Global mRNA Vaccines Market By Clinical Trial Phase (Preclinical , Phase I , Phase II , Phase III, Commercial); By Type (Non-replicating mRNA, In vivo self-replicating mRNA, In vitro dendritic cell non-replicating mRNA vaccine); By Indication/Application (Oncology (Solid Tumors, Melanoma, Prostate cancer, Ovarian cancer, Glioblastoma, HPV + Cancer , Renal Cancer , Others ), Infectious Diseases (COVID-19 vaccine, Rabies, Lassa, yellow fever, Respirational syncytial virus, Rota, Malaria , Universal influenza , Zika virus, Multiple sclerosis (MS) , Epstein-Barr virus (EBV), HIV vaccine, Others), Rare Diseases); By Vaccine Modality (Therapeutic, TME Modulation, Prophylactic, DC Vaccine, T Cell regulation); By Route of Administration (Intravenous/Intradermal, Intratumoral, Intramuscular); By Applications (Adolescents & Pediatric, Adults); By End-User (Healthcare Facilities, Government Vaccination Campaigns, Research Facilities, Others); By Region (North America (U.S., Canada, Mexico, Rest Of North America),
Industry Trends
Messenger RNA (mRNA) technology helps in training our cells to develop certain proteins that will induce an immune response in the human body. Research on mRNA vaccines has been ongoing for decades. The ease in mass production of mRNA vaccines, coupled with its ability to train certain cells to produce proteins that will attack foreign organisms that are entering the human body makes mRNA vaccines a viable source for preventing diseases like cancer, Zika, Yellow Fever, HIV, COVID-19, amongst others. The rise in demand for preventing various diseases at a faster rate with minimal side effects is leading to the growth of the global mRNA vaccines market.
COVID-19 Impact:
The COVID-19 pandemic has taken the world by storm. Pharmaceutical companies and research organizations are increasingly working on developing vaccines for coronavirus, and mRNA technology has received significant attention in the last couple of years. The U.S. Centers for Disease Control and Prevention (CDC) has recommended the use of mRNA COVID-19 vaccines from either Pfizer-BioNTech or Moderna. The greatest advantage with mRNA COVID-19 vaccines is that they do not cause infection in humans, as live viruses that cause the disease aren't used in these vaccines. These vaccines can be developed in the lab using easily available materials, which in turn increases the mass producing capability of mRNA vaccines. The success of COVID-19 mRNA vaccines has raised the investment in clinical trials for numerous diseases like renal cancer, malignant brain tumour, amongst others. Hence, the pandemic has raised the awareness regarding mRNA vaccines globally, which in turn is expected to lead to the growth in revenue in the global mRNA vaccines market in the coming years.
In terms of revenue, the global mRNA vaccine market is growing at a CAGR of 19.76% over the forecast period (2021 – 2029). The study analyses the market in terms of revenue across all the major regions, which have been bifurcated into countries.
Global mRNA Vaccines Market Revenue & Forecast, (US$ Million), 2017 – 2029
Clinical Trial Phase Outlook:
Companies are increasingly working on developing mRNA-based vaccines for lethal diseases. HIV, for instance, has affected around 37.7 million individuals till the end of 2020, and over 66.6% of these individuals are living in low income countries which are in the WHO Africa region. An estimated 680,000 perished due to HIV infection in 2020. HIV diagnostic, prevention and treatment is gaining significant attention, especially in Africa. Pharmaceutical giant Moderna has launched the clinical trials of mRNA HIV vaccines. The company has partnered with International AIDS Vaccine Initiative (IAVI), a non-profit organization that is working on reducing the global disease burden caused by HIV infections. The company hopes to trigger certain immune response in the human body to keep out HIV. mRNA technology is expected to speed up the immune response process in humans, which in turn will shorten the time required for commercializing the vaccine. Significant investment in the clinical trials of mRNA vaccine is being seen globally, especially in regions like Europe, North America and the Asia Pacific, which is propelling the demand of mRNA vaccines market.
Region Outlook:
North America accounted for the highest revenue share in the global mRNA vaccines market in 2020. The higher share in revenue can be attributed to the presence of prominent companies such as Moderna and Pfizer in the U.S. and Canada that are investing heavily in clinical trials in North America. Furthermore, the availability of advanced research facilities in the U.S. for the development of mRNA vaccines, especially for cancer related illnesses, is positively impacting the growth of the mRNA vaccines market in North America. Government bodies are increasingly concerned with the rise in cancer cases, especially among young adults. The change in lifestyle in North America as a result of factors such as industrialization and globalization is resulting in the deterioration of health in the region. Obesity, for instance, has seen a massive spike in cases in the last decade in the U.S. About 42.4% of the adults in the U.S. were reported to be obese in 2018. Therefore, companies are increasingly trying to develop vaccines that can prevent the onset of diseases like cancer, and other infectious diseases.
Competitive Landscape
The report provides both, qualitative and quantitative research of the global mRNA vaccines market, as well as provides comprehensive insights and development methods adopted by the key contenders. The report also offers extensive research on the key players in this market and details on the competitiveness of these players. Key business strategies such as mergers and acquisitions (M&A), affiliations, collaborations, and contracts adopted by these major market participants are also recognized and analysed in the report. For each company, the report studies their global presence, competitors, service offerings and specification amongst others.
Some of key players operating in the global mRNA vaccines market are:
- Argos Therapeutics
- BioNTech SE
- cōIMMUNE
- CureVac AG
- eTheRNA
- Genentech, Inc
- Immunomic Therapeutics, Inc.
- Moderna, Inc.
- Pfizer Inc.
- Other Market Participants
Global mRNA Vaccines Market
By Clinical Trial Phase
- Preclinical
- Phase I
- Phase II
- Phase III
- Commercial
By Type
- Non-replicating mRNA
- In vivo self-replicating mRNA
- In vitro dendritic cell non-replicating mRNA vaccine
By Indication/Application
- Oncology
- Solid Tumors
- Melanoma
- Prostate cancer
- Ovarian cancer
- Glioblastoma
- HPV + Cancer
- Renal Cancer
- Others
- Infectious Diseases
- COVID-19 vaccine
- Rabies
- Lassa, yellow fever
- Respirational syncytial virus
- Rota
- Malaria
- Universal influenza
- Zika virus
- Multiple sclerosis (MS)
- Epstein-Barr virus (EBV)
- HIV vaccine
- Others
- Rare Diseases
By Vaccine Modality
- Therapeutic
- TME Modulation
- Prophylactic
- DC Vaccine
- T Cell regulation
By Route of Administration
- Intravenous/Intradermal
- Intratumoral
- Intramuscular
By Applications
- Adolescents & Pediatric
- Adults
By End-User
- Healthcare Facilities
- Government Vaccination Campaigns
- Research Facilities
- Others
By Region
- North America
- U.S.
- Canada
- Mexico
- Rest of North America
- Europe
- France
- The UK
- Spain
- Germany
- Italy
- Nordic Countries
- Denmark
- Finland
- Iceland
- Sweden
- Norway
- Benelux Union
- Belgium
- The Netherlands
- Luxembourg
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- New Zealand
- Australia
- South Korea
- Southeast Asia
- Indonesia
- Thailand
- Malaysia
- Singapore
- Rest of Southeast Asia
- Rest of Asia Pacific
- Middle East & Africa
- Saudi Arabia
- UAE
- Egypt
- Kuwait
- South Africa
- Rest of Middle East & Africa
- Latin America
- Brazil
- Argentina
- Rest of Latin America
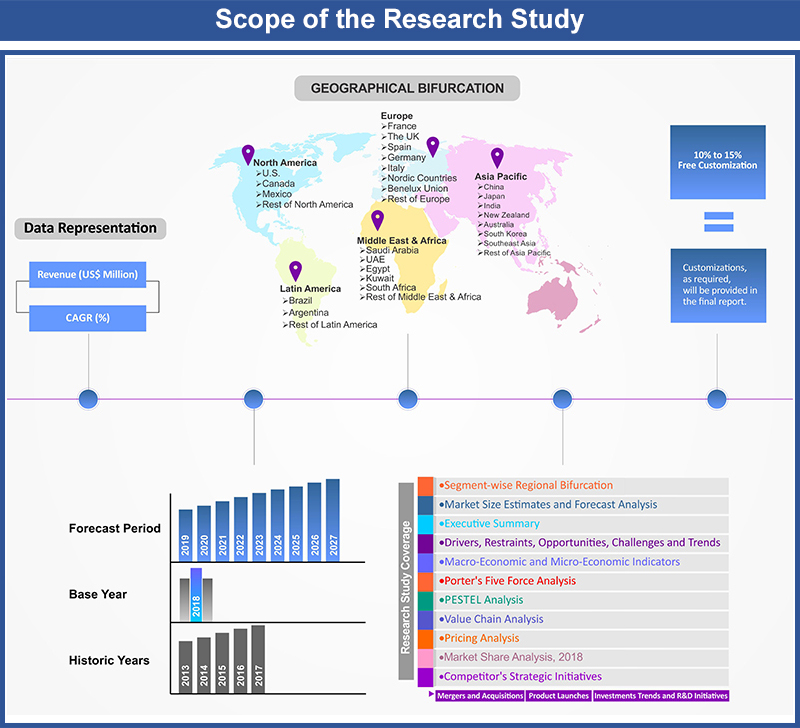
Table of Contents
1.
Market
Scope
1.1. Market Segmentation
1.2. Years Considered
1.2.1. Historic Years: 2015 - 2019
1.2.2. Base Year: 2020
1.2.3. Forecast Years: 2021 – 2029
2.
Key
Target Audiences
3.
Research
Methodology
3.1. Primary Research
3.1.1. Research Questionnaire
3.1.2. Global Percentage Breakdown
3.1.3. Primary Interviews: Key Opinion Leaders
(KOLs)
3.2. Secondary Research
3.2.1. Paid Databases
3.2.2. Secondary Sources
3.3. Market Size Estimates
3.3.1. Top-Down Approach
3.3.2. Bottom-Up Approach
3.4. Data Triangulation Methodology
3.5. Research Assumptions
4.
Recommendations
and Insights from AMI’s Perspective**
5.
Holistic
Overview of mRNA Vaccines Market
6.
Market
Synopsis: mRNA Vaccines Market
7.
mRNA
Vaccines Market Analysis: Qualitative Perspective
7.1. Introduction
7.1.1. Product Definition
7.1.2. Industry Development
7.2. Market Dynamics
7.2.1. Drivers
7.2.2. Restraints
7.2.3. Opportunities
7.2.4. Challenges
7.3. Trends in mRNA Vaccines Market
7.4. Market Determinants Radar Chart
7.5. Macro-Economic and Micro-Economic Indicators:
mRNA Vaccines Market
7.6. Porter’s Five Force Analysis
7.7. Impact of Covid-19 on mRNA Vaccines Market
8.
Global mRNA
Vaccines Market Analysis and Forecasts, 2021 – 2029
8.1. Overview
8.1.1. Global mRNA Vaccines Market Revenue (US$ Mn)
8.2. Global mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Clinical Trial Phase
8.2.1. Preclinical
8.2.1.1. Definition
8.2.1.2. Market Estimation and Penetration, 2015 -
2020
8.2.1.3. Market Forecast, 2021 - 2029
8.2.1.4. Compound Annual Growth Rate (CAGR)
8.2.1.5. Regional Bifurcation
8.2.1.5.1.
North
America
8.2.1.5.1.1. Market Estimation, 2015 - 2020
8.2.1.5.1.2. Market Forecast, 2021 - 2029
8.2.1.5.2.
Europe
8.2.1.5.2.1. Market Estimation, 2015 - 2020
8.2.1.5.2.2. Market Forecast, 2021 - 2029
8.2.1.5.3.
Asia
Pacific
8.2.1.5.3.1. Market Estimation, 2015 - 2020
8.2.1.5.3.2. Market Forecast, 2021 - 2029
8.2.1.5.4.
Middle
East and Africa
8.2.1.5.4.1. Market Estimation, 2015 - 2020
8.2.1.5.4.2. Market Forecast, 2021 - 2029
8.2.1.5.5.
Latin
America
8.2.1.5.5.1. Market Estimation, 2015 - 2020
8.2.1.5.5.2. Market Forecast, 2021 - 2029
8.2.2. Phase
I
8.2.2.1. Definition
8.2.2.2. Market Estimation and Penetration, 2015 -
2020
8.2.2.3. Market Forecast, 2021 - 2029
8.2.2.4. Compound Annual Growth Rate (CAGR)
8.2.2.5. Regional Bifurcation
8.2.2.5.1.
North
America
8.2.2.5.1.1. Market Estimation, 2015 - 2020
8.2.2.5.1.2. Market Forecast, 2021 - 2029
8.2.2.5.2.
Europe
8.2.2.5.2.1. Market Estimation, 2015 - 2020
8.2.2.5.2.2. Market Forecast, 2021 - 2029
8.2.2.5.3.
