Global Recombinant Factor C Assay Market By Type (PyroGene – Routine, Inhibition/ Enhancement, RSE/ CSE, Initial Qualification, EndoNext – EndoZyme II, EndoZyme II GO, EndoLisa); By Application (In-process testing, Final product testing, Water testing, Testing plant-based material); By End User (Pharmaceutical Companies, Biotechnology Companies, Medical Device Companies, Contract Research Organizations (CROs), Research Centers, Non-profits and Federal Government, Others); By Region (North America (U.S., Canada, Mexico, Rest Of North America), Europe (France, The UK, Spain, Germany, Italy, Denmark, Finland, Iceland, Sweden, Norway, Belgium, The Netherlands, Luxembourg, Rest of Europe), Asia Pacific (China, Japan, India, New Zealand, Australia, South Korea, Southeast Asia, Indonesia, Thailand, Malaysia, Singapore, Rest of Southeast Asia, Rest of Asia Pacific, Middle East & Africa (Saudi Arabia, UAE, Egypt, Kuwait, South Africa, Rest of Middle East & Africa) Latin America (Brazil, Argentina, Rest of Latin America)) - Global Insights, Growth, Size, Comparative Analysis, Trends and Forecast, 2023 – 2031
Industry Trends
Today the medical science has definitely arrived a long way and quick diagnosis and treatment of most of the diseases is possible. Several tests have gained immense popularity over the last decade and one such test is the recombinant factor c assay. The recombinant Factor C (rFC) assay is a test used to detect the presence of bacterial endotoxins, also known as lipopolysaccharides (LPS), in a variety of samples, such as pharmaceutical products, medical devices, and food. Endotoxins are harmful substances produced by the cell walls of gram-negative bacteria that can cause fever, shock, and even death in humans and animals. Traditionally, the Limulus Amebocyte Lysate (LAL) assay has been used to detect endotoxins. However, the rFC assay has emerged as an alternative to the LAL assay, as it is based on recombinant technology and does not require the use of horseshoe crabs, which are becoming increasingly endangered due to over-harvesting.
The rFC assay involves the use of a recombinant form of Factor C, which is a key protein involved in the LPS recognition pathway in horseshoe crabs. The rFC protein is produced using genetic engineering techniques and can be manufactured in large quantities, ensuring a consistent supply for testing purposes. The rFC assay works by detecting the activation of the recombinant Factor C protein in response to the presence of endotoxins. The activation results in the production of a fluorescent or chromogenic signal, which is then measured using specialized equipment. The amount of fluorescence or color produced is proportional to the concentration of endotoxins in the sample. There are many factors why the global recombinant factor C assay market is growing. Pharma giants like Elli Lilly are leaving no unturned in increasing the efficacy of this medical test.
In terms of revenue, the global recombinant factor C assay market was valued at US$ 21 Mn in 2022 growing at a CAGR of 14% over the forecast period (2023 – 2031). The study analyses the market in terms of revenue across all the major regions, which have been bifurcated into countries.
Component Outlook
Based on the type outlook, the global recombinant factor C assay market has been segregated into pyroGene and endoNext. The pyroGene segment is further divided into routine, inhibition/ enhancement, RSE/ CSE and initial qualification. While the endoNext segment has been again fragmented into endoZyme II, endoZyme II GO and endoLisA Couple of difference among pyroGene and endoNex are that they are manufactured by different companies. PyroGene posseses a lower limit of detection (LOD) compared to endoNext. This is the reason why the endoNext segment accounts for most of the money made by this market.
Region Outlook
On the basis of the region, the North America has been minting most of the money for the global recombinant factor C assay market in the year 2022. Countries like the United States and Canada definitely are the best in terms of healthcare, and several companies in the medical sector are providing greater solutions for enhancing the accuracy of this test.
Competitive Landscape
The report provides both, qualitative and quantitative research of global recombinant factor C assay market, as well as provides comprehensive insights and development methods adopted by the key contenders. The report also offers extensive research on the key players in this market and details on the competitiveness of these players. Key business strategies such as mergers and acquisitions (M&A), affiliations, collaborations, and contracts adopted by these major market participants are also recognized and analysed in the report. For each company, the report studies their global presence, competitors, service offerings and specification amongst others.
Some of the key players operating in the global recombinant factor C assay market are
- BIOMERIEUX SA
- BioVendor, LLC
- Creative Biolabs
- Eli Lilly
- Lonza
- Other Market Participants
Global Recombinant Factor C Assay Market:
By Type
- PyroGene
- Routine
- Inhibition/ Enhancement
- RSE/ CSE
- Initial Qualification
- EndoNext
- EndoZyme II
- EndoZyme II GO
- EndoLisa
By Application
- In-process testing
- Final product testing
- Water testing
- Testing plant-based material
By End User
- Pharmaceutical Companies
- Biotechnology Companies
- Medical Device Companies
- Contract Research Organizations (CROs)
- Research Centers
- Non-profits and Federal Government
- Others
By Region
- North America (U.S., Canada, Mexico, Rest of North America)
- Europe (France, The UK, Spain, Germany, Italy, Nordic Countries (Denmark, Finland, Iceland, Sweden, Norway), Benelux Union (Belgium, The Netherlands, Luxembourg), Rest of Europe
- Asia Pacific (China, Japan, India, New Zealand, Australia, South Korea, Southeast Asia (Indonesia, Thailand, Malaysia, Singapore, Rest of Southeast Asia), Rest of Asia Pacific
- Middle East & Africa (Saudi Arabia, UAE, Egypt, Kuwait, South Africa, Rest of Middle East & Africa)
- Latin America (Brazil, Argentina, Rest of Latin America)
Table of Contents
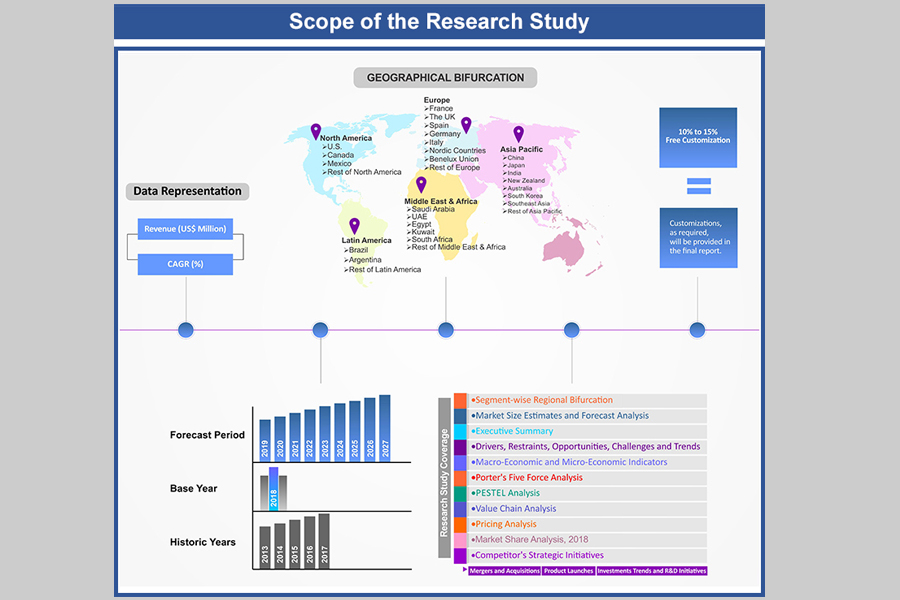
1. Market Scope
1.1. Market Segmentation
1.2. Years Considered
1.2.1. Historic Years: 2015 - 2021
1.2.2. Base Year: 2022
1.2.3. Forecast Years: 2023 - 2031
2. Key Target Audiences
3. Research Methodology
3.1. Primary Research
3.1.1. Research Questionnaire
3.1.2. Global Percentage Breakdown
3.1.3. Primary Interviews: Key Opinion Leaders (KOLs)
3.2. Secondary Research
3.2.1. Paid Databases
3.2.2. Secondary Sources
3.3. Market Size Estimates
3.3.1. Top-Down Approach
3.3.2. Bottom-Up Approach
3.4. Data Triangulation Methodology
3.5. Research Assumptions
4. Recommendations and Insights from AMI’s Perspective**
