.1.1.1. EMR/EHR/Clinical
Data
12.5.7.1.1.2. Administrative
Claims and Billing Data
12.5.7.1.1.3. Pharmacy
Data
12.5.7.1.1.4. Cancer
Registries
12.5.7.1.1.5. Specialty
Data Providers
12.5.7.1.1.6. Others
12.5.7.1.2. Real-world Consulting and Analytics Services
12.5.7.2. Benelux Union RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Application
12.5.7.2.1. Clinical Trials
12.5.7.2.2. Drug Development and approvals
12.5.7.2.3. Post marketing safety studies
12.5.7.2.4. Market Access and Reimbursement
12.5.7.2.5. Medical device development
12.5.7.2.6. Others
12.5.7.3. Benelux Union RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By End Users
12.5.7.3.1. Biotechnology companies
12.5.7.3.2. Medical device companies
12.5.7.3.3. Healthcare Payers
12.5.7.3.4. Healthcare Providers
12.5.7.3.5. Others
12.5.7.4. Benelux Union RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Country
12.5.7.4.1. Belgium
12.5.7.4.2. The Netherlands
12.5.7.4.3. Luxembourg
12.5.8. Rest of
Europe
12.5.8.1. Rest of Europe RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Component
12.5.8.1.1. Real-world Datasets
12.5.8.1.1.1. EMR/EHR/Clinical
Data
12.5.8.1.1.2. Administrative
Claims and Billing Data
12.5.8.1.1.3. Pharmacy
Data
12.5.8.1.1.4. Cancer
Registries
12.5.8.1.1.5. Specialty
Data Providers
12.5.8.1.1.6. Others
12.5.8.1.2. Real-world Consulting and Analytics Services
12.5.8.2. Rest of Europe RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Application
12.5.8.2.1. Clinical Trials
12.5.8.2.2. Drug Development and approvals
12.5.8.2.3. Post marketing safety studies
12.5.8.2.4. Market Access and Reimbursement
12.5.8.2.5. Medical device development
12.5.8.2.6. Others
12.5.8.3. Rest of Europe RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By End Users
12.5.8.3.1. Biotechnology companies
12.5.8.3.2. Medical device companies
12.5.8.3.3. Healthcare Payers
12.5.8.3.4. Healthcare Providers
12.5.8.3.5. Others
12.6. Key
Segment for Channeling Investments
12.6.1. By
Country
12.6.2. By
Component
12.6.3. By
Application
12.6.4. By End Users
13. Asia Pacific RWE Oncology Market Analysis and Forecasts, 2023
- 2031
13.1. Overview
13.1.1. Asia
Pacific RWE Oncology Market Revenue (US$ Mn)
13.2. Asia
Pacific RWE Oncology Market Revenue (US$ Mn) and Forecasts, By Component
13.2.1. Real-world
Datasets
13.2.1.1. EMR/EHR/Clinical Data
13.2.1.2. Administrative Claims and Billing Data
13.2.1.3. Pharmacy Data
13.2.1.4. Cancer Registries
13.2.1.5. Specialty Data Providers
13.2.1.6. Others
13.2.2. Real-world
Consulting and Analytics Services
13.3. Asia
Pacific RWE Oncology Market Revenue (US$ Mn) and Forecasts, By Application
13.3.1. Clinical
Trials
13.3.2. Drug
Development and approvals
13.3.3. Post
marketing safety studies
13.3.4. Market
Access and Reimbursement
13.3.5. Medical
device development
13.3.6. Others
13.4. Asia
Pacific RWE Oncology Market Revenue (US$ Mn) and Forecasts, By End Users
13.4.1. Biotechnology
companies
13.4.2. Medical
device companies
13.4.3. Healthcare
Payers
13.4.4. Healthcare
Providers
13.4.5. Others
13.5. Asia
Pacific RWE Oncology Market Revenue (US$ Mn) and Forecasts, By Country
13.5.1. China
13.5.1.1. China RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Component
13.5.1.1.1. Real-world Datasets
13.5.1.1.1.1. EMR/EHR/Clinical
Data
13.5.1.1.1.2. Administrative
Claims and Billing Data
13.5.1.1.1.3. Pharmacy
Data
13.5.1.1.1.4. Cancer
Registries
13.5.1.1.1.5. Specialty
Data Providers
13.5.1.1.1.6. Others
13.5.1.1.2. Real-world Consulting and Analytics Services