Asia
Pacific
8.2.2.5.3.1. Market Estimation, 2015 - 2020
8.2.2.5.3.2. Market Forecast, 2021 - 2029
8.2.2.5.4.
Middle
East and Africa
8.2.2.5.4.1. Market Estimation, 2015 - 2020
8.2.2.5.4.2. Market Forecast, 2021 - 2029
8.2.2.5.5.
Latin
America
8.2.2.5.5.1. Market Estimation, 2015 - 2020
8.2.2.5.5.2. Market Forecast, 2021 - 2029
8.2.3. Phase II
8.2.3.1. Definition
8.2.3.2. Market Estimation and Penetration, 2015 -
2020
8.2.3.3. Market Forecast, 2021 - 2029
8.2.3.4. Compound Annual Growth Rate (CAGR)
8.2.3.5. Regional Bifurcation
8.2.3.5.1.
North
America
8.2.3.5.1.1. Market Estimation, 2015 - 2020
8.2.3.5.1.2. Market Forecast, 2021 - 2029
8.2.3.5.2.
Europe
8.2.3.5.2.1. Market Estimation, 2015 - 2020
8.2.3.5.2.2. Market Forecast, 2021 - 2029
8.2.3.5.3.
Asia
Pacific
8.2.3.5.3.1. Market Estimation, 2015 - 2020
8.2.3.5.3.2. Market Forecast, 2021 - 2029
8.2.3.5.4.
Middle
East and Africa
8.2.3.5.4.1. Market Estimation, 2015 - 2020
8.2.3.5.4.2. Market Forecast, 2021 - 2029
8.2.3.5.5.
Latin
America
8.2.3.5.5.1. Market Estimation, 2015 - 2020
8.2.3.5.5.2. Market Forecast, 2021 - 2029
8.2.4. Phase III
8.2.4.1. Definition
8.2.4.2. Market Estimation and Penetration, 2015 -
2020
8.2.4.3. Market Forecast, 2021 - 2029
8.2.4.4. Compound Annual Growth Rate (CAGR)
8.2.4.5. Regional Bifurcation
8.2.4.5.1.
North
America
8.2.4.5.1.1. Market Estimation, 2015 - 2020
8.2.4.5.1.2. Market Forecast, 2021 - 2029
8.2.4.5.2.
Europe
8.2.4.5.2.1. Market Estimation, 2015 - 2020
8.2.4.5.2.2. Market Forecast, 2021 - 2029
8.2.4.5.3.
Asia
Pacific
8.2.4.5.3.1. Market Estimation, 2015 - 2020
8.2.4.5.3.2. Market Forecast, 2021 - 2029
8.2.4.5.4.
Middle
East and Africa
8.2.4.5.4.1. Market Estimation, 2015 - 2020
8.2.4.5.4.2. Market Forecast, 2021 - 2029
8.2.4.5.5.
Latin
America
8.2.4.5.5.1. Market Estimation, 2015 - 2020
8.2.4.5.5.2. Market Forecast, 2021 - 2029
8.2.5. Commercial
8.2.5.1. Definition
8.2.5.2. Market Estimation and Penetration, 2015 -
2020
8.2.5.3. Market Forecast, 2021 - 2029
8.2.5.4. Compound Annual Growth Rate (CAGR)
8.2.5.5. Regional Bifurcation
8.2.5.5.1.
North
America
8.2.5.5.1.1. Market Estimation, 2015 - 2020
8.2.5.5.1.2. Market Forecast, 2021 - 2029
8.2.5.5.2.
Europe
8.2.5.5.2.1. Market Estimation, 2015 - 2020
8.2.5.5.2.2. Market Forecast, 2021 - 2029
8.2.5.5.3.
Asia
Pacific
8.2.5.5.3.1. Market Estimation, 2015 - 2020
8.2.5.5.3.2. Market Forecast, 2021 - 2029
8.2.5.5.4.
Middle
East and Africa
8.2.5.5.4.1. Market Estimation, 2015 - 2020
8.2.5.5.4.2. Market Forecast, 2021 - 2029
8.2.5.5.5.
Latin
America
8.2.5.5.5.1. Market Estimation, 2015 - 2020
8.2.5.5.5.2. Market Forecast, 2021 - 2029
8.3. Key Segment for Channeling Investments
8.3.1. By Clinical Trial Phase
9.
Global mRNA
Vaccines Market Analysis and Forecasts, 2021 – 2029
9.1. Overview
9.2. Global mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Type
9.2.1. Non-replicating mRNA
9.2.1.1. Definition
9.2.1.2. Market Estimation and Penetration, 2015 -
2020
9.2.1.3. Market Forecast, 2021 - 2029
9.2.1.4. Compound Annual Growth Rate (CAGR)
9.2.1.5. Regional Bifurcation
9.2.1.5.1.
North
America
9.2.1.5.1.1. Market Estimation, 2015 - 2020
9.2.1.5.1.2. Market Forecast, 2021 - 2029
9.2.1.5.2.
Europe
9.2.1.5.2.1. Market Estimation, 2015 - 2020
9.2.1.5.2.2. Market Forecast, 2021 - 2029
9.2.1.5.3.
Asia
Pacific
9.2.1.5.3.1. Market Estimation, 2015 - 2020
9.2.1.5.3.2. Market Forecast, 2021 - 2029
9.2.1.5.4.
Middle
East and Africa
9.2.1.5.4.1. Market Estimation, 2015 - 2020
9.2.1.5.4.2. Market Forecast, 2021 - 2029
9.2.1.5.5.
Latin
America
9.2.1.5.5.1. Market Estimation, 2015 - 2020
9.2.1.5.5.2. Market Forecast, 2021 - 2029
9.2.2. In vivo self-replicating mRNA
9.2.2.1. Definition
9.2.2.2. Market Estimation and Penetration, 2015 -
2020
9.2.2.3. Market Forecast, 2021 - 2029
9.2.2.4. Compound Annual Growth Rate (CAGR)
9.2.2.5. Regional Bifurcation
9.2.2.5.1.
North
America
9.2.2.5.1.1. Market Estimation, 2015 - 2020
9.2.2.5.1.2. Market Forecast, 2021 - 2029
9.2.2.5.2.
Europe
9.2.2.5.2.1. Market Estimation, 2015 - 2020
9.2.2.5.2.2. Market Forecast, 2021 - 2029
9.2.2.5.3.
Asia
Pacific
9.2.2.5.3.1. Market Estimation, 2015 - 2020
9.2.2.5.3.2. Market Forecast, 2021 - 2029
9.2.2.5.4.
Middle
East and Africa
9.2.2.5.4.1. Market Estimation, 2015 - 2020
9.2.2.5.4.2. Market Forecast, 2021 - 2029
9.2.2.5.5.
Latin
America
9.2.2.5.5.1. Market Estimation, 2015 - 2020
9.2.2.5.5.2. Market Forecast, 2021 - 2029
9.2.3. In vitro dendritic cell non-replicating mRNA
vaccine
9.2.3.1. Definition
9.2.3.2. Market Estimation and Penetration, 2015 -
2020
9.2.3.3. Market Forecast, 2021 - 2029
9.2.3.4. Compound Annual Growth Rate (CAGR)
9.2.3.5. Regional Bifurcation
9.2.3.5.1.
North
America
9.2.3.5.1.1. Market Estimation, 2015 - 2020
9.2.3.5.1.2. Market Forecast, 2021 - 2029
9.2.3.5.2.
Europe
9.2.3.5.2.1. Market Estimation, 2015 - 2020
9.2.3.5.2.2. Market Forecast, 2021 - 2029
9.2.3.5.3.
Asia
Pacific
9.2.3.5.3.1. Market Estimation, 2015 - 2020
9.2.3.5.3.2. Market Forecast, 2021 - 2029
9.2.3.5.4.
Middle
East and Africa
9.2.3.5.4.1. Market Estimation, 2015 - 2020
9.2.3.5.4.2. Market Forecast, 2021 - 2029
9.2.3.5.5.
Latin
America
9.2.3.5.5.1. Market Estimation, 2015 - 2020
9.2.3.5.5.2. Market Forecast, 2021 - 2029
9.3. Key Segment for Channeling Investments
9.3.1. By Type
10. Global mRNA Vaccines Market Analysis and
Forecasts, 2021 – 2029
10.1. Overview
10.2. Global mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Vaccine Modality
10.2.1. Therapeutic
10.2.1.1. Definition
10.2.1.2. Market Estimation and Penetration, 2015 -
2020
10.2.1.3. Market Forecast, 2021 - 2029
10.2.1.4. Compound Annual Growth Rate (CAGR)
10.2.1.5. Regional Bifurcation
10.2.1.5.1.
North
America
10.2.1.5.1.1. Market Estimation, 2015 - 2020
10.2.1.5.1.2. Market Forecast, 2021 - 2029
10.2.1.5.2.
Europe
10.2.1.5.2.1. Market Estimation, 2015 - 2020
10.2.1.5.2.2. Market Forecast, 2021 - 2029
10.2.1.5.3.
Asia
Pacific
10.2.1.5.3.1. Market Estimation, 2015 - 2020
10.2.1.5.3.2. Market Forecast, 2021 - 2029
10.2.1.5.4.
Middle
East and Africa
10.2.1.5.4.1. Market Estimation, 2015 - 2020
10.2.1.5.4.2. Market Forecast, 2021 - 2029
10.2.1.5.5.
Latin
America
10.2.1.5.5.1. Market Estimation, 2015 - 2020
10.2.1.5.5.2. Market Forecast, 2021 - 2029
10.2.2. TME Modulation
10.2.2.1. Definition
10.2.2.2. Market Estimation and Penetration, 2015 -
2020
10.2.2.3. Market Forecast, 2021 - 2029
10.2.2.4. Compound Annual Growth Rate (CAGR)
10.2.2.5. Regional Bifurcation
10.2.2.5.1.
North
America
10.2.2.5.1.1. Market Estimation, 2015 - 2020
10.2.2.5.1.2. Market Forecast, 2021 - 2029
10.2.2.5.2.
Europe
10.2.2.5.2.1. Market Estimation, 2015 - 2020
10.2.2.5.2.2. Market Forecast, 2021 - 2029
10.2.2.5.3.
Asia
Pacific
10.2.2.5.3.1. Market Estimation, 2015 - 2020
10.2.2.5.3.2. Market Forecast, 2021 - 2029
10.2.2.5.4.
Middle
East and Africa
10.2.2.5.4.1. Market Estimation, 2015 - 2020
10.2.2.5.4.2. Market Forecast, 2021 - 2029
10.2.2.5.5.
Latin America
10.2.2.5.5.1. Market Estimation, 2015 - 2020
10.2.2.5.5.2. Market Forecast, 2021 - 2029
10.2.3. Prophylactic
10.2.3.1. Definition
10.2.3.2. Market Estimation and Penetration, 2015 -
2020
10.2.3.3. Market Forecast, 2021 - 2029
10.2.3.4. Compound Annual Growth Rate (CAGR)
10.2.3.5. Regional Bifurcation
10.2.3.5.1.
North
America
10.2.3.5.1.1. Market Estimation, 2015 - 2020
10.2.3.5.1.2. Market Forecast, 2021 - 2029
10.2.3.5.2.
Europe
10.2.3.5.2.1. Market Estimation, 2015 - 2020
10.2.3.5.2.2. Market Forecast, 2021 - 2029
10.2.3.5.3.
Asia
Pacific
10.2.3.5.3.1. Market Estimation, 2015 - 2020
10.2.3.5.3.2. Market Forecast, 2021 - 2029
10.2.3.5.4.
Middle
East and Africa
10.2.3.5.4.1. Market Estimation, 2015 - 2020
10.2.3.5.4.2. Market Forecast, 2021 - 2029
10.2.3.5.5.
Latin
America
10.2.3.5.5.1. Market Estimation, 2015 - 2020
10.2.3.5.5.2. Market Forecast, 2021 - 2029
10.2.4. DC Vaccine
10.2.4.1. Definition
10.2.4.2. Market Estimation and Penetration, 2015 -
2020
10.2.4.3. Market Forecast, 2021 - 2029
10.2.4.4. Compound Annual Growth Rate (CAGR)
10.2.4.5. Regional Bifurcation
10.2.4.5.1.
North America
10.2.4.5.1.1. Market Estimation, 2015 - 2020
10.2.4.5.1.2. Market Forecast, 2021 - 2029
10.2.4.5.2.
Europe
10.2.4.5.2.1. Market Estimation, 2015 - 2020
10.2.4.5.2.2. Market Forecast, 2021 - 2029
10.2.4.5.3.
Asia
Pacific
10.2.4.5.3.1. Market Estimation, 2015 - 2020
10.2.4.5.3.2. Market Forecast, 2021 - 2029
10.2.4.5.4.
Middle
East and Africa
10.2.4.5.4.1. Market Estimation, 2015 - 2020
10.2.4.5.4.2. Market Forecast, 2021 - 2029
10.2.4.5.5.