5. Holistic Overview of Recombinant Factor C Assay Market
6. Market Synopsis:
Recombinant Factor C Assay Market
7. Recombinant Factor C Assay Market Analysis: Qualitative
Perspective
7.1. Introduction
7.1.1. Product Definition
7.1.2. Industry Development
7.2. Market Dynamics
7.2.1. Drivers
7.2.2. Restraints
7.2.3. Opportunities
7.2.4. Challenges
7.3. Trends in Recombinant Factor C Assay Market
7.4. Market Determinants Radar Chart
7.5. Macro-Economic and Micro-Economic Indicators: Recombinant
Factor C Assay Market
7.6. Porter’s Five Force Analysis
7.7. Impact of Covid-19 on Recombinant Factor C Assay Market
8. Global Recombinant Factor C Assay Market Analysis and
Forecasts, 2023 - 2031
8.1. Overview
8.1.1. Global Recombinant Factor C Assay Market Revenue (US$ Mn)
8.2. Global Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Type
8.2.1. PyroGene (Definition, Market Estimation and Penetration, 2015
- 2022, Market Estimation (2015 - 2022), Market Forecast (2023 - 2031),
Compound Annual Growth Rate (CAGR), Regional Bifurcation (North America,
Europe, Asia Pacific, Middle East and Africa, Latin America) and Information on
Routine, Inhibition/ Enhancement, RSE/ CSE, Initial Qualification)
8.2.1.1. Routine
8.2.1.2. Inhibition/ Enhancement
8.2.1.3. RSE/ CSE
8.2.1.4. Initial Qualification
8.2.2. EndoNext (Definition, Market Estimation and Penetration, 2015
- 2022, Market Estimation (2015 - 2022), Market Forecast (2023 - 2031),
Compound Annual Growth Rate (CAGR), Regional Bifurcation (North America,
Europe, Asia Pacific, Middle East and Africa, Latin America) and Information on
EndoZyme II, EndoZyme II GO, EndoLisa)
8.2.2.1. EndoZyme II
8.2.2.2. EndoZyme II GO
8.2.2.3. EndoLisa
8.3. Key Segment for Channeling Investments
8.3.1. By Type
9. Global Recombinant Factor C Assay Market Analysis and
Forecasts, 2023 - 2031
9.1. Overview
9.2. Global Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Application
9.2.1. In-process testing
9.2.1.1. Definition
9.2.1.2. Market Estimation and Penetration, 2015 - 2022
9.2.1.3. Market Forecast, 2023 - 2031
9.2.1.4. Compound Annual Growth Rate (CAGR)
9.2.1.5. Regional Bifurcation
9.2.1.5.1. North America
9.2.1.5.1.1. Market
Estimation, 2015 - 2022
9.2.1.5.1.2. Market
Forecast, 2023 - 2031
9.2.1.5.2. Europe
9.2.1.5.2.1. Market
Estimation, 2015 - 2022
9.2.1.5.2.2. Market
Forecast, 2023 - 2031
9.2.1.5.3. Asia Pacific
9.2.1.5.3.1. Market
Estimation, 2015 - 2022
9.2.1.5.3.2. Market
Forecast, 2023 - 2031
9.2.1.5.4. Middle East and Africa
9.2.1.5.4.1. Market
Estimation, 2015 - 2022
9.2.1.5.4.2. Market
Forecast, 2023 - 2031
9.2.1.5.5. Latin America
9.2.1.5.5.1. Market
Estimation, 2015 - 2022
9.2.1.5.5.2. Market
Forecast, 2023 - 2031
9.2.2. Final product testing
9.2.2.1. Definition
9.2.2.2. Market Estimation and Penetration, 2015 - 2022
9.2.2.3. Market Forecast, 2023 - 2031
9.2.2.4. Compound Annual Growth Rate (CAGR)
9.2.2.5. Regional Bifurcation
9.2.2.5.1. North America
9.2.2.5.1.1. Market
Estimation, 2015 - 2022
9.2.2.5.1.2. Market
Forecast, 2023 - 2031
9.2.2.5.2. Europe
9.2.2.5.2.1. Market
Estimation, 2015 - 2022
9.2.2.5.2.2. Market
Forecast, 2023 - 2031
9.2.2.5.3. Asia Pacific
9.2.2.5.3.1. Market
Estimation, 2015 - 2022
9.2.2.5.3.2. Market
Forecast, 2023 - 2031
9.2.2.5.4. Middle East and Africa
9.2.2.5.4.1. Market
Estimation, 2015 - 2022
9.2.2.5.4.2. Market
Forecast, 2023 - 2031
9.2.2.5.5. Latin America
9.2.2.5.5.1. Market
Estimation, 2015 - 2022
9.2.2.5.5.2. Market
Forecast, 2023 - 2031
9.2.3. Water testing
9.2.3.1. Definition
9.2.3.2. Market Estimation and Penetration, 2015 - 2022
9.2.3.3. Market Forecast, 2023 - 2031
9.2.3.4. Compound Annual Growth Rate (CAGR)
9.2.3.5. Regional Bifurcation
9.2.3.5.1. North America
9.2.3.5.1.1. Market
Estimation, 2015 - 2022
9.2.3.5.1.2. Market
Forecast, 2023 - 2031
9.2.3.5.2. Europe
9.2.3.5.2.1. Market
Estimation, 2015 - 2022
9.2.3.5.2.2. Market
Forecast, 2023 - 2031
9.2.3.5.3. Asia Pacific
9.2.3.5.3.1. Market
Estimation, 2015 - 2022
9.2.3.5.3.2. Market
Forecast, 2023 - 2031
9.2.3.5.4. Middle East and Africa
9.2.3.5.4.1. Market
Estimation, 2015 - 2022
9.2.3.5.4.2. Market
Forecast, 2023 - 2031
9.2.3.5.5. Latin America
9.2.3.5.5.1. Market
Estimation, 2015 - 2022
9.2.3.5.5.2. Market
Forecast, 2023 - 2031
9.2.4. Testing plant-based material
9.2.4.1. Definition
9.2.4.2. Market Estimation and Penetration, 2015 - 2022
9.2.4.3. Market Forecast, 2023 - 2031
9.2.4.4. Compound Annual Growth Rate (CAGR)
9.2.4.5. Regional Bifurcation
9.2.4.5.1. North America
9.2.4.5.1.1. Market
Estimation, 2015 - 2022
9.2.4.5.1.2. Market
Forecast, 2023 - 2031
9.2.4.5.2. Europe
9.2.4.5.2.1. Market
Estimation, 2015 - 2022
9.2.4.5.2.2. Market
Forecast, 2023 - 2031
9.2.4.5.3. Asia Pacific
9.2.4.5.3.1. Market
Estimation, 2015 - 2022
9.2.4.5.3.2. Market
Forecast, 2023 - 2031
9.2.4.5.4. Middle East and Africa
9.2.4.5.4.1. Market
Estimation, 2015 - 2022
9.2.4.5.4.2. Market
Forecast, 2023 - 2031
9.2.4.5.5. Latin America
9.2.4.5.5.1. Market
Estimation, 2015 - 2022
9.2.4.5.5.2. Market
Forecast, 2023 - 2031
9.3. Key Segment for Channeling Investments
9.3.1. By Application
10. Global Recombinant Factor C Assay Market Analysis and
Forecasts, 2023 - 2031
10.1. Overview
10.2. Global Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By End User
10.2.1. Pharmaceutical Companies
10.2.1.1. Definition
10.2.1.2. Market Estimation and Penetration, 2015 - 2022
10.2.1.3. Market Forecast, 2023 - 2031
10.2.1.4. Compound Annual Growth Rate (CAGR)
10.2.1.5. Regional Bifurcation
10.2.1.5.1. North America
10.2.1.5.1.1. Market
Estimation, 2015 - 2022
10.2.1.5.1.2. Market
Forecast, 2023 - 2031
10.2.1.5.2. Europe
10.2.1.5.2.1. Market
Estimation, 2015 - 2022
10.2.1.5.2.2. Market
Forecast, 2023 - 2031
10.2.1.5.3. Asia Pacific
10.2.1.5.3.1. Market
Estimation, 2015 - 2022
10.2.1.5.3.2. Market
Forecast, 2023 - 2031
10.2.1.5.4. Middle East and Africa
10.2.1.5.4.1. Market
Estimation, 2015 - 2022
10.2.1.5.4.2. Market
Forecast, 2023 - 2031
10.2.1.5.5. Latin America
10.2.1.5.5.1. Market
Estimation, 2015 - 2022
10.2.1.5.5.2. Market
Forecast, 2023 - 2031
10.2.2. Biotechnology Companies
10.2.2.1. Definition
10.2.2.2. Market Estimation and Penetration, 2015 - 2022
10.2.2.3. Market Forecast, 2023 - 2031
10.2.2.4. Compound Annual Growth Rate (CAGR)
10.2.2.5. Regional Bifurcation
10.2.2.5.1. North America
10.2.2.5.1.1. Market
Estimation, 2015 - 2022
10.2.2.5.1.2. Market
Forecast, 2023 - 2031
10.2.2.5.2. Europe
10.2.2.5.2.1. Market
Estimation, 2015 - 2022
10.2.2.5.2.2. Market
Forecast, 2023 - 2031
10.2.2.5.3. Asia Pacific
10.2.2.5.3.1. Market
Estimation, 2015 - 2022
10.2.2.5.3.2. Market
Forecast, 2023 - 2031
10.2.2.5.4. Middle East and Africa
10.2.2.5.4.1. Market
Estimation, 2015 - 2022
10.2.2.5.4.2. Market
Forecast, 2023 - 2031
10.2.2.5.5. Latin America
10.2.2.5.5.1. Market
Estimation, 2015 - 2022
10.2.2.5.5.2. Market
Forecast, 2023 - 2031
10.2.3. Medical Device Companies
10.2.3.1. Definition