13.5.1.2. China RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Application
13.5.1.2.1. Clinical Trials
13.5.1.2.2. Drug Development and approvals
13.5.1.2.3. Post marketing safety studies
13.5.1.2.4. Market Access and Reimbursement
13.5.1.2.5. Medical device development
13.5.1.2.6. Others
13.5.1.3. China RWE Oncology Market Revenue (US$ Mn) and Forecasts,
By End Users
13.5.1.3.1. Biotechnology companies
13.5.1.3.2. Medical device companies
13.5.1.3.3. Healthcare Payers
13.5.1.3.4. Healthcare Providers
13.5.1.3.5. Others
13.5.2. Japan
13.5.2.1. Japan RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Component
13.5.2.1.1. Real-world Datasets
13.5.2.1.1.1. EMR/EHR/Clinical
Data
13.5.2.1.1.2. Administrative
Claims and Billing Data
13.5.2.1.1.3. Pharmacy
Data
13.5.2.1.1.4. Cancer
Registries
13.5.2.1.1.5. Specialty
Data Providers
13.5.2.1.1.6. Others
13.5.2.1.2. Real-world Consulting and Analytics Services
13.5.2.2. Japan RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Application
13.5.2.2.1. Clinical Trials
13.5.2.2.2. Drug Development and approvals
13.5.2.2.3. Post marketing safety studies
13.5.2.2.4. Market Access and Reimbursement
13.5.2.2.5. Medical device development
13.5.2.2.6. Others
13.5.2.3. Japan RWE Oncology Market Revenue (US$ Mn) and Forecasts,
By End Users
13.5.2.3.1. Biotechnology companies
13.5.2.3.2. Medical device companies
13.5.2.3.3. Healthcare Payers
13.5.2.3.4. Healthcare Providers
13.5.2.3.5. Others
13.5.3. India
13.5.3.1. India RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Component
13.5.3.1.1. Real-world Datasets
13.5.3.1.1.1. EMR/EHR/Clinical
Data
13.5.3.1.1.2. Administrative
Claims and Billing Data
13.5.3.1.1.3. Pharmacy
Data
13.5.3.1.1.4. Cancer
Registries
13.5.3.1.1.5. Specialty
Data Providers
13.5.3.1.1.6. Others
13.5.3.1.2. Real-world Consulting and Analytics Services
13.5.3.2. India RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Application
13.5.3.2.1. Clinical Trials
13.5.3.2.2. Drug Development and approvals
13.5.3.2.3. Post marketing safety studies
13.5.3.2.4. Market Access and Reimbursement
13.5.3.2.5. Medical device development
13.5.3.2.6. Others
13.5.3.3. India RWE Oncology Market Revenue (US$ Mn) and Forecasts,
By End Users
13.5.3.3.1. Biotechnology companies
13.5.3.3.2. Medical device companies
13.5.3.3.3. Healthcare Payers
13.5.3.3.4. Healthcare Providers
13.5.3.3.5. Others
13.5.4. New
Zealand
13.5.4.1. New Zealand RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Component
13.5.4.1.1. Real-world Datasets
13.5.4.1.1.1. EMR/EHR/Clinical
Data
13.5.4.1.1.2. Administrative
Claims and Billing Data
13.5.4.1.1.3. Pharmacy
Data
13.5.4.1.1.4. Cancer
Registries
13.5.4.1.1.5. Specialty
Data Providers
13.5.4.1.1.6. Others
13.5.4.1.2. Real-world Consulting and Analytics Services
13.5.4.2. New Zealand RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Application
13.5.4.2.1. Clinical Trials
13.5.4.2.2. Drug Development and approvals
13.5.4.2.3. Post marketing safety studies
13.5.4.2.4. Market Access and Reimbursement
13.5.4.2.5. Medical device development
13.5.4.2.6. Others
13.5.4.3. New Zealand RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By End Users
13.5.4.3.1. Biotechnology companies
13.5.4.3.2. Medical device companies
13.5.4.3.3. Healthcare Payers
13.5.4.3.4. Healthcare Providers
13.5.4.3.5. Others
13.5.5. Australia
13.5.5.1. Australia RWE Oncology Market Revenue (US$ Mn) and Forecasts,
By Component
13.5.5.1.1. Real-world Datasets