Latin
America
10.2.4.5.5.1. Market Estimation, 2015 - 2020
10.2.4.5.5.2. Market Forecast, 2021 - 2029
10.2.5. T Cell regulation
10.2.5.1. Definition
10.2.5.2. Market Estimation and Penetration, 2015 -
2020
10.2.5.3. Market Forecast, 2021 - 2029
10.2.5.4. Compound Annual Growth Rate (CAGR)
10.2.5.5. Regional Bifurcation
10.2.5.5.1.
North
America
10.2.5.5.1.1. Market Estimation, 2015 - 2020
10.2.5.5.1.2. Market Forecast, 2021 - 2029
10.2.5.5.2.
Europe
10.2.5.5.2.1. Market Estimation, 2015 - 2020
10.2.5.5.2.2. Market Forecast, 2021 - 2029
10.2.5.5.3.
Asia
Pacific
10.2.5.5.3.1. Market Estimation, 2015 - 2020
10.2.5.5.3.2. Market Forecast, 2021 - 2029
10.2.5.5.4.
Middle East
and Africa
10.2.5.5.4.1. Market Estimation, 2015 - 2020
10.2.5.5.4.2. Market Forecast, 2021 - 2029
10.2.5.5.5.
Latin
America
10.2.5.5.5.1. Market Estimation, 2015 - 2020
10.2.5.5.5.2. Market Forecast, 2021 - 2029
10.3. Key Segment for Channeling Investments
10.3.1. By Vaccine Modality
11. Global mRNA Vaccines Market Analysis and Forecasts,
2021 – 2029
11.1. Overview
11.2. Global mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Route of Administration
11.2.1. Intravenous/Intradermal
11.2.1.1. Definition
11.2.1.2. Market Estimation and Penetration, 2015 -
2020
11.2.1.3. Market Forecast, 2021 - 2029
11.2.1.4. Compound Annual Growth Rate (CAGR)
11.2.1.5. Regional Bifurcation
11.2.1.5.1.
North
America
11.2.1.5.1.1. Market Estimation, 2015 - 2020
11.2.1.5.1.2. Market Forecast, 2021 - 2029
11.2.1.5.2.
Europe
11.2.1.5.2.1. Market Estimation, 2015 - 2020
11.2.1.5.2.2. Market Forecast, 2021 - 2029
11.2.1.5.3.
Asia
Pacific
11.2.1.5.3.1. Market Estimation, 2015 - 2020
11.2.1.5.3.2. Market Forecast, 2021 - 2029
11.2.1.5.4.
Middle East
and Africa
11.2.1.5.4.1. Market Estimation, 2015 - 2020
11.2.1.5.4.2. Market Forecast, 2021 - 2029
11.2.1.5.5.
Latin
America
11.2.1.5.5.1. Market Estimation, 2015 - 2020
11.2.1.5.5.2. Market Forecast, 2021 - 2029
11.2.2. Intratumoral
11.2.2.1. Definition
11.2.2.2. Market Estimation and Penetration, 2015 -
2020
11.2.2.3. Market Forecast, 2021 - 2029
11.2.2.4. Compound Annual Growth Rate (CAGR)
11.2.2.5. Regional Bifurcation
11.2.2.5.1.
North
America
11.2.2.5.1.1. Market Estimation, 2015 - 2020
11.2.2.5.1.2. Market Forecast, 2021 - 2029
11.2.2.5.2.
Europe
11.2.2.5.2.1. Market Estimation, 2015 - 2020
11.2.2.5.2.2. Market Forecast, 2021 - 2029
11.2.2.5.3.
Asia
Pacific
11.2.2.5.3.1. Market Estimation, 2015 - 2020
11.2.2.5.3.2. Market Forecast, 2021 - 2029
11.2.2.5.4.
Middle
East and Africa
11.2.2.5.4.1. Market Estimation, 2015 - 2020
11.2.2.5.4.2. Market Forecast, 2021 - 2029
11.2.2.5.5.
Latin
America
11.2.2.5.5.1. Market Estimation, 2015 - 2020
11.2.2.5.5.2. Market Forecast, 2021 - 2029
11.2.3. Intramuscular
11.2.3.1. Definition
11.2.3.2. Market Estimation and Penetration, 2015 -
2020
11.2.3.3. Market Forecast, 2021 - 2029
11.2.3.4. Compound Annual Growth Rate (CAGR)
11.2.3.5. Regional Bifurcation
11.2.3.5.1.
North
America
11.2.3.5.1.1. Market Estimation, 2015 - 2020
11.2.3.5.1.2. Market Forecast, 2021 - 2029
11.2.3.5.2.
Europe
11.2.3.5.2.1. Market Estimation, 2015 - 2020
11.2.3.5.2.2. Market Forecast, 2021 - 2029
11.2.3.5.3.
Asia
Pacific
11.2.3.5.3.1. Market Estimation, 2015 - 2020
11.2.3.5.3.2. Market Forecast, 2021 - 2029
11.2.3.5.4.
Middle
East and Africa
11.2.3.5.4.1. Market Estimation, 2015 - 2020
11.2.3.5.4.2. Market Forecast, 2021 - 2029
11.2.3.5.5.
Latin
America
11.2.3.5.5.1. Market Estimation, 2015 - 2020
11.2.3.5.5.2. Market Forecast, 2021 - 2029
11.3. Key Segment for Channeling Investments
11.3.1. By Route of Administration
12. Global mRNA Vaccines Market Analysis and
Forecasts, 2021 – 2029
12.1. Overview
12.2. Global mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Indication
12.2.1. Oncology (Definition, Market Estimation and
Penetration, 2015 - 2020, Market Estimation (2015 - 2020), Market Forecast
(2021 - 2029), Compound Annual Growth Rate (CAGR), Regional Bifurcation (North
America, Europe, Asia Pacific, Middle East and Africa, Latin America) and
Information on Solid Tumors, Melanoma, Prostate cancer, Ovarian cancer,
Glioblastoma, HPV + Cancer, Renal Cancer, Others)
12.2.1.1. Solid Tumors
12.2.1.2. Melanoma
12.2.1.3. Prostate cancer
12.2.1.4. Ovarian cancer
12.2.1.5. Glioblastoma
12.2.1.6. HPV + Cancer
12.2.1.7. Renal Cancer
12.2.1.8. Others
12.2.2. Infectious Diseases (Definition, Market
Estimation and Penetration, 2015 - 2020, Market Estimation (2015 - 2020),
Market Forecast (2021 - 2029), Compound Annual Growth Rate (CAGR), Regional
Bifurcation (North America, Europe, Asia Pacific, Middle East and Africa, Latin
America) and Information on COVID-19 vaccine, Rabies, Lassa, yellow fever,
Respirational syncytial virus, Rota,
Malaria, Universal influenza, Zika virus, Multiple sclerosis (MS), Epstein-Barr
virus (EBV), HIV vaccine, Others)
12.2.2.1. COVID-19 vaccine
12.2.2.2. Rabies
12.2.2.3. Lassa, yellow fever
12.2.2.4. Respirational syncytial virus
12.2.2.5. Rota
12.2.2.6. Malaria
12.2.2.7. Universal influenza
12.2.2.8. Zika virus
12.2.2.9. Multiple sclerosis (MS)
12.2.2.10. Epstein-Barr virus (EBV)
12.2.2.11. HIV vaccine
12.2.2.12. Others
12.2.3. Rare Diseases
12.2.3.1. Definition
12.2.3.2. Market Estimation and Penetration, 2015 -
2020
12.2.3.3. Market Forecast, 2021 - 2029
12.2.3.4. Compound Annual Growth Rate (CAGR)
12.2.3.5. Regional Bifurcation
12.2.3.5.1.
North
America
12.2.3.5.1.1. Market Estimation, 2015 - 2020
12.2.3.5.1.2. Market Forecast, 2021 - 2029
12.2.3.5.2.
Europe
12.2.3.5.2.1. Market Estimation, 2015 - 2020
12.2.3.5.2.2. Market Forecast, 2021 - 2029
12.2.3.5.3.
Asia
Pacific
12.2.3.5.3.1. Market Estimation, 2015 - 2020
12.2.3.5.3.2. Market Forecast, 2021 - 2029
12.2.3.5.4.
Middle
East and Africa
12.2.3.5.4.1. Market Estimation, 2015 - 2020
12.2.3.5.4.2. Market Forecast, 2021 - 2029
12.2.3.5.5.
Latin
America
12.2.3.5.5.1. Market Estimation, 2015 - 2020
12.2.3.5.5.2. Market Forecast, 2021 - 2029
12.3. Key Segment for Channeling Investments
12.3.1. By Indication
13. Global mRNA Vaccines Market Analysis and
Forecasts, 2021 – 2029
13.1. Overview
13.2. Global mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Applications
13.2.1. Adolescents and Pediatric
13.2.1.1. Definition
13.2.1.2. Market Estimation and Penetration, 2015 -
2020
13.2.1.3. Market Forecast, 2021 - 2029
13.2.1.4. Compound Annual Growth Rate (CAGR)
13.2.1.5. Regional Bifurcation
13.2.1.5.1.
North
America
13.2.1.5.1.1. Market Estimation, 2015 - 2020
13.2.1.5.1.2. Market Forecast, 2021 - 2029
13.2.1.5.2.
Europe
13.2.1.5.2.1. Market Estimation, 2015 - 2020
13.2.1.5.2.2. Market Forecast, 2021 - 2029
13.2.1.5.3.
Asia
Pacific
13.2.1.5.3.1. Market Estimation, 2015 - 2020
13.2.1.5.3.2. Market Forecast, 2021 - 2029
13.2.1.5.4.
Middle
East and Africa
13.2.1.5.4.1. Market Estimation, 2015 - 2020
13.2.1.5.4.2. Market Forecast, 2021 - 2029
13.2.1.5.5.
Latin
America
13.2.1.5.5.1. Market Estimation, 2015 - 2020
13.2.1.5.5.2. Market Forecast, 2021 - 2029
13.2.2. Adults
13.2.2.1. Definition
13.2.2.2. Market Estimation and Penetration, 2015 -
2020
13.2.2.3. Market Forecast, 2021 - 2029
13.2.2.4. Compound Annual Growth Rate (CAGR)
13.2.2.5. Regional Bifurcation
13.2.2.5.1.
North
America
13.2.2.5.1.1. Market Estimation, 2015 - 2020
13.2.2.5.1.2. Market Forecast, 2021 - 2029
13.2.2.5.2.
Europe
13.2.2.5.2.1. Market Estimation, 2015 - 2020
13.2.2.5.2.2. Market Forecast, 2021 - 2029
13.2.2.5.3.
Asia
Pacific
13.2.2.5.3.1. Market Estimation, 2015 - 2020
13.2.2.5.3.2. Market Forecast, 2021 - 2029
13.2.2.5.4.
Middle
East and Africa
13.2.2.5.4.1. Market Estimation, 2015 - 2020
13.2.2.5.4.2. Market Forecast, 2021 - 2029
13.2.2.5.5.
Latin
America
13.2.2.5.5.1. Market Estimation, 2015 - 2020
13.2.2.5.5.2. Market Forecast, 2021 - 2029
13.3. Key Segment for Channeling Investments
13.3.1. By Applications
14. Global mRNA Vaccines Market Analysis and
Forecasts, 2021 – 2029
14.1. Overview
14.2. Global mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By End-User
14.2.1. Healthcare Facilities
14.2.1.1. Definition
14.2.1.2. Market Estimation and Penetration, 2015 -
2020
14.2.1.3. Market Forecast, 2021 - 2029
14.2.1.4. Compound Annual Growth Rate (CAGR)
14.2.1.5. Regional Bifurcation
14.2.1.5.1.
North
America
14.2.1.5.1.1. Market Estimation, 2015 - 2020
14.2.1.5.1.2. Market Forecast, 2021 - 2029
14.2.1.5.2.
Europe
14.2.1.5.2.1. Market Estimation, 2015 - 2020
14.2.1.5.2.2. Market Forecast, 2021 - 2029
14.2.1.5.3.
Asia
Pacific
14.2.1.5.3.1. Market Estimation, 2015 - 2020
14.2.1.5.3.2. Market Forecast, 2021 - 2029
14.2.1.5.4.
Middle
East and Africa
14.2.1.5.4.1. Market Estimation, 2015 - 2020
14.2.1.5.4.2. Market Forecast, 2021 - 2029
14.2.1.5.5.
Latin
America
14.2.1.5.5.1. Market Estimation, 2015 - 2020
14.2.1.5.5.2. Market Forecast, 2021 - 2029
14.2.2. Government Vaccination Campaigns
14.2.2.1. Definition
14.2.2.2. Market Estimation and Penetration, 2015 -
2020
14.2.2.3. Market Forecast, 2021 - 2029
14.2.2.4. Compound Annual Growth Rate (CAGR)
14.2.2.5. Regional Bifurcation
14.2.2.5.1.
North
America
14.2.2.5.1.1. Market Estimation, 2015 - 2020
14.2.2.5.1.2. Market Forecast, 2021 - 2029
14.2.2.5.2.