10.2.3.2. Market Estimation and Penetration, 2015 - 2022
10.2.3.3. Market Forecast, 2023 - 2031
10.2.3.4. Compound Annual Growth Rate (CAGR)
10.2.3.5. Regional Bifurcation
10.2.3.5.1. North America
10.2.3.5.1.1. Market
Estimation, 2015 - 2022
10.2.3.5.1.2. Market
Forecast, 2023 - 2031
10.2.3.5.2. Europe
10.2.3.5.2.1. Market
Estimation, 2015 - 2022
10.2.3.5.2.2. Market
Forecast, 2023 - 2031
10.2.3.5.3. Asia Pacific
10.2.3.5.3.1. Market
Estimation, 2015 - 2022
10.2.3.5.3.2. Market
Forecast, 2023 - 2031
10.2.3.5.4. Middle East and Africa
10.2.3.5.4.1. Market
Estimation, 2015 - 2022
10.2.3.5.4.2. Market
Forecast, 2023 - 2031
10.2.3.5.5. Latin America
10.2.3.5.5.1. Market
Estimation, 2015 - 2022
10.2.3.5.5.2. Market
Forecast, 2023 - 2031
10.2.4. Contract Research Organizations (CROs)
10.2.4.1. Definition
10.2.4.2. Market Estimation and Penetration, 2015 - 2022
10.2.4.3. Market Forecast, 2023 - 2031
10.2.4.4. Compound Annual Growth Rate (CAGR)
10.2.4.5. Regional Bifurcation
10.2.4.5.1. North America
10.2.4.5.1.1. Market
Estimation, 2015 - 2022
10.2.4.5.1.2. Market
Forecast, 2023 - 2031
10.2.4.5.2. Europe
10.2.4.5.2.1. Market
Estimation, 2015 - 2022
10.2.4.5.2.2. Market
Forecast, 2023 - 2031
10.2.4.5.3. Asia Pacific
10.2.4.5.3.1. Market
Estimation, 2015 - 2022
10.2.4.5.3.2. Market
Forecast, 2023 - 2031
10.2.4.5.4. Middle East and Africa
10.2.4.5.4.1. Market
Estimation, 2015 - 2022
10.2.4.5.4.2. Market
Forecast, 2023 - 2031
10.2.4.5.5. Latin America
10.2.4.5.5.1. Market
Estimation, 2015 - 2022
10.2.4.5.5.2. Market
Forecast, 2023 - 2031
10.2.5. Research Centers
10.2.5.1. Definition
10.2.5.2. Market Estimation and Penetration, 2015 - 2022
10.2.5.3. Market Forecast, 2023 - 2031
10.2.5.4. Compound Annual Growth Rate (CAGR)
10.2.5.5. Regional Bifurcation
10.2.5.5.1. North America
10.2.5.5.1.1. Market
Estimation, 2015 - 2022
10.2.5.5.1.2. Market
Forecast, 2023 - 2031
10.2.5.5.2. Europe
10.2.5.5.2.1. Market
Estimation, 2015 - 2022
10.2.5.5.2.2. Market
Forecast, 2023 - 2031
10.2.5.5.3. Asia Pacific
10.2.5.5.3.1. Market
Estimation, 2015 - 2022
10.2.5.5.3.2. Market
Forecast, 2023 - 2031
10.2.5.5.4. Middle East and Africa
10.2.5.5.4.1. Market
Estimation, 2015 - 2022
10.2.5.5.4.2. Market
Forecast, 2023 - 2031
10.2.5.5.5. Latin America
10.2.5.5.5.1. Market
Estimation, 2015 - 2022
10.2.5.5.5.2. Market
Forecast, 2023 - 2031
10.2.6. Non-profits and Federal Government
10.2.6.1. Definition
10.2.6.2. Market Estimation and Penetration, 2015 - 2022
10.2.6.3. Market Forecast, 2023 - 2031
10.2.6.4. Compound Annual Growth Rate (CAGR)
10.2.6.5. Regional Bifurcation
10.2.6.5.1. North America
10.2.6.5.1.1. Market
Estimation, 2015 - 2022
10.2.6.5.1.2. Market
Forecast, 2023 - 2031
10.2.6.5.2. Europe
10.2.6.5.2.1. Market
Estimation, 2015 - 2022
10.2.6.5.2.2. Market
Forecast, 2023 - 2031
10.2.6.5.3. Asia Pacific
10.2.6.5.3.1. Market
Estimation, 2015 - 2022
10.2.6.5.3.2. Market
Forecast, 2023 - 2031
10.2.6.5.4. Middle East and Africa
10.2.6.5.4.1. Market
Estimation, 2015 - 2022
10.2.6.5.4.2. Market
Forecast, 2023 - 2031
10.2.6.5.5. Latin America
10.2.6.5.5.1. Market
Estimation, 2015 - 2022
10.2.6.5.5.2. Market
Forecast, 2023 - 2031
10.2.7. Others
10.2.7.1. Definition
10.2.7.2. Market Estimation and Penetration, 2015 - 2022
10.2.7.3. Market Forecast, 2023 - 2031
10.2.7.4. Compound Annual Growth Rate (CAGR)
10.2.7.5. Regional Bifurcation
10.2.7.5.1. North America
10.2.7.5.1.1. Market
Estimation, 2015 - 2022
10.2.7.5.1.2. Market
Forecast, 2023 - 2031
10.2.7.5.2. Europe
10.2.7.5.2.1. Market
Estimation, 2015 - 2022
10.2.7.5.2.2. Market
Forecast, 2023 - 2031
10.2.7.5.3. Asia Pacific
10.2.7.5.3.1. Market
Estimation, 2015 - 2022
10.2.7.5.3.2. Market
Forecast, 2023 - 2031
10.2.7.5.4. Middle East and Africa
10.2.7.5.4.1. Market
Estimation, 2015 - 2022
10.2.7.5.4.2. Market
Forecast, 2023 - 2031
10.2.7.5.5. Latin America
10.2.7.5.5.1. Market
Estimation, 2015 - 2022
10.2.7.5.5.2. Market
Forecast, 2023 - 2031
10.3. Key Segment for Channeling Investments
10.3.1. By End User
11. North America Recombinant Factor C Assay Market Analysis and
Forecasts, 2023 - 2031
11.1. Overview
11.1.1. North America Recombinant Factor C Assay Market Revenue (US$
Mn)
11.2. North America Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Type
11.2.1. PyroGene
11.2.1.1. Routine
11.2.1.2. Inhibition/ Enhancement
11.2.1.3. RSE/ CSE
11.2.1.4. Initial Qualification
11.2.2. EndoNext
11.2.2.1. EndoZyme II
11.2.2.2. EndoZyme II GO
11.2.2.3. EndoLisa
11.3. North America Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Application
11.3.1. In-process testing
11.3.2. Final product testing
11.3.3. Water testing
11.3.4. Testing plant-based material
11.4. North America Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By End User
11.4.1. Pharmaceutical Companies
11.4.2. Biotechnology Companies
11.4.3. Medical Device Companies
11.4.4. Contract Research Organizations (CROs)
11.4.5. Research Centers
11.4.6. Non-profits and Federal Government
11.4.7. Others
11.5. North America Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Country
11.5.1. U.S
11.5.1.1. U.S Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Type
11.5.1.1.1. PyroGene
11.5.1.1.1.1. Routine
11.5.1.1.1.2. Inhibition/
Enhancement
11.5.1.1.1.3. RSE/
CSE
11.5.1.1.1.4. Initial
Qualification
11.5.1.1.2. EndoNext
11.5.1.1.2.1. EndoZyme
II
11.5.1.1.2.2. EndoZyme
II GO
11.5.1.1.2.3. EndoLisa
11.5.1.2. U.S Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Application
11.5.1.2.1. In-process testing
11.5.1.2.2. Final product testing
11.5.1.2.3. Water testing
11.5.1.2.4. Testing plant-based material
11.5.1.3. U.S Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By End User
11.5.1.3.1. Pharmaceutical Companies
11.5.1.3.2. Biotechnology Companies
11.5.1.3.3. Medical Device Companies
11.5.1.3.4. Contract Research Organizations (CROs)
11.5.1.3.5. Research Centers
11.5.1.3.6. Non-profits and Federal Government
11.5.1.3.7. Others
11.5.2. Canada
11.5.2.1. Canada Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Type
11.5.2.1.1. PyroGene
11.5.2.1.1.1. Routine
11.5.2.1.1.2. Inhibition/
Enhancement
11.5.2.1.1.3. RSE/
CSE
11.5.2.1.1.4. Initial
Qualification
11.5.2.1.2. EndoNext
11.5.2.1.2.1. EndoZyme
II
11.5.2.1.2.2. EndoZyme
II GO
11.5.2.1.2.3. EndoLisa
11.5.2.2. Canada Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Application
11.5.2.2.1. In-process testing
11.5.2.2.2. Final product testing
11.5.2.2.3. Water testing
11.5.2.2.4. Testing plant-based material
11.5.2.3. Canada Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By End User
11.5.2.3.1. Pharmaceutical Companies
11.5.2.3.2. Biotechnology Companies
11.5.2.3.3. Medical Device Companies
11.5.2.3.4. Contract Research Organizations (CROs)
11.5.2.3.5. Research Centers
11.5.2.3.6. Non-profits and Federal Government
11.5.2.3.7. Others
11.5.3. Mexico
11.5.3.1. Mexico Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Type
11.5.3.1.1. PyroGene