13.5.5.1.1.1. EMR/EHR/Clinical
Data
13.5.5.1.1.2. Administrative
Claims and Billing Data
13.5.5.1.1.3. Pharmacy
Data
13.5.5.1.1.4. Cancer
Registries
13.5.5.1.1.5. Specialty
Data Providers
13.5.5.1.1.6. Others
13.5.5.1.2. Real-world Consulting and Analytics Services
13.5.5.2. Australia RWE Oncology Market Revenue (US$ Mn) and Forecasts,
By Application
13.5.5.2.1. Clinical Trials
13.5.5.2.2. Drug Development and approvals
13.5.5.2.3. Post marketing safety studies
13.5.5.2.4. Market Access and Reimbursement
13.5.5.2.5. Medical device development
13.5.5.2.6. Others
13.5.5.3. Australia RWE Oncology Market Revenue (US$ Mn) and Forecasts,
By End Users
13.5.5.3.1. Biotechnology companies
13.5.5.3.2. Medical device companies
13.5.5.3.3. Healthcare Payers
13.5.5.3.4. Healthcare Providers
13.5.5.3.5. Others
13.5.6. South
Korea
13.5.6.1. South Korea RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Component
13.5.6.1.1. Real-world Datasets
13.5.6.1.1.1. EMR/EHR/Clinical
Data
13.5.6.1.1.2. Administrative
Claims and Billing Data
13.5.6.1.1.3. Pharmacy
Data
13.5.6.1.1.4. Cancer
Registries
13.5.6.1.1.5. Specialty
Data Providers
13.5.6.1.1.6. Others
13.5.6.1.2. Real-world Consulting and Analytics Services
13.5.6.2. South Korea RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Application
13.5.6.2.1. Clinical Trials
13.5.6.2.2. Drug Development and approvals
13.5.6.2.3. Post marketing safety studies
13.5.6.2.4. Market Access and Reimbursement
13.5.6.2.5. Medical device development
13.5.6.2.6. Others
13.5.6.3. South Korea RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By End Users
13.5.6.3.1. Biotechnology companies
13.5.6.3.2. Medical device companies
13.5.6.3.3. Healthcare Payers
13.5.6.3.4. Healthcare Providers
13.5.6.3.5. Others
13.5.7. Southeast
Asia
13.5.7.1. Southeast Asia RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Component
13.5.7.1.1. Real-world Datasets
13.5.7.1.1.1. EMR/EHR/Clinical
Data
13.5.7.1.1.2. Administrative
Claims and Billing Data
13.5.7.1.1.3. Pharmacy
Data
13.5.7.1.1.4. Cancer
Registries
13.5.7.1.1.5. Specialty
Data Providers
13.5.7.1.1.6. Others
13.5.7.1.2. Real-world Consulting and Analytics Services
13.5.7.2. Southeast Asia RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Application
13.5.7.2.1. Clinical Trials
13.5.7.2.2. Drug Development and approvals
13.5.7.2.3. Post marketing safety studies
13.5.7.2.4. Market Access and Reimbursement
13.5.7.2.5. Medical device development
13.5.7.2.6. Others
13.5.7.3. Southeast Asia RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By End Users
13.5.7.3.1. Biotechnology companies
13.5.7.3.2. Medical device companies
13.5.7.3.3. Healthcare Payers
13.5.7.3.4. Healthcare Providers
13.5.7.3.5. Others
13.5.7.4. Southeast Asia RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Country
13.5.7.4.1. Indonesia
13.5.7.4.2. Thailand
13.5.7.4.3. Malaysia
13.5.7.4.4. Singapore
13.5.7.4.5. Rest of Southeast Asia
13.5.8. Rest of
Asia Pacific
13.5.8.1. Rest of Asia Pacific RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Component
13.5.8.1.1. Real-world Datasets
13.5.8.1.1.1. EMR/EHR/Clinical
Data
13.5.8.1.1.2. Administrative
Claims and Billing Data
13.5.8.1.1.3. Pharmacy
Data
13.5.8.1.1.4. Cancer
Registries
13.5.8.1.1.5. Specialty
Data Providers
13.5.8.1.1.6. Others
13.5.8.1.2. Real-world Consulting and Analytics Services
13.5.8.2. Rest of Asia Pacific RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Application
13.5.8.2.1. Clinical Trials
13.5.8.2.2. Drug Development and approvals