Europe
14.2.2.5.2.1. Market Estimation, 2015 - 2020
14.2.2.5.2.2. Market Forecast, 2021 - 2029
14.2.2.5.3.
Asia
Pacific
14.2.2.5.3.1. Market Estimation, 2015 - 2020
14.2.2.5.3.2. Market Forecast, 2021 - 2029
14.2.2.5.4.
Middle
East and Africa
14.2.2.5.4.1. Market Estimation, 2015 - 2020
14.2.2.5.4.2. Market Forecast, 2021 - 2029
14.2.2.5.5.
Latin
America
14.2.2.5.5.1. Market Estimation, 2015 - 2020
14.2.2.5.5.2. Market Forecast, 2021 - 2029
14.2.3. Research Facilities
14.2.3.1. Definition
14.2.3.2. Market Estimation and Penetration, 2015 -
2020
14.2.3.3. Market Forecast, 2021 - 2029
14.2.3.4. Compound Annual Growth Rate (CAGR)
14.2.3.5. Regional Bifurcation
14.2.3.5.1.
North
America
14.2.3.5.1.1. Market Estimation, 2015 - 2020
14.2.3.5.1.2. Market Forecast, 2021 - 2029
14.2.3.5.2.
Europe
14.2.3.5.2.1. Market Estimation, 2015 - 2020
14.2.3.5.2.2. Market Forecast, 2021 - 2029
14.2.3.5.3.
Asia
Pacific
14.2.3.5.3.1. Market Estimation, 2015 - 2020
14.2.3.5.3.2. Market Forecast, 2021 - 2029
14.2.3.5.4.
Middle
East and Africa
14.2.3.5.4.1. Market Estimation, 2015 - 2020
14.2.3.5.4.2. Market Forecast, 2021 - 2029
14.2.3.5.5.
Latin
America
14.2.3.5.5.1. Market Estimation, 2015 - 2020
14.2.3.5.5.2. Market Forecast, 2021 - 2029
14.2.4. Others
14.2.4.1. Definition
14.2.4.2. Market Estimation and Penetration, 2015 -
2020
14.2.4.3. Market Forecast, 2021 - 2029
14.2.4.4. Compound Annual Growth Rate (CAGR)
14.2.4.5. Regional Bifurcation
14.2.4.5.1.
North
America
14.2.4.5.1.1. Market Estimation, 2015 - 2020
14.2.4.5.1.2. Market Forecast, 2021 - 2029
14.2.4.5.2.
Europe
14.2.4.5.2.1. Market Estimation, 2015 - 2020
14.2.4.5.2.2. Market Forecast, 2021 - 2029
14.2.4.5.3.
Asia
Pacific
14.2.4.5.3.1. Market Estimation, 2015 - 2020
14.2.4.5.3.2. Market Forecast, 2021 - 2029
14.2.4.5.4.
Middle
East and Africa
14.2.4.5.4.1. Market Estimation, 2015 - 2020
14.2.4.5.4.2. Market Forecast, 2021 - 2029
14.2.4.5.5.
Latin
America
14.2.4.5.5.1. Market Estimation, 2015 - 2020
14.2.4.5.5.2. Market Forecast, 2021 - 2029
14.3. Key Segment for Channeling Investments
14.3.1. By End-User
15. North America mRNA Vaccines Market Analysis
and Forecasts, 2021 - 2029
15.1. Overview
15.1.1. North America mRNA Vaccines Market Revenue
(US$ Mn)
15.2. North America mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Clinical Trial Phase
15.2.1. Preclinical
15.2.2. Phase
I
15.2.3. Phase II
15.2.4. Phase III
15.2.5. Commercial
15.3. North America mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Type
15.3.1. Non-replicating mRNA
15.3.2. In vivo self-replicating mRNA
15.3.3. In vitro dendritic cell non-replicating mRNA
vaccine
15.4. North America mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Vaccine Modality
15.4.1. Therapeutic
15.4.2. TME Modulation
15.4.3. Prophylactic
15.4.4. DC Vaccine
15.4.5. T Cell regulation
15.5. North America mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Route of Administration
15.5.1. Intravenous/Intradermal
15.5.2. Intratumoral
15.5.3. Intramuscular
15.6. North America mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Indication
15.6.1. Oncology
15.6.1.1. Solid Tumors
15.6.1.2. Melanoma
15.6.1.3. Prostate cancer
15.6.1.4. Ovarian cancer
15.6.1.5. Glioblastoma
15.6.1.6. HPV + Cancer
15.6.1.7. Renal Cancer
15.6.1.8. Others
15.6.2. Infectious Diseases
15.6.2.1. COVID-19 vaccine
15.6.2.2. Rabies
15.6.2.3. Lassa, yellow fever
15.6.2.4. Respirational syncytial virus
15.6.2.5. Rota
15.6.2.6. Malaria
15.6.2.7. Universal influenza
15.6.2.8. Zika virus
15.6.2.9. Multiple sclerosis (MS)
15.6.2.10. Epstein-Barr virus (EBV)
15.6.2.11. HIV vaccine
15.6.2.12. Others
15.6.3. Rare Diseases
15.7. North America mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Applications
15.7.1. Adolescents and Pediatric
15.7.2. Adults
15.8. North America mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By End-User
15.8.1. Healthcare Facilities
15.8.2. Government Vaccination Campaigns
15.8.3. Research Facilities
15.8.4. Others
15.9. North America mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Country
15.9.1. U.S
15.9.1.1. U.S mRNA Vaccines Market Revenue (US$ Mn) and
Forecasts, By Clinical Trial Phase
15.9.1.1.1.
Preclinical
15.9.1.1.2.
Phase I
15.9.1.1.3.
Phase II
15.9.1.1.4.
Phase
III
15.9.1.1.5.
Commercial
15.9.1.2. U.S mRNA Vaccines Market Revenue (US$ Mn) and
Forecasts, By Type
15.9.1.2.1.
Non-replicating
mRNA
15.9.1.2.2.
In vivo
self-replicating mRNA
15.9.1.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
15.9.1.3. U.S mRNA Vaccines Market Revenue (US$ Mn) and
Forecasts, By Vaccine Modality
15.9.1.3.1.
Therapeutic
15.9.1.3.2.
TME
Modulation
15.9.1.3.3.
Prophylactic
15.9.1.3.4.
DC
Vaccine
15.9.1.3.5.
T Cell
regulation
15.9.1.4. U.S mRNA Vaccines Market Revenue (US$ Mn) and
Forecasts, By Route of Administration
15.9.1.4.1.
Intravenous/Intradermal
15.9.1.4.2.
Intratumoral
15.9.1.4.3.
Intramuscular
15.9.1.5. U.S mRNA Vaccines Market Revenue (US$ Mn) and
Forecasts, By Indication
15.9.1.5.1.
Oncology
15.9.1.5.1.1. Solid Tumors
15.9.1.5.1.2. Melanoma
15.9.1.5.1.3. Prostate cancer
15.9.1.5.1.4. Ovarian cancer
15.9.1.5.1.5. Glioblastoma
15.9.1.5.1.6. HPV + Cancer
15.9.1.5.1.7. Renal Cancer
15.9.1.5.1.8. Others
15.9.1.5.2.
Infectious
Diseases
15.9.1.5.2.1. COVID-19 vaccine
15.9.1.5.2.2. Rabies
15.9.1.5.2.3. Lassa, yellow fever
15.9.1.5.2.4. Respirational syncytial virus
15.9.1.5.2.5. Rota
15.9.1.5.2.6. Malaria
15.9.1.5.2.7. Universal influenza
15.9.1.5.2.8. Zika virus
15.9.1.5.2.9. Multiple sclerosis (MS)
15.9.1.5.2.10. Epstein-Barr virus (EBV)
15.9.1.5.2.11. HIV vaccine
15.9.1.5.2.12. Others
15.9.1.5.3.
Rare
Diseases
15.9.1.6. U.S mRNA Vaccines Market Revenue (US$ Mn) and
Forecasts, By Applications
15.9.1.6.1.
Adolescents
and Pediatric
15.9.1.6.2.
Adults
15.9.1.7. U.S mRNA Vaccines Market Revenue (US$ Mn) and
Forecasts, By End-User
15.9.1.7.1.
Healthcare
Facilities
15.9.1.7.2.
Government
Vaccination Campaigns
15.9.1.7.3.
Research
Facilities
15.9.1.7.4.
Others
15.9.2. Canada
15.9.2.1. Canada mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Clinical Trial Phase
15.9.2.1.1.
Preclinical
15.9.2.1.2.
Phase I
15.9.2.1.3.
Phase II
15.9.2.1.4.
Phase
III
15.9.2.1.5.
Commercial
15.9.2.2. Canada mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Type
15.9.2.2.1.
Non-replicating
mRNA
15.9.2.2.2.
In vivo
self-replicating mRNA
15.9.2.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
15.9.2.3. Canada mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Vaccine Modality
15.9.2.3.1.
Therapeutic
15.9.2.3.2.
TME
Modulation
15.9.2.3.3.
Prophylactic
15.9.2.3.4.
DC
Vaccine
15.9.2.3.5.
T Cell
regulation
15.9.2.4. Canada mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Route of Administration
15.9.2.4.1.
Intravenous/Intradermal
15.9.2.4.2.
Intratumoral
15.9.2.4.3.
Intramuscular
15.9.2.5. Canada mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Indication
15.9.2.5.1.
Oncology
15.9.2.5.1.1. Solid Tumors
15.9.2.5.1.2. Melanoma
15.9.2.5.1.3. Prostate cancer
15.9.2.5.1.4. Ovarian cancer
15.9.2.5.1.5. Glioblastoma
15.9.2.5.1.6. HPV + Cancer
15.9.2.5.1.7. Renal Cancer
15.9.2.5.1.8. Others
15.9.2.5.2.
Infectious
Diseases
15.9.2.5.2.1. COVID-19 vaccine
15.9.2.5.2.2. Rabies
15.9.2.5.2.3. Lassa, yellow fever
15.9.2.5.2.4. Respirational syncytial virus
15.9.2.5.2.5. Rota
15.9.2.5.2.6. Malaria
15.9.2.5.2.7. Universal influenza
15.9.2.5.2.8. Zika virus
15.9.2.5.2.9. Multiple sclerosis (MS)
15.9.2.5.2.10. Epstein-Barr virus (EBV)
15.9.2.5.2.11. HIV vaccine
15.9.2.5.2.12. Others
15.9.2.5.3.
Rare
Diseases
15.9.2.6. Canada mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Applications
15.9.2.6.1.
Adolescents
and Pediatric
15.9.2.6.2.
Adults
15.9.2.7. Canada mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By End-User
15.9.2.7.1.
Healthcare
Facilities
15.9.2.7.2.
Government
Vaccination Campaigns
15.9.2.7.3.
Research
Facilities
15.9.2.7.4.
Others
15.9.3. Mexico
15.9.3.1. Mexico mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Clinical Trial Phase
15.9.3.1.1.
Preclinical
15.9.3.1.2.
Phase I
15.9.3.1.3.
Phase II
15.9.3.1.4.
Phase
III
15.9.3.1.5.
Commercial
15.9.3.2. Mexico mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Type
15.9.3.2.1.
Non-replicating
mRNA
15.9.3.2.2.
In vivo
self-replicating mRNA
15.9.3.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
15.9.3.3. Mexico mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Vaccine Modality
15.9.3.3.1.
Therapeutic
15.9.3.3.2.
TME
Modulation
15.9.3.3.3.
Prophylactic
15.9.3.3.4.
DC
Vaccine
15.9.3.3.5.
T Cell
regulation
15.9.3.4. Mexico mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Route of Administration
15.9.3.4.1.
Intravenous/Intradermal
15.9.3.4.2.
Intratumoral
15.9.3.4.3.
Intramuscular
15.9.3.5. Mexico mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Indication
15.9.3.5.1.
Oncology
15.9.3.5.1.1. Solid Tumors
15.9.3.5.1.2. Melanoma
15.9.3.5.1.3. Prostate cancer
15.9.3.5.1.4. Ovarian cancer
15.9.3.5.1.5. Glioblastoma
15.9.3.5.1.6. HPV + Cancer
15.9.3.5.1.7. Renal Cancer
15.9.3.5.1.8. Others
15.9.3.5.2.
Infectious
Diseases
15.9.3.5.2.1. COVID-19 vaccine
15.9.3.5.2.2. Rabies
15.9.3.5.2.3. Lassa, yellow fever
15.9.3.5.2.4. Respirational syncytial virus
15.9.3.5.2.5. Rota
15.9.3.5.2.6. Malaria
15.9.3.5.2.7. Universal influenza
15.9.3.5.2.8. Zika virus
15.9.3.5.2.9. Multiple sclerosis (MS)
15.9.3.5.2.10. Epstein-Barr virus (EBV)
15.9.3.5.2.11. HIV vaccine
15.9.3.5.2.12. Others
15.9.3.5.3.
Rare
Diseases
15.9.3.6. Mexico mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Applications
15.9.3.6.1.