11.5.3.1.1.1. Routine
11.5.3.1.1.2. Inhibition/
Enhancement
11.5.3.1.1.3. RSE/
CSE
11.5.3.1.1.4. Initial
Qualification
11.5.3.1.2. EndoNext
11.5.3.1.2.1. EndoZyme
II
11.5.3.1.2.2. EndoZyme
II GO
11.5.3.1.2.3. EndoLisa
11.5.3.2. Mexico Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Application
11.5.3.2.1. In-process testing
11.5.3.2.2. Final product testing
11.5.3.2.3. Water testing
11.5.3.2.4. Testing plant-based material
11.5.3.3. Mexico Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By End User
11.5.3.3.1. Pharmaceutical Companies
11.5.3.3.2. Biotechnology Companies
11.5.3.3.3. Medical Device Companies
11.5.3.3.4. Contract Research Organizations (CROs)
11.5.3.3.5. Research Centers
11.5.3.3.6. Non-profits and Federal Government
11.5.3.3.7. Others
11.5.4. Rest of North America
11.5.4.1. Rest of North America Recombinant Factor C Assay Market Revenue
(US$ Mn) and Forecasts, By Type
11.5.4.1.1. PyroGene
11.5.4.1.1.1. Routine
11.5.4.1.1.2. Inhibition/
Enhancement
11.5.4.1.1.3. RSE/
CSE
11.5.4.1.1.4. Initial
Qualification
11.5.4.1.2. EndoNext
11.5.4.1.2.1. EndoZyme
II
11.5.4.1.2.2. EndoZyme
II GO
11.5.4.1.2.3. EndoLisa
11.5.4.2. Rest of North America Recombinant Factor C Assay Market
Revenue (US$ Mn) and Forecasts, By Application
11.5.4.2.1. In-process testing
11.5.4.2.2. Final product testing
11.5.4.2.3. Water testing
11.5.4.2.4. Testing plant-based material
11.5.4.3. Rest of North America Recombinant Factor C Assay Market
Revenue (US$ Mn) and Forecasts, By End User
11.5.4.3.1. Pharmaceutical Companies
11.5.4.3.2. Biotechnology Companies
11.5.4.3.3. Medical Device Companies
11.5.4.3.4. Contract Research Organizations (CROs)
11.5.4.3.5. Research Centers
11.5.4.3.6. Non-profits and Federal Government
11.5.4.3.7. Others
11.6. Key Segment for Channeling Investments
11.6.1. By Country
11.6.2. By Type
11.6.3. By Application
11.6.4. By End User
12. Europe Recombinant Factor C Assay Market Analysis and
Forecasts, 2023 - 2031
12.1. Overview
12.1.1. Europe Recombinant Factor C Assay Market Revenue (US$ Mn)
12.2. Europe Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Type
12.2.1. PyroGene
12.2.1.1. Routine
12.2.1.2. Inhibition/ Enhancement
12.2.1.3. RSE/ CSE
12.2.1.4. Initial Qualification
12.2.2. EndoNext
12.2.2.1. EndoZyme II
12.2.2.2. EndoZyme II GO
12.2.2.3. EndoLisa
12.3. Europe Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Application
12.3.1. In-process testing
12.3.2. Final product testing
12.3.3. Water testing
12.3.4. Testing plant-based material
12.4. Europe Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By End User
12.4.1. Pharmaceutical Companies
12.4.2. Biotechnology Companies
12.4.3. Medical Device Companies
12.4.4. Contract Research Organizations (CROs)
12.4.5. Research Centers
12.4.6. Non-profits and Federal Government
12.4.7. Others
12.5. Europe Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Country
12.5.1. France
12.5.1.1. France Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Type
12.5.1.1.1. PyroGene
12.5.1.1.1.1. Routine
12.5.1.1.1.2. Inhibition/
Enhancement
12.5.1.1.1.3. RSE/
CSE
12.5.1.1.1.4. Initial
Qualification
12.5.1.1.2. EndoNext
12.5.1.1.2.1. EndoZyme
II
12.5.1.1.2.2. EndoZyme
II GO
12.5.1.1.2.3. EndoLisa
12.5.1.2. France Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Application
12.5.1.2.1. In-process testing
12.5.1.2.2. Final product testing
12.5.1.2.3. Water testing
12.5.1.2.4. Testing plant-based material
12.5.1.3. France Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By End User
12.5.1.3.1. Pharmaceutical Companies
12.5.1.3.2. Biotechnology Companies
12.5.1.3.3. Medical Device Companies
12.5.1.3.4. Contract Research Organizations (CROs)
12.5.1.3.5. Research Centers
12.5.1.3.6. Non-profits and Federal Government
12.5.1.3.7. Others
12.5.2. The UK
12.5.2.1. The UK Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Type
12.5.2.1.1. PyroGene
12.5.2.1.1.1. Routine
12.5.2.1.1.2. Inhibition/
Enhancement
12.5.2.1.1.3. RSE/
CSE
12.5.2.1.1.4. Initial
Qualification
12.5.2.1.2. EndoNext
12.5.2.1.2.1. EndoZyme
II
12.5.2.1.2.2. EndoZyme
II GO
12.5.2.1.2.3. EndoLisa
12.5.2.2. The UK Recombinant Factor C Assay Market Revenue (US$ Mn) and Forecasts,
By Application
12.5.2.2.1. In-process testing
12.5.2.2.2. Final product testing
12.5.2.2.3. Water testing
12.5.2.2.4. Testing plant-based material
12.5.2.3. The UK Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By End User
12.5.2.3.1. Pharmaceutical Companies
12.5.2.3.2. Biotechnology Companies
12.5.2.3.3. Medical Device Companies
12.5.2.3.4. Contract Research Organizations (CROs)
12.5.2.3.5. Research Centers
12.5.2.3.6. Non-profits and Federal Government
12.5.2.3.7. Others
12.5.3. Spain
12.5.3.1. Spain Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Type
12.5.3.1.1. PyroGene
12.5.3.1.1.1. Routine
12.5.3.1.1.2. Inhibition/
Enhancement
12.5.3.1.1.3. RSE/
CSE
12.5.3.1.1.4. Initial
Qualification
12.5.3.1.2. EndoNext
12.5.3.1.2.1. EndoZyme
II
12.5.3.1.2.2. EndoZyme
II GO
12.5.3.1.2.3. EndoLisa
12.5.3.2. Spain Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Application
12.5.3.2.1. In-process testing
12.5.3.2.2. Final product testing
12.5.3.2.3. Water testing
12.5.3.2.4. Testing plant-based material
12.5.3.3. Spain Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By End User
12.5.3.3.1. Pharmaceutical Companies
12.5.3.3.2. Biotechnology Companies
12.5.3.3.3. Medical Device Companies
12.5.3.3.4. Contract Research Organizations (CROs)
12.5.3.3.5. Research Centers
12.5.3.3.6. Non-profits and Federal Government
12.5.3.3.7. Others
12.5.4. Germany
12.5.4.1. Germany Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Type
12.5.4.1.1. PyroGene
12.5.4.1.1.1. Routine
12.5.4.1.1.2. Inhibition/
Enhancement
12.5.4.1.1.3. RSE/
CSE
12.5.4.1.1.4. Initial
Qualification
12.5.4.1.2. EndoNext
12.5.4.1.2.1. EndoZyme
II
12.5.4.1.2.2. EndoZyme
II GO
12.5.4.1.2.3. EndoLisa
12.5.4.2. Germany Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Application
12.5.4.2.1. In-process testing
12.5.4.2.2. Final product testing
12.5.4.2.3. Water testing
12.5.4.2.4. Testing plant-based material
12.5.4.3. Germany Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By End User
12.5.4.3.1. Pharmaceutical Companies
12.5.4.3.2. Biotechnology Companies
12.5.4.3.3. Medical Device Companies
12.5.4.3.4. Contract Research Organizations (CROs)
12.5.4.3.5. Research Centers
12.5.4.3.6. Non-profits and Federal Government
12.5.4.3.7. Others
12.5.5. Italy
12.5.5.1. Italy Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Type
12.5.5.1.1. PyroGene
12.5.5.1.1.1. Routine
12.5.5.1.1.2. Inhibition/
Enhancement
12.5.5.1.1.3. RSE/
CSE
12.5.5.1.1.4. Initial
Qualification
12.5.5.1.2. EndoNext
12.5.5.1.2.1. EndoZyme
II
12.5.5.1.2.2. EndoZyme
II GO
12.5.5.1.2.3. EndoLisa
12.5.5.2. Italy Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Application
12.5.5.2.1. In-process testing
12.5.5.2.2. Final product testing