13.5.8.2.3. Post marketing safety studies
13.5.8.2.4. Market Access and Reimbursement
13.5.8.2.5. Medical device development
13.5.8.2.6. Others
13.5.8.3. Rest of Asia Pacific RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By End Users
13.5.8.3.1. Biotechnology companies
13.5.8.3.2. Medical device companies
13.5.8.3.3. Healthcare Payers
13.5.8.3.4. Healthcare Providers
13.5.8.3.5. Others
13.6. Key
Segment for Channeling Investments
13.6.1. By
Country
13.6.2. By
Component
13.6.3. By
Application
13.6.4. By End Users
14. Middle East and Africa RWE Oncology Market Analysis and
Forecasts, 2023 - 2031
14.1. Overview
14.1.1. Middle
East and Africa RWE Oncology Market Revenue (US$ Mn)
14.2. Middle
East and Africa RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Component
14.2.1. Real-world
Datasets
14.2.1.1. EMR/EHR/Clinical Data
14.2.1.2. Administrative Claims and Billing Data
14.2.1.3. Pharmacy Data
14.2.1.4. Cancer Registries
14.2.1.5. Specialty Data Providers
14.2.1.6. Others
14.2.2. Real-world
Consulting and Analytics Services
14.3. Middle
East and Africa RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Application
14.3.1. Clinical
Trials
14.3.2. Drug
Development and approvals
14.3.3. Post
marketing safety studies
14.3.4. Market
Access and Reimbursement
14.3.5. Medical
device development
14.3.6. Others
14.4. Middle
East and Africa RWE Oncology Market Revenue (US$ Mn) and Forecasts, By End Users
14.4.1. Biotechnology
companies
14.4.2. Medical
device companies
14.4.3. Healthcare
Payers
14.4.4. Healthcare
Providers
14.4.5. Others
14.5. Middle
East and Africa RWE Oncology Market Revenue (US$ Mn) and Forecasts, By Country
14.5.1. Saudi
Arabia
14.5.1.1. Saudi Arabia RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Component
14.5.1.1.1. Real-world Datasets
14.5.1.1.1.1. EMR/EHR/Clinical
Data
14.5.1.1.1.2. Administrative
Claims and Billing Data
14.5.1.1.1.3. Pharmacy
Data
14.5.1.1.1.4. Cancer
Registries
14.5.1.1.1.5. Specialty
Data Providers
14.5.1.1.1.6. Others
14.5.1.1.2. Real-world Consulting and Analytics Services
14.5.1.2. Saudi Arabia RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Application
14.5.1.2.1. Clinical Trials
14.5.1.2.2. Drug Development and approvals
14.5.1.2.3. Post marketing safety studies
14.5.1.2.4. Market Access and Reimbursement
14.5.1.2.5. Medical device development
14.5.1.2.6. Others
14.5.1.3. Saudi Arabia RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By End Users
14.5.1.3.1. Biotechnology companies
14.5.1.3.2. Medical device companies
14.5.1.3.3. Healthcare Payers
14.5.1.3.4. Healthcare Providers
14.5.1.3.5. Others
14.5.2. UAE
14.5.2.1. UAE RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Component
14.5.2.1.1. Real-world Datasets
14.5.2.1.1.1. EMR/EHR/Clinical
Data
14.5.2.1.1.2. Administrative
Claims and Billing Data
14.5.2.1.1.3. Pharmacy
Data
14.5.2.1.1.4. Cancer
Registries
14.5.2.1.1.5. Specialty
Data Providers
14.5.2.1.1.6. Others
14.5.2.1.2. Real-world Consulting and Analytics Services
14.5.2.2. UAE RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Application
14.5.2.2.1. Clinical Trials
14.5.2.2.2. Drug Development and approvals
14.5.2.2.3. Post marketing safety studies
14.5.2.2.4. Market Access and Reimbursement
14.5.2.2.5. Medical device development
14.5.2.2.6. Others
14.5.2.3. UAE RWE Oncology Market Revenue (US$ Mn) and Forecasts,
By End Users
14.5.2.3.1. Biotechnology companies
14.5.2.3.2. Medical device companies
14.5.2.3.3. Healthcare Payers
14.5.2.3.4. Healthcare Providers