Adolescents
and Pediatric
15.9.3.6.2.
Adults
15.9.3.7. Mexico mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By End-User
15.9.3.7.1.
Healthcare
Facilities
15.9.3.7.2.
Government
Vaccination Campaigns
15.9.3.7.3.
Research
Facilities
15.9.3.7.4.
Others
15.9.4. Rest of North America
15.9.4.1. Rest of North America mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Clinical Trial Phase
15.9.4.1.1.
Preclinical
15.9.4.1.2.
Phase I
15.9.4.1.3.
Phase II
15.9.4.1.4.
Phase
III
15.9.4.1.5.
Commercial
15.9.4.2. Rest of North America mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Type
15.9.4.2.1.
Non-replicating
mRNA
15.9.4.2.2.
In vivo
self-replicating mRNA
15.9.4.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
15.9.4.3. Rest of North America mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Vaccine Modality
15.9.4.3.1.
Therapeutic
15.9.4.3.2.
TME
Modulation
15.9.4.3.3.
Prophylactic
15.9.4.3.4.
DC
Vaccine
15.9.4.3.5.
T Cell
regulation
15.9.4.4. Rest of North America mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Route of Administration
15.9.4.4.1.
Intravenous/Intradermal
15.9.4.4.2.
Intratumoral
15.9.4.4.3.
Intramuscular
15.9.4.5. Rest of North America mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Indication
15.9.4.5.1.
Oncology
15.9.4.5.1.1. Solid Tumors
15.9.4.5.1.2. Melanoma
15.9.4.5.1.3. Prostate cancer
15.9.4.5.1.4. Ovarian cancer
15.9.4.5.1.5. Glioblastoma
15.9.4.5.1.6. HPV + Cancer
15.9.4.5.1.7. Renal Cancer
15.9.4.5.1.8. Others
15.9.4.5.2.
Infectious
Diseases
15.9.4.5.2.1. COVID-19 vaccine
15.9.4.5.2.2. Rabies
15.9.4.5.2.3. Lassa, yellow fever
15.9.4.5.2.4. Respirational syncytial virus
15.9.4.5.2.5. Rota
15.9.4.5.2.6. Malaria
15.9.4.5.2.7. Universal influenza
15.9.4.5.2.8. Zika virus
15.9.4.5.2.9. Multiple sclerosis (MS)
15.9.4.5.2.10. Epstein-Barr virus (EBV)
15.9.4.5.2.11. HIV vaccine
15.9.4.5.2.12. Others
15.9.4.5.3.
Rare
Diseases
15.9.4.6. Rest of North America mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Applications
15.9.4.6.1.
Adolescents
and Pediatric
15.9.4.6.2.
Adults
15.9.4.7. Rest of North America mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By End-User
15.9.4.7.1.
Healthcare
Facilities
15.9.4.7.2.
Government
Vaccination Campaigns
15.9.4.7.3.
Research
Facilities
15.9.4.7.4.
Others
15.10. Key Segment for Channeling Investments
15.10.1. By Country
15.10.2. By Clinical Trial Phase
15.10.3. By Type
15.10.4. By Vaccine Modality
15.10.5. By Route of Administration
15.10.6. By Indication
15.10.7. By Applications
15.10.8. By End-User
16. Europe mRNA Vaccines Market Analysis and
Forecasts, 2021 - 2029
16.1. Overview
16.1.1. Europe mRNA Vaccines Market Revenue (US$ Mn)
16.2. Europe mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Clinical Trial Phase
16.2.1. Preclinical
16.2.2. Phase
I
16.2.3. Phase II
16.2.4. Phase III
16.2.5. Commercial
16.3. Europe mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Type
16.3.1. Non-replicating mRNA
16.3.2. In vivo self-replicating mRNA
16.3.3. In vitro dendritic cell non-replicating mRNA
vaccine
16.4. Europe mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Vaccine Modality
16.4.1. Therapeutic
16.4.2. TME Modulation
16.4.3. Prophylactic
16.4.4. DC Vaccine
16.4.5. T Cell regulation
16.5. Europe mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Route of Administration
16.5.1. Intravenous/Intradermal
16.5.2. Intratumoral
16.5.3. Intramuscular
16.6. Europe mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Indication
16.6.1. Oncology
16.6.1.1. Solid Tumors
16.6.1.2. Melanoma
16.6.1.3. Prostate cancer
16.6.1.4. Ovarian cancer
16.6.1.5. Glioblastoma
16.6.1.6. HPV + Cancer
16.6.1.7. Renal Cancer
16.6.1.8. Others
16.6.2. Infectious Diseases
16.6.2.1. COVID-19 vaccine
16.6.2.2. Rabies
16.6.2.3. Lassa, yellow fever
16.6.2.4. Respirational syncytial virus
16.6.2.5. Rota
16.6.2.6. Malaria
16.6.2.7. Universal influenza
16.6.2.8. Zika virus
16.6.2.9. Multiple sclerosis (MS)
16.6.2.10. Epstein-Barr virus (EBV)
16.6.2.11. HIV vaccine
16.6.2.12. Others
16.6.3. Rare Diseases
16.7. Europe mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Applications
16.7.1. Adolescents and Pediatric
16.7.2. Adults
16.8. Europe mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By End-User
16.8.1. Healthcare Facilities
16.8.2. Government Vaccination Campaigns
16.8.3. Research Facilities
16.8.4. Others
16.9. Europe mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Country
16.9.1. France
16.9.1.1. France mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Clinical Trial Phase
16.9.1.1.1.
Preclinical
16.9.1.1.2.
Phase I
16.9.1.1.3.
Phase II
16.9.1.1.4.
Phase
III
16.9.1.1.5.
Commercial
16.9.1.2. France mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Type
16.9.1.2.1.
Non-replicating
mRNA
16.9.1.2.2.
In vivo
self-replicating mRNA
16.9.1.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
16.9.1.3. France mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Vaccine Modality
16.9.1.3.1.
Therapeutic
16.9.1.3.2.
TME
Modulation
16.9.1.3.3.
Prophylactic
16.9.1.3.4.
DC
Vaccine
16.9.1.3.5.
T Cell regulation
16.9.1.4. France mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Route of Administration
16.9.1.4.1.
Intravenous/Intradermal
16.9.1.4.2.
Intratumoral
16.9.1.4.3.
Intramuscular
16.9.1.5. France mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Indication
16.9.1.5.1.
Oncology
16.9.1.5.1.1. Solid Tumors
16.9.1.5.1.2. Melanoma
16.9.1.5.1.3. Prostate cancer
16.9.1.5.1.4. Ovarian cancer
16.9.1.5.1.5. Glioblastoma
16.9.1.5.1.6. HPV + Cancer
16.9.1.5.1.7. Renal Cancer
16.9.1.5.1.8. Others
16.9.1.5.2.
Infectious
Diseases
16.9.1.5.2.1. COVID-19 vaccine
16.9.1.5.2.2. Rabies
16.9.1.5.2.3. Lassa, yellow fever
16.9.1.5.2.4. Respirational syncytial virus
16.9.1.5.2.5. Rota
16.9.1.5.2.6. Malaria
16.9.1.5.2.7. Universal influenza
16.9.1.5.2.8. Zika virus
16.9.1.5.2.9. Multiple sclerosis (MS)
16.9.1.5.2.10. Epstein-Barr virus (EBV)
16.9.1.5.2.11. HIV vaccine
16.9.1.5.2.12. Others
16.9.1.5.3.
Rare
Diseases
16.9.1.6. France mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Applications
16.9.1.6.1.
Adolescents
and Pediatric
16.9.1.6.2.
Adults
16.9.1.7. France mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By End-User
16.9.1.7.1.
Healthcare
Facilities
16.9.1.7.2.
Government
Vaccination Campaigns
16.9.1.7.3.
Research
Facilities
16.9.1.7.4.
Others
16.9.2. The UK
16.9.2.1. The UK mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Clinical Trial Phase
16.9.2.1.1.
Preclinical
16.9.2.1.2.
Phase I
16.9.2.1.3.
Phase II
16.9.2.1.4.
Phase
III
16.9.2.1.5.
Commercial
16.9.2.2. The UK mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Type
16.9.2.2.1.
Non-replicating
mRNA
16.9.2.2.2.
In vivo
self-replicating mRNA
16.9.2.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
16.9.2.3. The UK mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Vaccine Modality
16.9.2.3.1.
Therapeutic
16.9.2.3.2.
TME
Modulation
16.9.2.3.3.
Prophylactic
16.9.2.3.4.
DC
Vaccine
16.9.2.3.5.
T Cell regulation
16.9.2.4. The UK mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Route of Administration
16.9.2.4.1.
Intravenous/Intradermal
16.9.2.4.2.
Intratumoral
16.9.2.4.3.
Intramuscular
16.9.2.5. The UK mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Indication
16.9.2.5.1.
Oncology
16.9.2.5.1.1. Solid Tumors
16.9.2.5.1.2. Melanoma
16.9.2.5.1.3. Prostate cancer
16.9.2.5.1.4. Ovarian cancer
16.9.2.5.1.5. Glioblastoma
16.9.2.5.1.6. HPV + Cancer
16.9.2.5.1.7. Renal Cancer
16.9.2.5.1.8. Others
16.9.2.5.2.
Infectious
Diseases
16.9.2.5.2.1. COVID-19 vaccine
16.9.2.5.2.2. Rabies
16.9.2.5.2.3. Lassa, yellow fever
16.9.2.5.2.4. Respirational syncytial virus
16.9.2.5.2.5. Rota
16.9.2.5.2.6. Malaria
16.9.2.5.2.7. Universal influenza
16.9.2.5.2.8. Zika virus
16.9.2.5.2.9. Multiple sclerosis (MS)
16.9.2.5.2.10. Epstein-Barr virus (EBV)
16.9.2.5.2.11. HIV vaccine
16.9.2.5.2.12. Others
16.9.2.5.3.
Rare
Diseases
16.9.2.6. The UK mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Applications
16.9.2.6.1.
Adolescents
and Pediatric
16.9.2.6.2.
Adults
16.9.2.7. The UK mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By End-User
16.9.2.7.1.
Healthcare
Facilities
16.9.2.7.2.
Government
Vaccination Campaigns
16.9.2.7.3.
Research
Facilities
16.9.2.7.4.
Others
16.9.3. Spain
16.9.3.1. Spain mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Clinical Trial Phase
16.9.3.1.1.
Preclinical
16.9.3.1.2.
Phase I
16.9.3.1.3.
Phase II
16.9.3.1.4.
Phase
III
16.9.3.1.5.
Commercial
16.9.3.2. Spain mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Type
16.9.3.2.1.
Non-replicating
mRNA
16.9.3.2.2.
In vivo
self-replicating mRNA
16.9.3.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
16.9.3.3. Spain mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Vaccine Modality
16.9.3.3.1.
Therapeutic
16.9.3.3.2.
TME
Modulation
16.9.3.3.3.
Prophylactic
16.9.3.3.4.
DC
Vaccine
16.9.3.3.5.
T Cell
regulation
16.9.3.4. Spain mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Route of Administration
16.9.3.4.1.
Intravenous/Intradermal
16.9.3.4.2.
Intratumoral
16.9.3.4.3.
Intramuscular
16.9.3.5. Spain mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Indication
16.9.3.5.1.
Oncology
16.9.3.5.1.1. Solid Tumors
16.9.3.5.1.2. Melanoma
16.9.3.5.1.3. Prostate cancer
16.9.3.5.1.4. Ovarian cancer
16.9.3.5.1.5. Glioblastoma
16.9.3.5.1.6. HPV + Cancer
16.9.3.5.1.7. Renal Cancer
16.9.3.5.1.8. Others
16.9.3.5.2.
Infectious
Diseases
16.9.3.5.2.1. COVID-19 vaccine
16.9.3.5.2.2. Rabies
16.9.3.5.2.3. Lassa, yellow fever
16.9.3.5.2.4. Respirational syncytial virus
16.9.3.5.2.5. Rota
16.9.3.5.2.6. Malaria
16.9.3.5.2.7. Universal influenza
16.9.3.5.2.8. Zika virus
16.9.3.5.2.9. Multiple sclerosis (MS)
16.9.3.5.2.10. Epstein-Barr virus (EBV)
16.9.3.5.2.11. HIV vaccine
16.9.3.5.2.12. Others
16.9.3.5.3.
Rare
Diseases
16.9.3.6. Spain mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Applications
16.9.3.6.1.
Adolescents
and Pediatric
16.9.3.6.2.
Adults
16.9.3.7. Spain mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By End-User
16.9.3.7.1.
Healthcare
Facilities
16.9.3.7.2.
Government
Vaccination Campaigns
16.9.3.7.3.
Research
Facilities
16.9.3.7.4.
Others
16.9.4. Germany
16.9.4.1. Germany mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Clinical Trial Phase
16.9.4.1.1.
Preclinical
16.9.4.1.2.
Phase I
16.9.4.1.3.