12.5.5.2.3. Water testing
12.5.5.2.4. Testing plant-based material
12.5.5.3. Italy Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By End User
12.5.5.3.1. Pharmaceutical Companies
12.5.5.3.2. Biotechnology Companies
12.5.5.3.3. Medical Device Companies
12.5.5.3.4. Contract Research Organizations (CROs)
12.5.5.3.5. Research Centers
12.5.5.3.6. Non-profits and Federal Government
12.5.5.3.7. Others
12.5.6. Nordic Countries
12.5.6.1. Nordic Countries Recombinant Factor C Assay Market Revenue
(US$ Mn) and Forecasts, By Type
12.5.6.1.1. PyroGene
12.5.6.1.1.1. Routine
12.5.6.1.1.2. Inhibition/
Enhancement
12.5.6.1.1.3. RSE/
CSE
12.5.6.1.1.4. Initial
Qualification
12.5.6.1.2. EndoNext
12.5.6.1.2.1. EndoZyme
II
12.5.6.1.2.2. EndoZyme
II GO
12.5.6.1.2.3. EndoLisa
12.5.6.2. Nordic Countries Recombinant Factor C Assay Market Revenue
(US$ Mn) and Forecasts, By Application
12.5.6.2.1. In-process testing
12.5.6.2.2. Final product testing
12.5.6.2.3. Water testing
12.5.6.2.4. Testing plant-based material
12.5.6.3. Nordic Countries Recombinant Factor C Assay Market Revenue
(US$ Mn) and Forecasts, By End User
12.5.6.3.1. Pharmaceutical Companies
12.5.6.3.2. Biotechnology Companies
12.5.6.3.3. Medical Device Companies
12.5.6.3.4. Contract Research Organizations (CROs)
12.5.6.3.5. Research Centers
12.5.6.3.6. Non-profits and Federal Government
12.5.6.3.7. Others
12.5.6.4. Nordic Countries Recombinant Factor C Assay Market Revenue
(US$ Mn) and Forecasts, By Country
12.5.6.4.1. Denmark
12.5.6.4.2. Finland
12.5.6.4.3. Iceland
12.5.6.4.4. Sweden
12.5.6.4.5. Norway
12.5.7. Benelux Union
12.5.7.1. Benelux Union Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Type
12.5.7.1.1. PyroGene
12.5.7.1.1.1. Routine
12.5.7.1.1.2. Inhibition/
Enhancement
12.5.7.1.1.3. RSE/
CSE
12.5.7.1.1.4. Initial
Qualification
12.5.7.1.2. EndoNext
12.5.7.1.2.1. EndoZyme
II
12.5.7.1.2.2. EndoZyme
II GO
12.5.7.1.2.3. EndoLisa
12.5.7.2. Benelux Union Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Application
12.5.7.2.1. In-process testing
12.5.7.2.2. Final product testing
12.5.7.2.3. Water testing
12.5.7.2.4. Testing plant-based material
12.5.7.3. Benelux Union Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By End User
12.5.7.3.1. Pharmaceutical Companies
12.5.7.3.2. Biotechnology Companies
12.5.7.3.3. Medical Device Companies
12.5.7.3.4. Contract Research Organizations (CROs)
12.5.7.3.5. Research Centers
12.5.7.3.6. Non-profits and Federal Government
12.5.7.3.7. Others
12.5.7.4. Benelux Union Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Country
12.5.7.4.1. Belgium
12.5.7.4.2. The Netherlands
12.5.7.4.3. Luxembourg
12.5.8. Rest of Europe
12.5.8.1. Rest of Europe Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Type
12.5.8.1.1. PyroGene
12.5.8.1.1.1. Routine
12.5.8.1.1.2. Inhibition/
Enhancement
12.5.8.1.1.3. RSE/
CSE
12.5.8.1.1.4. Initial
Qualification
12.5.8.1.2. EndoNext
12.5.8.1.2.1. EndoZyme
II
12.5.8.1.2.2. EndoZyme
II GO
12.5.8.1.2.3. EndoLisa
12.5.8.2. Rest of Europe Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Application
12.5.8.2.1. In-process testing
12.5.8.2.2. Final product testing
12.5.8.2.3. Water testing
12.5.8.2.4. Testing plant-based material
12.5.8.3. Rest of Europe Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By End User
12.5.8.3.1. Pharmaceutical Companies
12.5.8.3.2. Biotechnology Companies
12.5.8.3.3. Medical Device Companies
12.5.8.3.4. Contract Research Organizations (CROs)
12.5.8.3.5. Research Centers
12.5.8.3.6. Non-profits and Federal Government
12.5.8.3.7. Others
12.6. Key Segment for Channeling Investments
12.6.1. By Country
12.6.2. By Type
12.6.3. By Application
12.6.4. By End User
13. Asia Pacific Recombinant Factor C Assay Market Analysis and
Forecasts, 2023 - 2031
13.1. Overview
13.1.1. Asia Pacific Recombinant Factor C Assay Market Revenue (US$
Mn)
13.2. Asia Pacific Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Type
13.2.1. PyroGene
13.2.1.1. Routine
13.2.1.2. Inhibition/ Enhancement
13.2.1.3. RSE/ CSE
13.2.1.4. Initial Qualification
13.2.2. EndoNext
13.2.2.1. EndoZyme II
13.2.2.2. EndoZyme II GO
13.2.2.3. EndoLisa
13.3. Asia Pacific Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Application
13.3.1. In-process testing
13.3.2. Final product testing
13.3.3. Water testing
13.3.4. Testing plant-based material
13.4. Asia Pacific Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By End User
13.4.1. Pharmaceutical Companies
13.4.2. Biotechnology Companies
13.4.3. Medical Device Companies
13.4.4. Contract Research Organizations (CROs)
13.4.5. Research Centers
13.4.6. Non-profits and Federal Government
13.4.7. Others
13.5. Asia Pacific Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Country
13.5.1. China
13.5.1.1. China Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Type
13.5.1.1.1. PyroGene
13.5.1.1.1.1. Routine
13.5.1.1.1.2. Inhibition/
Enhancement
13.5.1.1.1.3. RSE/
CSE
13.5.1.1.1.4. Initial
Qualification
13.5.1.1.2. EndoNext
13.5.1.1.2.1. EndoZyme
II
13.5.1.1.2.2. EndoZyme
II GO
13.5.1.1.2.3. EndoLisa
13.5.1.2. China Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Application
13.5.1.2.1. In-process testing
13.5.1.2.2. Final product testing
13.5.1.2.3. Water testing
13.5.1.2.4. Testing plant-based material
13.5.1.3. China Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By End User
13.5.1.3.1. Pharmaceutical Companies
13.5.1.3.2. Biotechnology Companies
13.5.1.3.3. Medical Device Companies
13.5.1.3.4. Contract Research Organizations (CROs)
13.5.1.3.5. Research Centers
13.5.1.3.6. Non-profits and Federal Government
13.5.1.3.7. Others
13.5.2. Japan
13.5.2.1. Japan Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Type
13.5.2.1.1. PyroGene
13.5.2.1.1.1. Routine
13.5.2.1.1.2. Inhibition/
Enhancement
13.5.2.1.1.3. RSE/
CSE
13.5.2.1.1.4. Initial
Qualification
13.5.2.1.2. EndoNext
13.5.2.1.2.1. EndoZyme
II
13.5.2.1.2.2. EndoZyme
II GO
13.5.2.1.2.3. EndoLisa
13.5.2.2. Japan Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Application
13.5.2.2.1. In-process testing
13.5.2.2.2. Final product testing
13.5.2.2.3. Water testing
13.5.2.2.4. Testing plant-based material
13.5.2.3. Japan Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By End User
13.5.2.3.1. Pharmaceutical Companies
13.5.2.3.2. Biotechnology Companies
13.5.2.3.3. Medical Device Companies
13.5.2.3.4. Contract Research Organizations (CROs)
13.5.2.3.5. Research Centers
13.5.2.3.6. Non-profits and Federal Government
13.5.2.3.7. Others
13.5.3. India
13.5.3.1. India Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Type
13.5.3.1.1. PyroGene
13.5.3.1.1.1. Routine
13.5.3.1.1.2. Inhibition/
Enhancement
13.5.3.1.1.3. RSE/
CSE
13.5.3.1.1.4. Initial
Qualification
13.5.3.1.2. EndoNext
13.5.3.1.2.1. EndoZyme
II
13.5.3.1.2.2. EndoZyme
II GO
13.5.3.1.2.3. EndoLisa
13.5.3.2. India Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Application