14.5.2.3.5. Others
14.5.3. Egypt
14.5.3.1. Egypt RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Component
14.5.3.1.1. Real-world Datasets
14.5.3.1.1.1. EMR/EHR/Clinical
Data
14.5.3.1.1.2. Administrative
Claims and Billing Data
14.5.3.1.1.3. Pharmacy
Data
14.5.3.1.1.4. Cancer
Registries
14.5.3.1.1.5. Specialty
Data Providers
14.5.3.1.1.6. Others
14.5.3.1.2. Real-world Consulting and Analytics Services
14.5.3.2. Egypt RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Application
14.5.3.2.1. Clinical Trials
14.5.3.2.2. Drug Development and approvals
14.5.3.2.3. Post marketing safety studies
14.5.3.2.4. Market Access and Reimbursement
14.5.3.2.5. Medical device development
14.5.3.2.6. Others
14.5.3.3. Egypt RWE Oncology Market Revenue (US$ Mn) and Forecasts,
By End Users
14.5.3.3.1. Biotechnology companies
14.5.3.3.2. Medical device companies
14.5.3.3.3. Healthcare Payers
14.5.3.3.4. Healthcare Providers
14.5.3.3.5. Others
14.5.4. Kuwait
14.5.4.1. Kuwait RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Component
14.5.4.1.1. Real-world Datasets
14.5.4.1.1.1. EMR/EHR/Clinical
Data
14.5.4.1.1.2. Administrative
Claims and Billing Data
14.5.4.1.1.3. Pharmacy
Data
14.5.4.1.1.4. Cancer
Registries
14.5.4.1.1.5. Specialty
Data Providers
14.5.4.1.1.6. Others
14.5.4.1.2. Real-world Consulting and Analytics Services
14.5.4.2. Kuwait RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Application
14.5.4.2.1. Clinical Trials
14.5.4.2.2. Drug Development and approvals
14.5.4.2.3. Post marketing safety studies
14.5.4.2.4. Market Access and Reimbursement
14.5.4.2.5. Medical device development
14.5.4.2.6. Others
14.5.4.3. Kuwait RWE Oncology Market Revenue (US$ Mn) and Forecasts,
By End Users
14.5.4.3.1. Biotechnology companies
14.5.4.3.2. Medical device companies
14.5.4.3.3. Healthcare Payers
14.5.4.3.4. Healthcare Providers
14.5.4.3.5. Others
14.5.5. South
Africa
14.5.5.1. South Africa RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Component
14.5.5.1.1. Real-world Datasets
14.5.5.1.1.1. EMR/EHR/Clinical
Data
14.5.5.1.1.2. Administrative
Claims and Billing Data
14.5.5.1.1.3. Pharmacy
Data
14.5.5.1.1.4. Cancer
Registries
14.5.5.1.1.5. Specialty
Data Providers
14.5.5.1.1.6. Others
14.5.5.1.2. Real-world Consulting and Analytics Services
14.5.5.2. South Africa RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Application
14.5.5.2.1. Clinical Trials
14.5.5.2.2. Drug Development and approvals
14.5.5.2.3. Post marketing safety studies
14.5.5.2.4. Market Access and Reimbursement
14.5.5.2.5. Medical device development
14.5.5.2.6. Others
14.5.5.3. South Africa RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By End Users
14.5.5.3.1. Biotechnology companies
14.5.5.3.2. Medical device companies
14.5.5.3.3. Healthcare Payers
14.5.5.3.4. Healthcare Providers
14.5.5.3.5. Others
14.5.6. Rest of
Middle East & Africa
14.5.6.1. Rest of Middle East & Africa RWE Oncology Market Revenue
(US$ Mn) and Forecasts, By Component
14.5.6.1.1. Real-world Datasets
14.5.6.1.1.1. EMR/EHR/Clinical
Data
14.5.6.1.1.2. Administrative
Claims and Billing Data
14.5.6.1.1.3. Pharmacy
Data
14.5.6.1.1.4. Cancer
Registries
14.5.6.1.1.5. Specialty
Data Providers
14.5.6.1.1.6. Others
14.5.6.1.2. Real-world Consulting and Analytics Services
14.5.6.2. Rest of Middle East & Africa RWE Oncology Market Revenue
(US$ Mn) and Forecasts, By Application
14.5.6.2.1. Clinical Trials
14.5.6.2.2. Drug Development and approvals
14.5.6.2.3. Post marketing safety studies