Phase II
16.9.4.1.4.
Phase
III
16.9.4.1.5.
Commercial
16.9.4.2. Germany mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Type
16.9.4.2.1.
Non-replicating
mRNA
16.9.4.2.2.
In vivo
self-replicating mRNA
16.9.4.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
16.9.4.3. Germany mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Vaccine Modality
16.9.4.3.1.
Therapeutic
16.9.4.3.2.
TME
Modulation
16.9.4.3.3.
Prophylactic
16.9.4.3.4.
DC
Vaccine
16.9.4.3.5.
T Cell
regulation
16.9.4.4. Germany mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Route of Administration
16.9.4.4.1.
Intravenous/Intradermal
16.9.4.4.2.
Intratumoral
16.9.4.4.3.
Intramuscular
16.9.4.5. Germany mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Indication
16.9.4.5.1.
Oncology
16.9.4.5.1.1. Solid Tumors
16.9.4.5.1.2. Melanoma
16.9.4.5.1.3. Prostate cancer
16.9.4.5.1.4. Ovarian cancer
16.9.4.5.1.5. Glioblastoma
16.9.4.5.1.6. HPV + Cancer
16.9.4.5.1.7. Renal Cancer
16.9.4.5.1.8. Others
16.9.4.5.2.
Infectious
Diseases
16.9.4.5.2.1. COVID-19 vaccine
16.9.4.5.2.2. Rabies
16.9.4.5.2.3. Lassa, yellow fever
16.9.4.5.2.4. Respirational syncytial virus
16.9.4.5.2.5. Rota
16.9.4.5.2.6. Malaria
16.9.4.5.2.7. Universal influenza
16.9.4.5.2.8. Zika virus
16.9.4.5.2.9. Multiple sclerosis (MS)
16.9.4.5.2.10. Epstein-Barr virus (EBV)
16.9.4.5.2.11. HIV vaccine
16.9.4.5.2.12. Others
16.9.4.5.3.
Rare
Diseases
16.9.4.6. Germany mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Applications
16.9.4.6.1.
Adolescents
and Pediatric
16.9.4.6.2.
Adults
16.9.4.7. Germany mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By End-User
16.9.4.7.1.
Healthcare
Facilities
16.9.4.7.2.
Government
Vaccination Campaigns
16.9.4.7.3.
Research
Facilities
16.9.4.7.4.
Others
16.9.5. Italy
16.9.5.1. Italy mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Clinical Trial Phase
16.9.5.1.1.
Preclinical
16.9.5.1.2.
Phase I
16.9.5.1.3.
Phase II
16.9.5.1.4.
Phase
III
16.9.5.1.5.
Commercial
16.9.5.2. Italy mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Type
16.9.5.2.1.
Non-replicating
mRNA
16.9.5.2.2.
In vivo
self-replicating mRNA
16.9.5.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
16.9.5.3. Italy mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Vaccine Modality
16.9.5.3.1.
Therapeutic
16.9.5.3.2.
TME
Modulation
16.9.5.3.3.
Prophylactic
16.9.5.3.4.
DC
Vaccine
16.9.5.3.5.
T Cell
regulation
16.9.5.4. Italy mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Route of Administration
16.9.5.4.1.
Intravenous/Intradermal
16.9.5.4.2.
Intratumoral
16.9.5.4.3.
Intramuscular
16.9.5.5. Italy mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Indication
16.9.5.5.1.
Oncology
16.9.5.5.1.1. Solid Tumors
16.9.5.5.1.2. Melanoma
16.9.5.5.1.3. Prostate cancer
16.9.5.5.1.4. Ovarian cancer
16.9.5.5.1.5. Glioblastoma
16.9.5.5.1.6. HPV + Cancer
16.9.5.5.1.7. Renal Cancer
16.9.5.5.1.8. Others
16.9.5.5.2.
Infectious
Diseases
16.9.5.5.2.1. COVID-19 vaccine
16.9.5.5.2.2. Rabies
16.9.5.5.2.3. Lassa, yellow fever
16.9.5.5.2.4. Respirational syncytial virus
16.9.5.5.2.5. Rota
16.9.5.5.2.6. Malaria
16.9.5.5.2.7. Universal influenza
16.9.5.5.2.8. Zika virus
16.9.5.5.2.9. Multiple sclerosis (MS)
16.9.5.5.2.10. Epstein-Barr virus (EBV)
16.9.5.5.2.11. HIV vaccine
16.9.5.5.2.12. Others
16.9.5.5.3.
Rare
Diseases
16.9.5.6. Italy mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Applications
16.9.5.6.1.
Adolescents
and Pediatric
16.9.5.6.2.
Adults
16.9.5.7. Italy mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By End-User
16.9.5.7.1.
Healthcare
Facilities
16.9.5.7.2.
Government
Vaccination Campaigns
16.9.5.7.3.
Research
Facilities
16.9.5.7.4.
Others
16.9.6. Nordic Countries
16.9.6.1. Nordic Countries mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Clinical Trial Phase
16.9.6.1.1.
Preclinical
16.9.6.1.2.
Phase I
16.9.6.1.3.
Phase II
16.9.6.1.4.
Phase
III
16.9.6.1.5.
Commercial
16.9.6.2. Nordic Countries mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Type
16.9.6.2.1.
Non-replicating
mRNA
16.9.6.2.2.
In vivo
self-replicating mRNA
16.9.6.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
16.9.6.3. Nordic Countries mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Vaccine Modality
16.9.6.3.1.
Therapeutic
16.9.6.3.2.
TME
Modulation
16.9.6.3.3.
Prophylactic
16.9.6.3.4.
DC
Vaccine
16.9.6.3.5.
T Cell
regulation
16.9.6.4. Nordic Countries mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Route of Administration
16.9.6.4.1.
Intravenous/Intradermal
16.9.6.4.2.
Intratumoral
16.9.6.4.3.
Intramuscular
16.9.6.5. Nordic Countries mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Indication
16.9.6.5.1.
Oncology
16.9.6.5.1.1. Solid Tumors
16.9.6.5.1.2. Melanoma
16.9.6.5.1.3. Prostate cancer
16.9.6.5.1.4. Ovarian cancer
16.9.6.5.1.5. Glioblastoma
16.9.6.5.1.6. HPV + Cancer
16.9.6.5.1.7. Renal Cancer
16.9.6.5.1.8. Others
16.9.6.5.2.
Infectious
Diseases
16.9.6.5.2.1. COVID-19 vaccine
16.9.6.5.2.2. Rabies
16.9.6.5.2.3. Lassa, yellow fever
16.9.6.5.2.4. Respirational syncytial virus
16.9.6.5.2.5. Rota
16.9.6.5.2.6. Malaria
16.9.6.5.2.7. Universal influenza
16.9.6.5.2.8. Zika virus
16.9.6.5.2.9. Multiple sclerosis (MS)
16.9.6.5.2.10. Epstein-Barr virus (EBV)
16.9.6.5.2.11. HIV vaccine
16.9.6.5.2.12. Others
16.9.6.5.3.
Rare
Diseases
16.9.6.6. Nordic Countries mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Applications
16.9.6.6.1.
Adolescents
and Pediatric
16.9.6.6.2.
Adults
16.9.6.7. Nordic Countries mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By End-User
16.9.6.7.1.
Healthcare
Facilities
16.9.6.7.2.
Government
Vaccination Campaigns
16.9.6.7.3.
Research
Facilities
16.9.6.7.4.
Others
16.9.6.8. Nordic Countries mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Country
16.9.6.8.1.
Denmark
16.9.6.8.2.
Finland
16.9.6.8.3.
Iceland
16.9.6.8.4.
Sweden
16.9.6.8.5.
Norway
16.9.7. Benelux Union
16.9.7.1. Benelux Union mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Clinical Trial Phase
16.9.7.1.1.
Preclinical
16.9.7.1.2.
Phase I
16.9.7.1.3.
Phase II
16.9.7.1.4.
Phase
III
16.9.7.1.5.
Commercial
16.9.7.2. Benelux Union mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Type
16.9.7.2.1.
Non-replicating
mRNA
16.9.7.2.2.
In vivo
self-replicating mRNA
16.9.7.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
16.9.7.3. Benelux Union mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Vaccine Modality
16.9.7.3.1.
Therapeutic
16.9.7.3.2.
TME
Modulation
16.9.7.3.3.
Prophylactic
16.9.7.3.4.
DC
Vaccine
16.9.7.3.5.
T Cell
regulation
16.9.7.4. Benelux Union mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Route of Administration
16.9.7.4.1.
Intravenous/Intradermal
16.9.7.4.2.
Intratumoral
16.9.7.4.3.
Intramuscular
16.9.7.5. Benelux Union mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Indication
16.9.7.5.1.
Oncology
16.9.7.5.1.1. Solid Tumors
16.9.7.5.1.2. Melanoma
16.9.7.5.1.3. Prostate cancer
16.9.7.5.1.4. Ovarian cancer
16.9.7.5.1.5. Glioblastoma
16.9.7.5.1.6. HPV + Cancer
16.9.7.5.1.7. Renal Cancer
16.9.7.5.1.8. Others
16.9.7.5.2.
Infectious
Diseases
16.9.7.5.2.1. COVID-19 vaccine
16.9.7.5.2.2. Rabies
16.9.7.5.2.3. Lassa, yellow fever
16.9.7.5.2.4. Respirational syncytial virus
16.9.7.5.2.5. Rota
16.9.7.5.2.6. Malaria
16.9.7.5.2.7. Universal influenza
16.9.7.5.2.8. Zika virus
16.9.7.5.2.9. Multiple sclerosis (MS)
16.9.7.5.2.10. Epstein-Barr virus (EBV)
16.9.7.5.2.11. HIV vaccine
16.9.7.5.2.12. Others
16.9.7.5.3.
Rare
Diseases
16.9.7.6. Benelux Union mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Applications
16.9.7.6.1.
Adolescents
and Pediatric
16.9.7.6.2.
Adults
16.9.7.7. Benelux Union mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By End-User
16.9.7.7.1.
Healthcare
Facilities
16.9.7.7.2.
Government
Vaccination Campaigns
16.9.7.7.3.
Research
Facilities
16.9.7.7.4.
Others
16.9.7.8. Benelux Union mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Country
16.9.7.8.1.
Belgium
16.9.7.8.2.
The
Netherlands
16.9.7.8.3.
Luxembourg
16.9.8. Rest of Europe
16.9.8.1. Rest of Europe mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Clinical Trial Phase
16.9.8.1.1.
Preclinical
16.9.8.1.2.
Phase I
16.9.8.1.3.
Phase II
16.9.8.1.4.
Phase
III
16.9.8.1.5.
Commercial
16.9.8.2. Rest of Europe mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Type
16.9.8.2.1.
Non-replicating
mRNA
16.9.8.2.2.
In vivo
self-replicating mRNA
16.9.8.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
16.9.8.3. Rest of Europe mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Vaccine Modality
16.9.8.3.1.
Therapeutic
16.9.8.3.2.
TME
Modulation
16.9.8.3.3.
Prophylactic
16.9.8.3.4.
DC
Vaccine
16.9.8.3.5.
T Cell
regulation
16.9.8.4. Rest of Europe mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Route of Administration
16.9.8.4.1.
Intravenous/Intradermal
16.9.8.4.2.
Intratumoral
16.9.8.4.3.
Intramuscular
16.9.8.5. Rest of Europe mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Indication
16.9.8.5.1.
Oncology
16.9.8.5.1.1. Solid Tumors
16.9.8.5.1.2. Melanoma
16.9.8.5.1.3. Prostate cancer
16.9.8.5.1.4. Ovarian cancer
16.9.8.5.1.5. Glioblastoma
16.9.8.5.1.6. HPV + Cancer
16.9.8.5.1.7. Renal Cancer
16.9.8.5.1.8. Others
16.9.8.5.2.
Infectious
Diseases
16.9.8.5.2.1. COVID-19 vaccine
16.9.8.5.2.2. Rabies
16.9.8.5.2.3. Lassa, yellow fever
16.9.8.5.2.4. Respirational syncytial virus
16.9.8.5.2.5. Rota
16.9.8.5.2.6. Malaria
16.9.8.5.2.7. Universal influenza
16.9.8.5.2.8. Zika virus
16.9.8.5.2.9. Multiple sclerosis (MS)
16.9.8.5.2.10. Epstein-Barr virus (EBV)
16.9.8.5.2.11. HIV vaccine
16.9.8.5.2.12. Others
16.9.8.5.3.
Rare
Diseases
16.9.8.6. Rest of Europe mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Applications
16.9.8.6.1.
Adolescents
and Pediatric
16.9.8.6.2.
Adults
16.9.8.7. Rest of Europe mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By End-User
16.9.8.7.1.
Healthcare
Facilities
16.9.8.7.2.
Government
Vaccination Campaigns
16.9.8.7.3.
Research
Facilities
16.9.8.7.4.