13.5.3.2.1. In-process testing
13.5.3.2.2. Final product testing
13.5.3.2.3. Water testing
13.5.3.2.4. Testing plant-based material
13.5.3.3. India Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By End User
13.5.3.3.1. Pharmaceutical Companies
13.5.3.3.2. Biotechnology Companies
13.5.3.3.3. Medical Device Companies
13.5.3.3.4. Contract Research Organizations (CROs)
13.5.3.3.5. Research Centers
13.5.3.3.6. Non-profits and Federal Government
13.5.3.3.7. Others
13.5.4. New Zealand
13.5.4.1. New Zealand Recombinant Factor C Assay Market Revenue (US$ Mn)
and Forecasts, By Type
13.5.4.1.1. PyroGene
13.5.4.1.1.1. Routine
13.5.4.1.1.2. Inhibition/
Enhancement
13.5.4.1.1.3. RSE/
CSE
13.5.4.1.1.4. Initial
Qualification
13.5.4.1.2. EndoNext
13.5.4.1.2.1. EndoZyme
II
13.5.4.1.2.2. EndoZyme
II GO
13.5.4.1.2.3. EndoLisa
13.5.4.2. New Zealand Recombinant Factor C Assay Market Revenue (US$ Mn)
and Forecasts, By Application
13.5.4.2.1. In-process testing
13.5.4.2.2. Final product testing
13.5.4.2.3. Water testing
13.5.4.2.4. Testing plant-based material
13.5.4.3. New Zealand Recombinant Factor C Assay Market Revenue (US$ Mn)
and Forecasts, By End User
13.5.4.3.1. Pharmaceutical Companies
13.5.4.3.2. Biotechnology Companies
13.5.4.3.3. Medical Device Companies
13.5.4.3.4. Contract Research Organizations (CROs)
13.5.4.3.5. Research Centers
13.5.4.3.6. Non-profits and Federal Government
13.5.4.3.7. Others
13.5.5. Australia
13.5.5.1. Australia Recombinant Factor C Assay Market Revenue (US$ Mn)
and Forecasts, By Type
13.5.5.1.1. PyroGene
13.5.5.1.1.1. Routine
13.5.5.1.1.2. Inhibition/
Enhancement
13.5.5.1.1.3. RSE/
CSE
13.5.5.1.1.4. Initial
Qualification
13.5.5.1.2. EndoNext
13.5.5.1.2.1. EndoZyme
II
13.5.5.1.2.2. EndoZyme
II GO
13.5.5.1.2.3. EndoLisa
13.5.5.2. Australia Recombinant Factor C Assay Market Revenue (US$ Mn)
and Forecasts, By Application
13.5.5.2.1. In-process testing
13.5.5.2.2. Final product testing
13.5.5.2.3. Water testing
13.5.5.2.4. Testing plant-based material
13.5.5.3. Australia Recombinant Factor C Assay Market Revenue (US$ Mn)
and Forecasts, By End User
13.5.5.3.1. Pharmaceutical Companies
13.5.5.3.2. Biotechnology Companies
13.5.5.3.3. Medical Device Companies
13.5.5.3.4. Contract Research Organizations (CROs)
13.5.5.3.5. Research Centers
13.5.5.3.6. Non-profits and Federal Government
13.5.5.3.7. Others
13.5.6. South Korea
13.5.6.1. South Korea Recombinant Factor C Assay Market Revenue (US$ Mn)
and Forecasts, By Type
13.5.6.1.1. PyroGene
13.5.6.1.1.1. Routine
13.5.6.1.1.2. Inhibition/
Enhancement
13.5.6.1.1.3. RSE/
CSE
13.5.6.1.1.4. Initial
Qualification
13.5.6.1.2. EndoNext
13.5.6.1.2.1. EndoZyme
II
13.5.6.1.2.2. EndoZyme
II GO
13.5.6.1.2.3. EndoLisa
13.5.6.2. South Korea Recombinant Factor C Assay Market Revenue (US$ Mn)
and Forecasts, By Application
13.5.6.2.1. In-process testing
13.5.6.2.2. Final product testing
13.5.6.2.3. Water testing
13.5.6.2.4. Testing plant-based material
13.5.6.3. South Korea Recombinant Factor C Assay Market Revenue (US$ Mn)
and Forecasts, By End User
13.5.6.3.1. Pharmaceutical Companies
13.5.6.3.2. Biotechnology Companies
13.5.6.3.3. Medical Device Companies
13.5.6.3.4. Contract Research Organizations (CROs)
13.5.6.3.5. Research Centers
13.5.6.3.6. Non-profits and Federal Government
13.5.6.3.7. Others
13.5.7. Southeast Asia
13.5.7.1. Southeast Asia Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Type
13.5.7.1.1. PyroGene
13.5.7.1.1.1. Routine
13.5.7.1.1.2. Inhibition/
Enhancement
13.5.7.1.1.3. RSE/
CSE
13.5.7.1.1.4. Initial
Qualification
13.5.7.1.2. EndoNext
13.5.7.1.2.1. EndoZyme
II
13.5.7.1.2.2. EndoZyme
II GO
13.5.7.1.2.3. EndoLisa
13.5.7.2. Southeast Asia Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Application
13.5.7.2.1. In-process testing
13.5.7.2.2. Final product testing
13.5.7.2.3. Water testing
13.5.7.2.4. Testing plant-based material
13.5.7.3. Southeast Asia Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By End User
13.5.7.3.1. Pharmaceutical Companies
13.5.7.3.2. Biotechnology Companies
13.5.7.3.3. Medical Device Companies
13.5.7.3.4. Contract Research Organizations (CROs)
13.5.7.3.5. Research Centers
13.5.7.3.6. Non-profits and Federal Government
13.5.7.3.7. Others
13.5.7.4. Southeast Asia Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Country
13.5.7.4.1. Indonesia
13.5.7.4.2. Thailand
13.5.7.4.3. Malaysia
13.5.7.4.4. Singapore
13.5.7.4.5. Rest of Southeast Asia
13.5.8. Rest of Asia Pacific
13.5.8.1. Rest of Asia Pacific Recombinant Factor C Assay Market Revenue
(US$ Mn) and Forecasts, By Type
13.5.8.1.1. PyroGene
13.5.8.1.1.1. Routine
13.5.8.1.1.2. Inhibition/
Enhancement
13.5.8.1.1.3. RSE/
CSE
13.5.8.1.1.4. Initial
Qualification
13.5.8.1.2. EndoNext
13.5.8.1.2.1. EndoZyme
II
13.5.8.1.2.2. EndoZyme
II GO
13.5.8.1.2.3. EndoLisa
13.5.8.2. Rest of Asia Pacific Recombinant Factor C Assay Market Revenue
(US$ Mn) and Forecasts, By Application
13.5.8.2.1. In-process testing
13.5.8.2.2. Final product testing
13.5.8.2.3. Water testing
13.5.8.2.4. Testing plant-based material
13.5.8.3. Rest of Asia Pacific Recombinant Factor C Assay Market Revenue
(US$ Mn) and Forecasts, By End User
13.5.8.3.1. Pharmaceutical Companies
13.5.8.3.2. Biotechnology Companies
13.5.8.3.3. Medical Device Companies
13.5.8.3.4. Contract Research Organizations (CROs)
13.5.8.3.5. Research Centers
13.5.8.3.6. Non-profits and Federal Government
13.5.8.3.7. Others
13.6. Key Segment for Channeling Investments
13.6.1. By Country
13.6.2. By Type
13.6.3. By Application
13.6.4. By End User
14. Middle East and Africa Recombinant Factor C Assay Market
Analysis and Forecasts, 2023 - 2031
14.1. Overview
14.1.1. Middle East and Africa Recombinant Factor C Assay Market
Revenue (US$ Mn)
14.2. Middle East and Africa Recombinant Factor C Assay Market
Revenue (US$ Mn) and Forecasts, By Type
14.2.1. PyroGene
14.2.1.1. Routine
14.2.1.2. Inhibition/ Enhancement
14.2.1.3. RSE/ CSE
14.2.1.4. Initial Qualification
14.2.2. EndoNext
14.2.2.1. EndoZyme II
14.2.2.2. EndoZyme II GO
14.2.2.3. EndoLisa
14.3. Middle East and Africa Recombinant Factor C Assay Market
Revenue (US$ Mn) and Forecasts, By Application
14.3.1. In-process testing
14.3.2. Final product testing
14.3.3. Water testing
14.3.4. Testing plant-based material
14.4. Middle East and Africa Recombinant Factor C Assay Market
Revenue (US$ Mn) and Forecasts, By End User
14.4.1. Pharmaceutical Companies
14.4.2. Biotechnology Companies
14.4.3. Medical Device Companies
14.4.4. Contract Research Organizations (CROs)
14.4.5. Research Centers
14.4.6. Non-profits and Federal Government
14.4.7. Others
14.5. Middle East and Africa Recombinant Factor C Assay Market
Revenue (US$ Mn) and Forecasts, By Country
14.5.1. Saudi Arabia
14.5.1.1. Saudi Arabia Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Type
14.5.1.1.1. PyroGene
14.5.1.1.1.1. Routine
14.5.1.1.1.2. Inhibition/
Enhancement
14.5.1.1.1.3. RSE/