14.5.6.2.4. Market Access and Reimbursement
14.5.6.2.5. Medical device development
14.5.6.2.6. Others
14.5.6.3. Rest of Middle East & Africa RWE Oncology Market Revenue
(US$ Mn) and Forecasts, By End Users
14.5.6.3.1. Biotechnology companies
14.5.6.3.2. Medical device companies
14.5.6.3.3. Healthcare Payers
14.5.6.3.4. Healthcare Providers
14.5.6.3.5. Others
14.6. Key
Segment for Channeling Investments
14.6.1. By
Country
14.6.2. By
Component
14.6.3. By
Application
14.6.4. By End Users
15. Latin America RWE Oncology Market Analysis and Forecasts,
2023 - 2031
15.1. Overview
15.1.1. Latin
America RWE Oncology Market Revenue (US$ Mn)
15.2. Latin
America RWE Oncology Market Revenue (US$ Mn) and Forecasts, By Component
15.2.1. Real-world
Datasets
15.2.1.1. EMR/EHR/Clinical Data
15.2.1.2. Administrative Claims and Billing Data
15.2.1.3. Pharmacy Data
15.2.1.4. Cancer Registries
15.2.1.5. Specialty Data Providers
15.2.1.6. Others
15.2.2. Real-world
Consulting and Analytics Services
15.3. Latin
America RWE Oncology Market Revenue (US$ Mn) and Forecasts, By Application
15.3.1. Clinical
Trials
15.3.2. Drug
Development and approvals
15.3.3. Post
marketing safety studies
15.3.4. Market
Access and Reimbursement
15.3.5. Medical
device development
15.3.6. Others
15.4. Latin
America RWE Oncology Market Revenue (US$ Mn) and Forecasts, By End Users
15.4.1. Biotechnology
companies
15.4.2. Medical
device companies
15.4.3. Healthcare
Payers
15.4.4. Healthcare
Providers
15.4.5. Others
15.5. Latin
America RWE Oncology Market Revenue (US$ Mn) and Forecasts, By Country
15.5.1. Brazil
15.5.1.1. Brazil RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Component
15.5.1.1.1. Real-world Datasets
15.5.1.1.1.1. EMR/EHR/Clinical
Data
15.5.1.1.1.2. Administrative
Claims and Billing Data
15.5.1.1.1.3. Pharmacy
Data
15.5.1.1.1.4. Cancer
Registries
15.5.1.1.1.5. Specialty
Data Providers
15.5.1.1.1.6. Others
15.5.1.1.2. Real-world Consulting and Analytics Services
15.5.1.2. Brazil RWE Oncology Market Revenue (US$ Mn) and Forecasts, By
Application
15.5.1.2.1. Clinical Trials
15.5.1.2.2. Drug Development and approvals
15.5.1.2.3. Post marketing safety studies
15.5.1.2.4. Market Access and Reimbursement
15.5.1.2.5. Medical device development
15.5.1.2.6. Others
15.5.1.3. Brazil RWE Oncology Market Revenue (US$ Mn) and Forecasts,
By End Users
15.5.1.3.1. Biotechnology companies
15.5.1.3.2. Medical device companies
15.5.1.3.3. Healthcare Payers
15.5.1.3.4. Healthcare Providers
15.5.1.3.5. Others
15.5.2. Argentina
15.5.2.1. Argentina RWE Oncology Market Revenue (US$ Mn) and Forecasts,
By Component
15.5.2.1.1. Real-world Datasets
15.5.2.1.1.1. EMR/EHR/Clinical
Data
15.5.2.1.1.2. Administrative
Claims and Billing Data
15.5.2.1.1.3. Pharmacy
Data
15.5.2.1.1.4. Cancer
Registries
15.5.2.1.1.5. Specialty
Data Providers
15.5.2.1.1.6. Others
15.5.2.1.2. Real-world Consulting and Analytics Services
15.5.2.2. Argentina RWE Oncology Market Revenue (US$ Mn) and Forecasts,
By Application
15.5.2.2.1. Clinical Trials
15.5.2.2.2. Drug Development and approvals
15.5.2.2.3. Post marketing safety studies
15.5.2.2.4. Market Access and Reimbursement
15.5.2.2.5. Medical device development
15.5.2.2.6. Others
15.5.2.3. Argentina RWE Oncology Market Revenue (US$ Mn) and Forecasts,
By End Users
15.5.2.3.1. Biotechnology companies
15.5.2.3.2. Medical device companies
15.5.2.3.3. Healthcare Payers
15.5.2.3.4. Healthcare Providers
15.5.2.3.5. Others
15.5.3. Rest of
Latin America