Others
16.10. Key Segment for Channeling Investments
16.10.1. By Country
16.10.2. By Clinical Trial Phase
16.10.3. By Type
16.10.4. By Vaccine Modality
16.10.5. By Route of Administration
16.10.6. By Indication
16.10.7. By Applications
16.10.8. By End-User
17. Asia Pacific mRNA Vaccines Market Analysis
and Forecasts, 2021 - 2029
17.1. Overview
17.1.1. Asia Pacific mRNA Vaccines Market Revenue
(US$ Mn)
17.2. Asia Pacific mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Clinical Trial Phase
17.2.1. Preclinical
17.2.2. Phase
I
17.2.3. Phase II
17.2.4. Phase III
17.2.5. Commercial
17.3. Asia Pacific mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Type
17.3.1. Non-replicating mRNA
17.3.2. In vivo self-replicating mRNA
17.3.3. In vitro dendritic cell non-replicating mRNA
vaccine
17.4. Asia Pacific mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Vaccine Modality
17.4.1. Therapeutic
17.4.2. TME Modulation
17.4.3. Prophylactic
17.4.4. DC Vaccine
17.4.5. T Cell regulation
17.5. Asia Pacific mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Route of Administration
17.5.1. Intravenous/Intradermal
17.5.2. Intratumoral
17.5.3. Intramuscular
17.6. Asia Pacific mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Indication
17.6.1. Oncology
17.6.1.1. Solid Tumors
17.6.1.2. Melanoma
17.6.1.3. Prostate cancer
17.6.1.4. Ovarian cancer
17.6.1.5. Glioblastoma
17.6.1.6. HPV + Cancer
17.6.1.7. Renal Cancer
17.6.1.8. Others
17.6.2. Infectious Diseases
17.6.2.1. COVID-19 vaccine
17.6.2.2. Rabies
17.6.2.3. Lassa, yellow fever
17.6.2.4. Respirational syncytial virus
17.6.2.5. Rota
17.6.2.6. Malaria
17.6.2.7. Universal influenza
17.6.2.8. Zika virus
17.6.2.9. Multiple sclerosis (MS)
17.6.2.10. Epstein-Barr virus (EBV)
17.6.2.11. HIV vaccine
17.6.2.12. Others
17.6.3. Rare Diseases
17.7. Asia Pacific mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Applications
17.7.1. Adolescents and Pediatric
17.7.2. Adults
17.8. Asia Pacific mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By End-User
17.8.1. Healthcare Facilities
17.8.2. Government Vaccination Campaigns
17.8.3. Research Facilities
17.8.4. Others
17.9. Asia Pacific mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Country
17.9.1. China
17.9.1.1. China mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Clinical Trial Phase
17.9.1.1.1.
Preclinical
17.9.1.1.2.
Phase I
17.9.1.1.3.
Phase II
17.9.1.1.4.
Phase
III
17.9.1.1.5.
Commercial
17.9.1.2. China mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Type
17.9.1.2.1.
Non-replicating
mRNA
17.9.1.2.2.
In vivo
self-replicating mRNA
17.9.1.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
17.9.1.3. China mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Vaccine Modality
17.9.1.3.1.
Therapeutic
17.9.1.3.2.
TME
Modulation
17.9.1.3.3.
Prophylactic
17.9.1.3.4.
DC
Vaccine
17.9.1.3.5.
T Cell
regulation
17.9.1.4. China mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Route of Administration
17.9.1.4.1.
Intravenous/Intradermal
17.9.1.4.2.
Intratumoral
17.9.1.4.3.
Intramuscular
17.9.1.5. China mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Indication
17.9.1.5.1.
Oncology
17.9.1.5.1.1. Solid Tumors
17.9.1.5.1.2. Melanoma
17.9.1.5.1.3. Prostate cancer
17.9.1.5.1.4. Ovarian cancer
17.9.1.5.1.5. Glioblastoma
17.9.1.5.1.6. HPV + Cancer
17.9.1.5.1.7. Renal Cancer
17.9.1.5.1.8. Others
17.9.1.5.2.
Infectious
Diseases
17.9.1.5.2.1. COVID-19 vaccine
17.9.1.5.2.2. Rabies
17.9.1.5.2.3. Lassa, yellow fever
17.9.1.5.2.4. Respirational syncytial virus
17.9.1.5.2.5. Rota
17.9.1.5.2.6. Malaria
17.9.1.5.2.7. Universal influenza
17.9.1.5.2.8. Zika virus
17.9.1.5.2.9. Multiple sclerosis (MS)
17.9.1.5.2.10. Epstein-Barr virus (EBV)
17.9.1.5.2.11. HIV vaccine
17.9.1.5.2.12. Others
17.9.1.5.3.
Rare
Diseases
17.9.1.6. China mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Applications
17.9.1.6.1.
Adolescents
and Pediatric
17.9.1.6.2.
Adults
17.9.1.7. China mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By End-User
17.9.1.7.1.
Healthcare
Facilities
17.9.1.7.2.
Government
Vaccination Campaigns
17.9.1.7.3.
Research
Facilities
17.9.1.7.4.
Others
17.9.2. Japan
17.9.2.1. Japan mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Clinical Trial Phase
17.9.2.1.1.
Preclinical
17.9.2.1.2.
Phase I
17.9.2.1.3.
Phase II
17.9.2.1.4.
Phase
III
17.9.2.1.5.
Commercial
17.9.2.2. Japan mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Type
17.9.2.2.1.
Non-replicating
mRNA
17.9.2.2.2.
In vivo
self-replicating mRNA
17.9.2.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
17.9.2.3. Japan mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Vaccine Modality
17.9.2.3.1.
Therapeutic
17.9.2.3.2.
TME
Modulation
17.9.2.3.3.
Prophylactic
17.9.2.3.4.
DC
Vaccine
17.9.2.3.5.
T Cell
regulation
17.9.2.4. Japan mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Route of Administration
17.9.2.4.1.
Intravenous/Intradermal
17.9.2.4.2.
Intratumoral
17.9.2.4.3.
Intramuscular
17.9.2.5. Japan mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Indication
17.9.2.5.1.
Oncology
17.9.2.5.1.1. Solid Tumors
17.9.2.5.1.2. Melanoma
17.9.2.5.1.3. Prostate cancer
17.9.2.5.1.4. Ovarian cancer
17.9.2.5.1.5. Glioblastoma
17.9.2.5.1.6. HPV + Cancer
17.9.2.5.1.7. Renal Cancer
17.9.2.5.1.8. Others
17.9.2.5.2.
Infectious
Diseases
17.9.2.5.2.1. COVID-19 vaccine
17.9.2.5.2.2. Rabies
17.9.2.5.2.3. Lassa, yellow fever
17.9.2.5.2.4. Respirational syncytial virus
17.9.2.5.2.5. Rota
17.9.2.5.2.6. Malaria
17.9.2.5.2.7. Universal influenza
17.9.2.5.2.8. Zika virus
17.9.2.5.2.9. Multiple sclerosis (MS)
17.9.2.5.2.10. Epstein-Barr virus (EBV)
17.9.2.5.2.11. HIV vaccine
17.9.2.5.2.12. Others
17.9.2.5.3.
Rare
Diseases
17.9.2.6. Japan mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Applications
17.9.2.6.1.
Adolescents
and Pediatric
17.9.2.6.2.
Adults
17.9.2.7. Japan mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By End-User
17.9.2.7.1.
Healthcare
Facilities
17.9.2.7.2.
Government
Vaccination Campaigns
17.9.2.7.3.
Research
Facilities
17.9.2.7.4.
Others
17.9.3. India
17.9.3.1. India mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Clinical Trial Phase
17.9.3.1.1.
Preclinical
17.9.3.1.2.
Phase I
17.9.3.1.3.
Phase II
17.9.3.1.4.
Phase
III
17.9.3.1.5.
Commercial
17.9.3.2. India mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Type
17.9.3.2.1.
Non-replicating
mRNA
17.9.3.2.2.
In vivo
self-replicating mRNA
17.9.3.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
17.9.3.3. India mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Vaccine Modality
17.9.3.3.1.
Therapeutic
17.9.3.3.2.
TME
Modulation
17.9.3.3.3.
Prophylactic
17.9.3.3.4.
DC
Vaccine
17.9.3.3.5.
T Cell
regulation
17.9.3.4. India mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Route of Administration
17.9.3.4.1.
Intravenous/Intradermal
17.9.3.4.2.
Intratumoral
17.9.3.4.3.
Intramuscular
17.9.3.5. India mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Indication
17.9.3.5.1.
Oncology
17.9.3.5.1.1. Solid Tumors
17.9.3.5.1.2. Melanoma
17.9.3.5.1.3. Prostate cancer
17.9.3.5.1.4. Ovarian cancer
17.9.3.5.1.5. Glioblastoma
17.9.3.5.1.6. HPV + Cancer
17.9.3.5.1.7. Renal Cancer
17.9.3.5.1.8. Others
17.9.3.5.2.
Infectious
Diseases
17.9.3.5.2.1. COVID-19 vaccine
17.9.3.5.2.2. Rabies
17.9.3.5.2.3. Lassa, yellow fever
17.9.3.5.2.4. Respirational syncytial virus
17.9.3.5.2.5. Rota
17.9.3.5.2.6. Malaria
17.9.3.5.2.7. Universal influenza
17.9.3.5.2.8. Zika virus
17.9.3.5.2.9. Multiple sclerosis (MS)
17.9.3.5.2.10. Epstein-Barr virus (EBV)
17.9.3.5.2.11. HIV vaccine
17.9.3.5.2.12. Others
17.9.3.5.3.
Rare
Diseases
17.9.3.6. India mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Applications
17.9.3.6.1.
Adolescents
and Pediatric
17.9.3.6.2.
Adults
17.9.3.7. India mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By End-User
17.9.3.7.1.
Healthcare
Facilities
17.9.3.7.2.
Government
Vaccination Campaigns
17.9.3.7.3.
Research
Facilities
17.9.3.7.4.
Others
17.9.4. New Zealand
17.9.4.1. New Zealand mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Clinical Trial Phase
17.9.4.1.1.
Preclinical
17.9.4.1.2.
Phase I
17.9.4.1.3.
Phase II
17.9.4.1.4.
Phase
III
17.9.4.1.5.
Commercial
17.9.4.2. New Zealand mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Type
17.9.4.2.1.
Non-replicating
mRNA
17.9.4.2.2.
In vivo
self-replicating mRNA
17.9.4.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
17.9.4.3. New Zealand mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Vaccine Modality
17.9.4.3.1.
Therapeutic
17.9.4.3.2.
TME
Modulation
17.9.4.3.3.
Prophylactic
17.9.4.3.4.
DC
Vaccine
17.9.4.3.5.
T Cell
regulation
17.9.4.4. New Zealand mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Route of Administration
17.9.4.4.1.
Intravenous/Intradermal
17.9.4.4.2.
Intratumoral
17.9.4.4.3.
Intramuscular
17.9.4.5. New Zealand mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Indication
17.9.4.5.1.
Oncology
17.9.4.5.1.1. Solid Tumors
17.9.4.5.1.2. Melanoma
17.9.4.5.1.3. Prostate cancer
17.9.4.5.1.4. Ovarian cancer
17.9.4.5.1.5. Glioblastoma
17.9.4.5.1.6. HPV + Cancer
17.9.4.5.1.7. Renal Cancer
17.9.4.5.1.8. Others
17.9.4.5.2.
Infectious
Diseases
17.9.4.5.2.1. COVID-19 vaccine
17.9.4.5.2.2. Rabies
17.9.4.5.2.3. Lassa, yellow fever
17.9.4.5.2.4. Respirational syncytial virus
17.9.4.5.2.5. Rota
17.9.4.5.2.6. Malaria
17.9.4.5.2.7. Universal influenza
17.9.4.5.2.8. Zika virus
17.9.4.5.2.9. Multiple sclerosis (MS)
17.9.4.5.2.10. Epstein-Barr virus (EBV)
17.9.4.5.2.11. HIV vaccine
17.9.4.5.2.12. Others
17.9.4.5.3.
Rare
Diseases
17.9.4.6. New Zealand mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Applications
17.9.4.6.1.
Adolescents
and Pediatric
17.9.4.6.2.
Adults
17.9.4.7. New Zealand mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By End-User
17.9.4.7.1.
Healthcare
Facilities
17.9.4.7.2.
Government
Vaccination Campaigns
17.9.4.7.3.
Research
Facilities
17.9.4.7.4.
Others
17.9.5. Australia
17.9.5.1. Australia mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Clinical Trial Phase
17.9.5.1.1.
Preclinical
17.9.5.1.2.
Phase I
17.9.5.1.3.
Phase II
17.9.5.1.4.
Phase
III
17.9.5.1.5.
Commercial
17.9.5.2. Australia mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Type
17.9.5.2.1.
Non-replicating
mRNA
17.9.5.2.2.
In vivo
self-replicating mRNA
17.9.5.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
17.9.5.3. Australia mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Vaccine Modality
17.9.5.3.1.
Therapeutic
17.9.5.3.2.