CSE
14.5.1.1.1.4. Initial
Qualification
14.5.1.1.2. EndoNext
14.5.1.1.2.1. EndoZyme
II
14.5.1.1.2.2. EndoZyme
II GO
14.5.1.1.2.3. EndoLisa
14.5.1.2. Saudi Arabia Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Application
14.5.1.2.1. In-process testing
14.5.1.2.2. Final product testing
14.5.1.2.3. Water testing
14.5.1.2.4. Testing plant-based material
14.5.1.3. Saudi Arabia Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By End User
14.5.1.3.1. Pharmaceutical Companies
14.5.1.3.2. Biotechnology Companies
14.5.1.3.3. Medical Device Companies
14.5.1.3.4. Contract Research Organizations (CROs)
14.5.1.3.5. Research Centers
14.5.1.3.6. Non-profits and Federal Government
14.5.1.3.7. Others
14.5.2. UAE
14.5.2.1. UAE Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Type
14.5.2.1.1. PyroGene
14.5.2.1.1.1. Routine
14.5.2.1.1.2. Inhibition/
Enhancement
14.5.2.1.1.3. RSE/
CSE
14.5.2.1.1.4. Initial
Qualification
14.5.2.1.2. EndoNext
14.5.2.1.2.1. EndoZyme
II
14.5.2.1.2.2. EndoZyme
II GO
14.5.2.1.2.3. EndoLisa
14.5.2.2. UAE Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Application
14.5.2.2.1. In-process testing
14.5.2.2.2. Final product testing
14.5.2.2.3. Water testing
14.5.2.2.4. Testing plant-based material
14.5.2.3. UAE Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By End User
14.5.2.3.1. Pharmaceutical Companies
14.5.2.3.2. Biotechnology Companies
14.5.2.3.3. Medical Device Companies
14.5.2.3.4. Contract Research Organizations (CROs)
14.5.2.3.5. Research Centers
14.5.2.3.6. Non-profits and Federal Government
14.5.2.3.7. Others
14.5.3. Egypt
14.5.3.1. Egypt Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Type
14.5.3.1.1. PyroGene
14.5.3.1.1.1. Routine
14.5.3.1.1.2. Inhibition/
Enhancement
14.5.3.1.1.3. RSE/
CSE
14.5.3.1.1.4. Initial
Qualification
14.5.3.1.2. EndoNext
14.5.3.1.2.1. EndoZyme
II
14.5.3.1.2.2. EndoZyme
II GO
14.5.3.1.2.3. EndoLisa
14.5.3.2. Egypt Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Application
14.5.3.2.1. In-process testing
14.5.3.2.2. Final product testing
14.5.3.2.3. Water testing
14.5.3.2.4. Testing plant-based material
14.5.3.3. Egypt Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By End User
14.5.3.3.1. Pharmaceutical Companies
14.5.3.3.2. Biotechnology Companies
14.5.3.3.3. Medical Device Companies
14.5.3.3.4. Contract Research Organizations (CROs)
14.5.3.3.5. Research Centers
14.5.3.3.6. Non-profits and Federal Government
14.5.3.3.7. Others
14.5.4. Kuwait
14.5.4.1. Kuwait Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Type
14.5.4.1.1. PyroGene
14.5.4.1.1.1. Routine
14.5.4.1.1.2. Inhibition/
Enhancement
14.5.4.1.1.3. RSE/
CSE
14.5.4.1.1.4. Initial
Qualification
14.5.4.1.2. EndoNext
14.5.4.1.2.1. EndoZyme
II
14.5.4.1.2.2. EndoZyme
II GO
14.5.4.1.2.3. EndoLisa
14.5.4.2. Kuwait Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Application
14.5.4.2.1. In-process testing
14.5.4.2.2. Final product testing
14.5.4.2.3. Water testing
14.5.4.2.4. Testing plant-based material
14.5.4.3. Kuwait Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By End User
14.5.4.3.1. Pharmaceutical Companies
14.5.4.3.2. Biotechnology Companies
14.5.4.3.3. Medical Device Companies
14.5.4.3.4. Contract Research Organizations (CROs)
14.5.4.3.5. Research Centers
14.5.4.3.6. Non-profits and Federal Government
14.5.4.3.7. Others
14.5.5. South Africa
14.5.5.1. South Africa Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Type
14.5.5.1.1. PyroGene
14.5.5.1.1.1. Routine
14.5.5.1.1.2. Inhibition/
Enhancement
14.5.5.1.1.3. RSE/
CSE
14.5.5.1.1.4. Initial
Qualification
14.5.5.1.2. EndoNext
14.5.5.1.2.1. EndoZyme
II
14.5.5.1.2.2. EndoZyme
II GO
14.5.5.1.2.3. EndoLisa
14.5.5.2. South Africa Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Application
14.5.5.2.1. In-process testing
14.5.5.2.2. Final product testing
14.5.5.2.3. Water testing
14.5.5.2.4. Testing plant-based material
14.5.5.3. South Africa Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By End User
14.5.5.3.1. Pharmaceutical Companies
14.5.5.3.2. Biotechnology Companies
14.5.5.3.3. Medical Device Companies
14.5.5.3.4. Contract Research Organizations (CROs)
14.5.5.3.5. Research Centers
14.5.5.3.6. Non-profits and Federal Government
14.5.5.3.7. Others
14.5.6. Rest of Middle East & Africa
14.5.6.1. Rest of Middle East & Africa Recombinant Factor C Assay
Market Revenue (US$ Mn) and Forecasts, By Type
14.5.6.1.1. PyroGene
14.5.6.1.1.1. Routine
14.5.6.1.1.2. Inhibition/
Enhancement
14.5.6.1.1.3. RSE/
CSE
14.5.6.1.1.4. Initial
Qualification
14.5.6.1.2. EndoNext
14.5.6.1.2.1. EndoZyme
II
14.5.6.1.2.2. EndoZyme
II GO
14.5.6.1.2.3. EndoLisa
14.5.6.2. Rest of Middle East & Africa Recombinant Factor C Assay
Market Revenue (US$ Mn) and Forecasts, By Application
14.5.6.2.1. In-process testing
14.5.6.2.2. Final product testing
14.5.6.2.3. Water testing
14.5.6.2.4. Testing plant-based material
14.5.6.3. Rest of Middle East & Africa Recombinant Factor C Assay
Market Revenue (US$ Mn) and Forecasts, By End User
14.5.6.3.1. Pharmaceutical Companies
14.5.6.3.2. Biotechnology Companies
14.5.6.3.3. Medical Device Companies
14.5.6.3.4. Contract Research Organizations (CROs)
14.5.6.3.5. Research Centers
14.5.6.3.6. Non-profits and Federal Government
14.5.6.3.7. Others
14.6. Key Segment for Channeling Investments
14.6.1. By Country
14.6.2. By Type
14.6.3. By Application
14.6.4. By End User
15. Latin America Recombinant Factor C Assay Market Analysis and
Forecasts, 2023 - 2031
15.1. Overview
15.1.1. Latin America Recombinant Factor C Assay Market Revenue (US$
Mn)
15.2. Latin America Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Type
15.2.1. PyroGene
15.2.1.1. Routine
15.2.1.2. Inhibition/ Enhancement
15.2.1.3. RSE/ CSE
15.2.1.4. Initial Qualification
15.2.2. EndoNext
15.2.2.1. EndoZyme II
15.2.2.2. EndoZyme II GO
15.2.2.3. EndoLisa
15.3. Latin America Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Application
15.3.1. In-process testing
15.3.2. Final product testing
15.3.3. Water testing
15.3.4. Testing plant-based material
15.4. Latin America Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By End User
15.4.1. Pharmaceutical Companies
15.4.2. Biotechnology Companies
15.4.3. Medical Device Companies
15.4.4. Contract Research Organizations (CROs)
15.4.5. Research Centers
15.4.6. Non-profits and Federal Government
15.4.7. Others
15.5. Latin America Recombinant Factor C Assay Market Revenue (US$
Mn) and Forecasts, By Country
15.5.1. Brazil
15.5.1.1. Brazil Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Type
15.5.1.1.1. PyroGene
15.5.1.1.1.1. Routine
15.5.1.1.1.2. Inhibition/
Enhancement
15.5.1.1.1.3. RSE/
CSE
15.5.1.1.1.4. Initial
Qualification
15.5.1.1.2. EndoNext
15.5.1.1.2.1. EndoZyme
II
15.5.1.1.2.2. EndoZyme
II GO
15.5.1.1.2.3. EndoLisa
15.5.1.2. Brazil Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By Application
15.5.1.2.1. In-process testing