15.5.3.1. Rest of Latin America RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Component
15.5.3.1.1. Real-world Datasets
15.5.3.1.1.1. EMR/EHR/Clinical
Data
15.5.3.1.1.2. Administrative
Claims and Billing Data
15.5.3.1.1.3. Pharmacy
Data
15.5.3.1.1.4. Cancer
Registries
15.5.3.1.1.5. Specialty
Data Providers
15.5.3.1.1.6. Others
15.5.3.1.2. Real-world Consulting and Analytics Services
15.5.3.2. Rest of Latin America RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By Application
15.5.3.2.1. Clinical Trials
15.5.3.2.2. Drug Development and approvals
15.5.3.2.3. Post marketing safety studies
15.5.3.2.4. Market Access and Reimbursement
15.5.3.2.5. Medical device development
15.5.3.2.6. Others
15.5.3.3. Rest of Latin America RWE Oncology Market Revenue (US$ Mn) and
Forecasts, By End Users
15.5.3.3.1. Biotechnology companies
15.5.3.3.2. Medical device companies
15.5.3.3.3. Healthcare Payers
15.5.3.3.4. Healthcare Providers
15.5.3.3.5. Others
15.6. Key
Segment for Channeling Investments
15.6.1. By
Country
15.6.2. By
Component
15.6.3. By
Application
15.6.4. By End Users
16. Competitive Benchmarking
16.1. Market Share
Analysis, 2022
16.2. Global
Presence and Growth Strategies
16.2.1. Mergers
and Acquisitions
16.2.2. Product
Launches
16.2.3. Investments
Trends
16.2.4. R&D
Initiatives
17. Player Profiles
17.1. Aetion,
Inc.
17.1.1. Company
Details
17.1.2. Company
Overview
17.1.3. Product
Offerings
17.1.4. Key
Developments
17.1.5. Financial
Analysis
17.1.6. SWOT Analysis
17.1.7. Business Strategies
17.2. AstraZeneca
17.2.1. Company
Details
17.2.2. Company
Overview
17.2.3. Product
Offerings
17.2.4. Key
Developments
17.2.5. Financial
Analysis
17.2.6. SWOT
Analysis
17.2.7. Business
Strategies
17.3. EVERSANA
17.3.1. Company
Details
17.3.2. Company
Overview
17.3.3. Product
Offerings
17.3.4. Key
Developments
17.3.5. Financial
Analysis
17.3.6. SWOT
Analysis
17.3.7. Business
Strategies
17.4. Flatiron
Health
17.4.1. Company
Details
17.4.2. Company
Overview
17.4.3. Product
Offerings
17.4.4. Key
Developments
17.4.5. Financial
Analysis
17.4.6. SWOT
Analysis
17.4.7. Business Strategies
17.5. ICON plc
17.5.1. Company
Details
17.5.2. Company
Overview
17.5.3. Product
Offerings
17.5.4. Key
Developments
17.5.5. Financial
Analysis
17.5.6. SWOT
Analysis
17.5.7. Business
Strategies
17.6. IQVIA
Holdings Inc
17.6.1. Company
Details
17.6.2. Company
Overview
17.6.3. Product
Offerings
17.6.4. Key
Developments
17.6.5. Financial
Analysis
17.6.6. SWOT
Analysis
17.6.7. Business
Strategies
17.7. MCKESSON
CORPORATION
17.7.1. Company
Details
17.7.2. Company
Overview
17.7.3. Product
Offerings
17.7.4. Key
Developments
17.7.5. Financial
Analysis
17.7.6. SWOT
Analysis
17.7.7. Business Strategies
17.8. Medpace
17.8.1. Company
Details
17.8.2. Company
Overview
17.8.3. Product
Offerings
17.8.4. Key
Developments
17.8.5. Financial
Analysis
17.8.6. SWOT
Analysis
17.8.7. Business
Strategies
17.9. Oracle
17.9.1. Company
Details
17.9.2. Company
Overview
17.9.3. Product
Offerings
17.9.4. Key
Developments
17.9.5. Financial
Analysis
17.9.6. SWOT
Analysis
17.9.7. Business
Strategies
17.10. Parexel
International Corporation.
17.10.1. Company
Details
17.10.2. Company
Overview
17.10.3. Product
Offerings
17.10.4. Key
Developments
17.10.5. Financial
Analysis
17.10.6. SWOT
Analysis
17.10.7. Business
Strategies
17.11. Syapse, Inc.
17.11.1. Company
Details
17.11.2. Company
Overview
17.11.3. Product
Offerings
17.11.4. Key
Developments
17.11.5. Financial
Analysis
17.11.6. SWOT
Analysis
17.11.7. Business Strategies
17.12. Syneos
Health.
17.12.1. Company
Details
17.12.2. Company
Overview
17.12.3. Product
Offerings
17.12.4. Key
Developments
17.12.5. Financial
Analysis
17.12.6. SWOT
Analysis
17.12.7. Business
Strategies
17.13. Tempus
Labs Inc.
17.13.1. Company
Details
17.13.2. Company
Overview
17.13.3. Product
Offerings
17.13.4. Key
Developments
17.13.5. Financial
Analysis
17.13.6. SWOT
Analysis
17.13.7. Business Strategies
17.14. Thermo
Fisher Scientific Inc.
17.14.1. Company
Details
17.14.2. Company
Overview
17.14.3. Product
Offerings
17.14.4. Key
Developments
17.14.5. Financial
Analysis
17.14.6. SWOT
Analysis
17.14.7. Business
Strategies
17.15. Other Market
Participants
18. Key Findings
**Exclusive for Multi-User and Enterprise User.
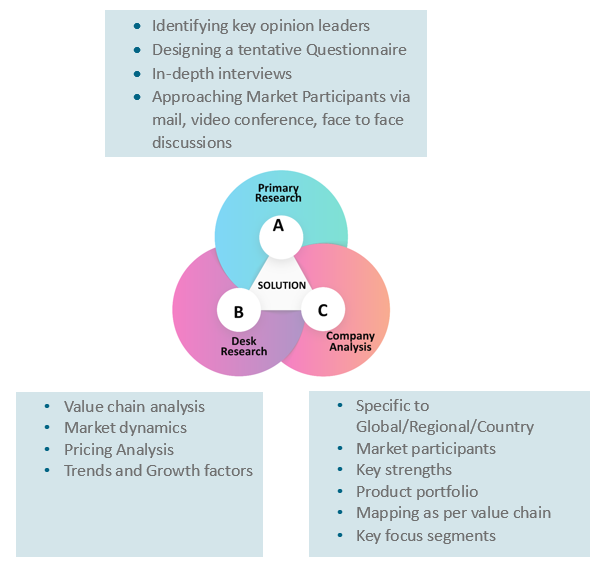
At Absolute Markets Insights, we are engaged in building both global as well as country specific reports. As a result, the approach taken for deriving the estimation and forecast for a specific country is a bit unique and different in comparison to the global research studies. In this case, we not only study the concerned market factors & trends prevailing in a particular country (from secondary research) but we also tend to calculate the actual market size & forecast from the revenue generated from the market participants involved in manufacturing or distributing the any concerned product. These companies can also be service providers. For analyzing any country specifically, we do consider the growth factors prevailing under the states/cities/county for the same. For instance, if we are analyzing an industry specific to United States, we primarily need to study about the states present under the same(where the product/service has the highest growth). Similar analysis will be followed by other countries. Our scope of the report changes with different markets.
Our research study is mainly implement through a mix of both secondary and primary research. Various sources such as industry magazines, trade journals, and government websites and trade associations are reviewed for gathering precise data. Primary interviews are conducted to validate the market size derived from secondary research. Industry experts, major manufacturers and distributors are contacted for further validation purpose on the current market penetration and growth trends.
Prominent participants in our primary research process include:
- Key Opinion Leaders namely the CEOs, CSOs, VPs, purchasing managers, amongst others
- Research and development participants, distributors/suppliers and subject matter experts
Secondary Research includes data extracted from paid data sources:
- Reuters
- Factiva
- Bloomberg
- One Source
- Hoovers
Research Methodology
Key Inclusions

Reach to us
Call us on
+91-74002-42424
Drop us an email at
sales@absolutemarketsinsights.com
Why Absolute Markets Insights?
An effective strategy is the entity that influences a business to stand out of the crowd. An organization with a phenomenal strategy for success dependably has the edge over the rivals in the market. It offers the organizations a head start in planning their strategy. Absolute Market Insights is the new initiation in the industry that will furnish you with the lead your business needs. Absolute Market Insights is the best destination for your business intelligence and analytical solutions; essentially because our qualitative and quantitative sources of information are competent to give one-stop solutions. We inventively combine qualitative and quantitative research in accurate proportions to have the best report, which not only gives the most recent insights but also assists you to grow.