15.5.1.2.2. Final product testing
15.5.1.2.3. Water testing
15.5.1.2.4. Testing plant-based material
15.5.1.3. Brazil Recombinant Factor C Assay Market Revenue (US$ Mn) and
Forecasts, By End User
15.5.1.3.1. Pharmaceutical Companies
15.5.1.3.2. Biotechnology Companies
15.5.1.3.3. Medical Device Companies
15.5.1.3.4. Contract Research Organizations (CROs)
15.5.1.3.5. Research Centers
15.5.1.3.6. Non-profits and Federal Government
15.5.1.3.7. Others
15.5.2. Argentina
15.5.2.1. Argentina Recombinant Factor C Assay Market Revenue (US$ Mn)
and Forecasts, By Type
15.5.2.1.1. PyroGene
15.5.2.1.1.1. Routine
15.5.2.1.1.2. Inhibition/
Enhancement
15.5.2.1.1.3. RSE/
CSE
15.5.2.1.1.4. Initial
Qualification
15.5.2.1.2. EndoNext
15.5.2.1.2.1. EndoZyme
II
15.5.2.1.2.2. EndoZyme
II GO
15.5.2.1.2.3. EndoLisa
15.5.2.2. Argentina Recombinant Factor C Assay Market Revenue (US$ Mn)
and Forecasts, By Application
15.5.2.2.1. In-process testing
15.5.2.2.2. Final product testing
15.5.2.2.3. Water testing
15.5.2.2.4. Testing plant-based material
15.5.2.3. Argentina Recombinant Factor C Assay Market Revenue (US$ Mn)
and Forecasts, By End User
15.5.2.3.1. Pharmaceutical Companies
15.5.2.3.2. Biotechnology Companies
15.5.2.3.3. Medical Device Companies
15.5.2.3.4. Contract Research Organizations (CROs)
15.5.2.3.5. Research Centers
15.5.2.3.6. Non-profits and Federal Government
15.5.2.3.7. Others
15.5.3. Rest of Latin America
15.5.3.1. Rest of Latin America Recombinant Factor C Assay Market
Revenue (US$ Mn) and Forecasts, By Type
15.5.3.1.1. PyroGene
15.5.3.1.1.1. Routine
15.5.3.1.1.2. Inhibition/
Enhancement
15.5.3.1.1.3. RSE/
CSE
15.5.3.1.1.4. Initial
Qualification
15.5.3.1.2. EndoNext
15.5.3.1.2.1. EndoZyme
II
15.5.3.1.2.2. EndoZyme
II GO
15.5.3.1.2.3. EndoLisa
15.5.3.2. Rest of Latin America Recombinant Factor C Assay Market
Revenue (US$ Mn) and Forecasts, By Application
15.5.3.2.1. In-process testing
15.5.3.2.2. Final product testing
15.5.3.2.3. Water testing
15.5.3.2.4. Testing plant-based material
15.5.3.3. Rest of Latin America Recombinant Factor C Assay Market
Revenue (US$ Mn) and Forecasts, By End User
15.5.3.3.1. Pharmaceutical Companies
15.5.3.3.2. Biotechnology Companies
15.5.3.3.3. Medical Device Companies
15.5.3.3.4. Contract Research Organizations (CROs)
15.5.3.3.5. Research Centers
15.5.3.3.6. Non-profits and Federal Government
15.5.3.3.7. Others
15.6. Key Segment for Channeling Investments
15.6.1. By Country
15.6.2. By Type
15.6.3. By Application
15.6.4. By End User
16. Competitive Benchmarking
16.1. Brand Benchmarking
16.2. Market Share Analysis, 2022
16.3. Global Presence and Growth Strategies
16.3.1. Mergers and Acquisitions
16.3.2. Product Launches
16.3.3. Investments Trends
16.3.4. R&D Initiatives
17. Player Profiles
17.1. BIOMERIEUX SA
17.1.1. Company Details
17.1.2. Company Overview
17.1.3. Product Offerings
17.1.4. Key Developments
17.1.5. Financial Analysis
17.1.6. SWOT Analysis
17.1.7. Business Strategies
17.2. BioVendor, LLC
17.2.1. Company Details
17.2.2. Company Overview
17.2.3. Product Offerings
17.2.4. Key Developments
17.2.5. Financial Analysis
17.2.6. SWOT Analysis
17.2.7. Business Strategies
17.3. Creative Biolabs
17.3.1. Company Details
17.3.2. Company Overview
17.3.3. Product Offerings
17.3.4. Key Developments
17.3.5. Financial Analysis
17.3.6. SWOT Analysis
17.3.7. Business Strategies
17.4. Eli Lilly
17.4.1. Company Details
17.4.2. Company Overview
17.4.3. Product Offerings
17.4.4. Key Developments
17.4.5. Financial Analysis
17.4.6. SWOT Analysis
17.4.7. Business Strategies
17.5. Lonza
17.5.1. Company Details
17.5.2. Company Overview
17.5.3. Product Offerings
17.5.4. Key Developments
17.5.5. Financial Analysis
17.5.6. SWOT Analysis
17.5.7. Business Strategies
17.6. Other Market Participants
18. Key Findings
Note: This ToC is tentative
and can be changed according to the research study conducted during the course
of report completion.
**Exclusive for Multi-User
and Enterprise User.
At Absolute Markets Insights, we are engaged in building both global as well as country specific reports. As a result, the approach taken for deriving the estimation and forecast for a specific country is a bit unique and different in comparison to the global research studies. In this case, we not only study the concerned market factors & trends prevailing in a particular country (from secondary research) but we also tend to calculate the actual market size & forecast from the revenue generated from the market participants involved in manufacturing or distributing the any concerned product. These companies can also be service providers. For analyzing any country specifically, we do consider the growth factors prevailing under the states/cities/county for the same. For instance, if we are analyzing an industry specific to United States, we primarily need to study about the states present under the same(where the product/service has the highest growth). Similar analysis will be followed by other countries. Our scope of the report changes with different markets.
Our research study is mainly implement through a mix of both secondary and primary research. Various sources such as industry magazines, trade journals, and government websites and trade associations are reviewed for gathering precise data. Primary interviews are conducted to validate the market size derived from secondary research. Industry experts, major manufacturers and distributors are contacted for further validation purpose on the current market penetration and growth trends.
Prominent participants in our primary research process include:
- Key Opinion Leaders namely the CEOs, CSOs, VPs, purchasing managers, amongst others
- Research and development participants, distributors/suppliers and subject matter experts
Secondary Research includes data extracted from paid data sources:
- Reuters
- Factiva
- Bloomberg
- One Source
- Hoovers
Research Methodology
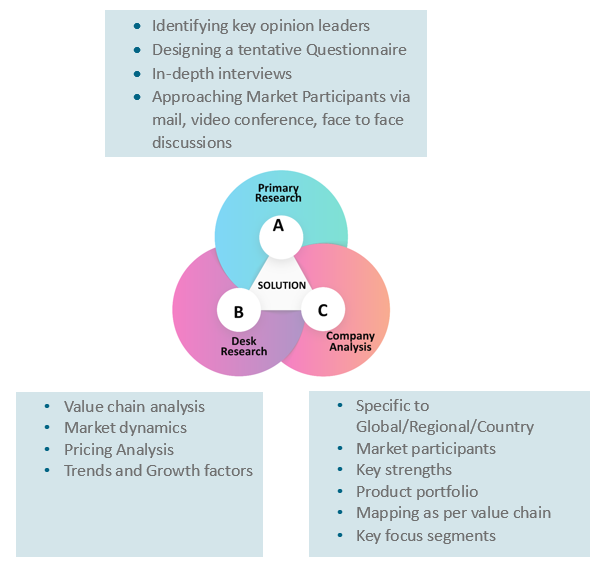
Key Inclusions

Reach to us
Call us on
+91-74002-42424
Drop us an email at
sales@absolutemarketsinsights.com
Why Absolute Markets Insights?
An effective strategy is the entity that influences a business to stand out of the crowd. An organization with a phenomenal strategy for success dependably has the edge over the rivals in the market. It offers the organizations a head start in planning their strategy. Absolute Market Insights is the new initiation in the industry that will furnish you with the lead your business needs. Absolute Market Insights is the best destination for your business intelligence and analytical solutions; essentially because our qualitative and quantitative sources of information are competent to give one-stop solutions. We inventively combine qualitative and quantitative research in accurate proportions to have the best report, which not only gives the most recent insights but also assists you to grow.