TME
Modulation
17.9.5.3.3.
Prophylactic
17.9.5.3.4.
DC
Vaccine
17.9.5.3.5.
T Cell
regulation
17.9.5.4. Australia mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Route of Administration
17.9.5.4.1.
Intravenous/Intradermal
17.9.5.4.2.
Intratumoral
17.9.5.4.3.
Intramuscular
17.9.5.5. Australia mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Indication
17.9.5.5.1.
Oncology
17.9.5.5.1.1. Solid Tumors
17.9.5.5.1.2. Melanoma
17.9.5.5.1.3. Prostate cancer
17.9.5.5.1.4. Ovarian cancer
17.9.5.5.1.5. Glioblastoma
17.9.5.5.1.6. HPV + Cancer
17.9.5.5.1.7. Renal Cancer
17.9.5.5.1.8. Others
17.9.5.5.2.
Infectious
Diseases
17.9.5.5.2.1. COVID-19 vaccine
17.9.5.5.2.2. Rabies
17.9.5.5.2.3. Lassa, yellow fever
17.9.5.5.2.4. Respirational syncytial virus
17.9.5.5.2.5. Rota
17.9.5.5.2.6. Malaria
17.9.5.5.2.7. Universal influenza
17.9.5.5.2.8. Zika virus
17.9.5.5.2.9. Multiple sclerosis (MS)
17.9.5.5.2.10. Epstein-Barr virus (EBV)
17.9.5.5.2.11. HIV vaccine
17.9.5.5.2.12. Others
17.9.5.5.3.
Rare
Diseases
17.9.5.6. Australia mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Applications
17.9.5.6.1.
Adolescents
and Pediatric
17.9.5.6.2.
Adults
17.9.5.7. Australia mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By End-User
17.9.5.7.1.
Healthcare
Facilities
17.9.5.7.2.
Government
Vaccination Campaigns
17.9.5.7.3.
Research
Facilities
17.9.5.7.4.
Others
17.9.6. South Korea
17.9.6.1. South Korea mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Clinical Trial Phase
17.9.6.1.1.
Preclinical
17.9.6.1.2.
Phase I
17.9.6.1.3.
Phase II
17.9.6.1.4.
Phase
III
17.9.6.1.5.
Commercial
17.9.6.2. South Korea mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Type
17.9.6.2.1.
Non-replicating
mRNA
17.9.6.2.2.
In vivo
self-replicating mRNA
17.9.6.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
17.9.6.3. South Korea mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Vaccine Modality
17.9.6.3.1.
Therapeutic
17.9.6.3.2.
TME
Modulation
17.9.6.3.3.
Prophylactic
17.9.6.3.4.
DC
Vaccine
17.9.6.3.5.
T Cell
regulation
17.9.6.4. South Korea mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Route of Administration
17.9.6.4.1.
Intravenous/Intradermal
17.9.6.4.2.
Intratumoral
17.9.6.4.3.
Intramuscular
17.9.6.5. South Korea mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Indication
17.9.6.5.1.
Oncology
17.9.6.5.1.1. Solid Tumors
17.9.6.5.1.2. Melanoma
17.9.6.5.1.3. Prostate cancer
17.9.6.5.1.4. Ovarian cancer
17.9.6.5.1.5. Glioblastoma
17.9.6.5.1.6. HPV + Cancer
17.9.6.5.1.7. Renal Cancer
17.9.6.5.1.8. Others
17.9.6.5.2.
Infectious
Diseases
17.9.6.5.2.1. COVID-19 vaccine
17.9.6.5.2.2. Rabies
17.9.6.5.2.3. Lassa, yellow fever
17.9.6.5.2.4. Respirational syncytial virus
17.9.6.5.2.5. Rota
17.9.6.5.2.6. Malaria
17.9.6.5.2.7. Universal influenza
17.9.6.5.2.8. Zika virus
17.9.6.5.2.9. Multiple sclerosis (MS)
17.9.6.5.2.10. Epstein-Barr virus (EBV)
17.9.6.5.2.11. HIV vaccine
17.9.6.5.2.12. Others
17.9.6.5.3.
Rare
Diseases
17.9.6.6. South Korea mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Applications
17.9.6.6.1.
Adolescents
and Pediatric
17.9.6.6.2.
Adults
17.9.6.7. South Korea mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By End-User
17.9.6.7.1.
Healthcare
Facilities
17.9.6.7.2.
Government
Vaccination Campaigns
17.9.6.7.3.
Research
Facilities
17.9.6.7.4.
Others
17.9.7. Southeast Asia
17.9.7.1. Southeast Asia mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Clinical Trial Phase
17.9.7.1.1.
Preclinical
17.9.7.1.2.
Phase I
17.9.7.1.3.
Phase II
17.9.7.1.4.
Phase
III
17.9.7.1.5.
Commercial
17.9.7.2. Southeast Asia mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Type
17.9.7.2.1.
Non-replicating
mRNA
17.9.7.2.2.
In vivo
self-replicating mRNA
17.9.7.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
17.9.7.3. Southeast Asia mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Vaccine Modality
17.9.7.3.1.
Therapeutic
17.9.7.3.2.
TME Modulation
17.9.7.3.3.
Prophylactic
17.9.7.3.4.
DC
Vaccine
17.9.7.3.5.
T Cell
regulation
17.9.7.4. Southeast Asia mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Route of Administration
17.9.7.4.1.
Intravenous/Intradermal
17.9.7.4.2.
Intratumoral
17.9.7.4.3.
Intramuscular
17.9.7.5. Southeast Asia mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Indication
17.9.7.5.1.
Oncology
17.9.7.5.1.1. Solid Tumors
17.9.7.5.1.2. Melanoma
17.9.7.5.1.3. Prostate cancer
17.9.7.5.1.4. Ovarian cancer
17.9.7.5.1.5. Glioblastoma
17.9.7.5.1.6. HPV + Cancer
17.9.7.5.1.7. Renal Cancer
17.9.7.5.1.8. Others
17.9.7.5.2.
Infectious
Diseases
17.9.7.5.2.1. COVID-19 vaccine
17.9.7.5.2.2. Rabies
17.9.7.5.2.3. Lassa, yellow fever
17.9.7.5.2.4. Respirational syncytial virus
17.9.7.5.2.5. Rota
17.9.7.5.2.6. Malaria
17.9.7.5.2.7. Universal influenza
17.9.7.5.2.8. Zika virus
17.9.7.5.2.9. Multiple sclerosis (MS)
17.9.7.5.2.10. Epstein-Barr virus (EBV)
17.9.7.5.2.11. HIV vaccine
17.9.7.5.2.12. Others
17.9.7.5.3.
Rare
Diseases
17.9.7.6. Southeast Asia mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Applications
17.9.7.6.1.
Adolescents
and Pediatric
17.9.7.6.2.
Adults
17.9.7.7. Southeast Asia mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By End-User
17.9.7.7.1.
Healthcare
Facilities
17.9.7.7.2.
Government
Vaccination Campaigns
17.9.7.7.3.
Research
Facilities
17.9.7.7.4.
Others
17.9.7.8. Southeast Asia mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Country
17.9.7.8.1.
Indonesia
17.9.7.8.2.
Thailand
17.9.7.8.3.
Malaysia
17.9.7.8.4.
Singapore
17.9.7.8.5.
Rest of Southeast
Asia
17.9.8. Rest of Asia Pacific
17.9.8.1. Rest of Asia Pacific mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Clinical Trial Phase
17.9.8.1.1.
Preclinical
17.9.8.1.2.
Phase I
17.9.8.1.3.
Phase II
17.9.8.1.4.
Phase
III
17.9.8.1.5.
Commercial
17.9.8.2. Rest of Asia Pacific mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Type
17.9.8.2.1.
Non-replicating
mRNA
17.9.8.2.2.
In vivo
self-replicating mRNA
17.9.8.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
17.9.8.3. Rest of Asia Pacific mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Vaccine Modality
17.9.8.3.1.
Therapeutic
17.9.8.3.2.
TME
Modulation
17.9.8.3.3.
Prophylactic
17.9.8.3.4.
DC
Vaccine
17.9.8.3.5.
T Cell
regulation
17.9.8.4. Rest of Asia Pacific mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Route of Administration
17.9.8.4.1.
Intravenous/Intradermal
17.9.8.4.2.
Intratumoral
17.9.8.4.3.
Intramuscular
17.9.8.5. Rest of Asia Pacific mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Indication
17.9.8.5.1.
Oncology
17.9.8.5.1.1. Solid Tumors
17.9.8.5.1.2. Melanoma
17.9.8.5.1.3. Prostate cancer
17.9.8.5.1.4. Ovarian cancer
17.9.8.5.1.5. Glioblastoma
17.9.8.5.1.6. HPV + Cancer
17.9.8.5.1.7. Renal Cancer
17.9.8.5.1.8. Others
17.9.8.5.2.
Infectious
Diseases
17.9.8.5.2.1. COVID-19 vaccine
17.9.8.5.2.2. Rabies
17.9.8.5.2.3. Lassa, yellow fever
17.9.8.5.2.4. Respirational syncytial virus
17.9.8.5.2.5. Rota
17.9.8.5.2.6. Malaria
17.9.8.5.2.7. Universal influenza
17.9.8.5.2.8. Zika virus
17.9.8.5.2.9. Multiple sclerosis (MS)
17.9.8.5.2.10. Epstein-Barr virus (EBV)
17.9.8.5.2.11. HIV vaccine
17.9.8.5.2.12. Others
17.9.8.5.3.
Rare
Diseases
17.9.8.6. Rest of Asia Pacific mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Applications
17.9.8.6.1.
Adolescents
and Pediatric
17.9.8.6.2.
Adults
17.9.8.7. Rest of Asia Pacific mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By End-User
17.9.8.7.1.
Healthcare
Facilities
17.9.8.7.2.
Government
Vaccination Campaigns
17.9.8.7.3.
Research
Facilities
17.9.8.7.4.
Others
17.10. Key Segment for Channeling Investments
17.10.1. By Country
17.10.2. By Clinical Trial Phase
17.10.3. By Type
17.10.4. By Vaccine Modality
17.10.5. By Route of Administration
17.10.6. By Indication
17.10.7. By Applications
17.10.8. By End-User
18. Middle East and Africa mRNA Vaccines Market
Analysis and Forecasts, 2021 - 2029
18.1. Overview
18.1.1. Middle East and Africa mRNA Vaccines Market
Revenue (US$ Mn)
18.2. Middle East and Africa mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Clinical Trial Phase
18.2.1. Preclinical
18.2.2. Phase
I
18.2.3. Phase II
18.2.4. Phase III
18.2.5. Commercial
18.3. Middle East and Africa mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Type
18.3.1. Non-replicating mRNA
18.3.2. In vivo self-replicating mRNA
18.3.3. In vitro dendritic cell non-replicating mRNA
vaccine
18.4. Middle East and Africa mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Vaccine Modality
18.4.1. Therapeutic
18.4.2. TME Modulation
18.4.3. Prophylactic
18.4.4. DC Vaccine
18.4.5. T Cell regulation
18.5. Middle East and Africa mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Route of Administration
18.5.1. Intravenous/Intradermal
18.5.2. Intratumoral
18.5.3. Intramuscular
18.6. Middle East and Africa mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Indication
18.6.1. Oncology
18.6.1.1. Solid Tumors
18.6.1.2. Melanoma
18.6.1.3. Prostate cancer
18.6.1.4. Ovarian cancer
18.6.1.5. Glioblastoma
18.6.1.6. HPV + Cancer
18.6.1.7. Renal Cancer
18.6.1.8. Others
18.6.2. Infectious Diseases
18.6.2.1. COVID-19 vaccine
18.6.2.2. Rabies
18.6.2.3. Lassa, yellow fever
18.6.2.4. Respirational syncytial virus
18.6.2.5. Rota
18.6.2.6. Malaria
18.6.2.7. Universal influenza
18.6.2.8. Zika virus
18.6.2.9. Multiple sclerosis (MS)
18.6.2.10. Epstein-Barr virus (EBV)
18.6.2.11. HIV vaccine
18.6.2.12. Others
18.6.3. Rare Diseases
18.7. Middle East and Africa mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Applications
18.7.1. Adolescents and Pediatric
18.7.2. Adults
18.8. Middle East and Africa mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By End-User
18.8.1. Healthcare Facilities
18.8.2. Government Vaccination Campaigns
18.8.3. Research Facilities
18.8.4. Others
18.9. Middle East and Africa mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Country
18.9.1. Saudi Arabia
18.9.1.1. Saudi Arabia mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Clinical Trial Phase
18.9.1.1.1.
Preclinical
18.9.1.1.2.
Phase I
18.9.1.1.3.
Phase II
18.9.1.1.4.
Phase
III
18.9.1.1.5.
Commercial
18.9.1.2. Saudi Arabia mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Type
18.9.1.2.1.
Non-replicating
mRNA
18.9.1.2.2.
In vivo
self-replicating mRNA
18.9.1.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
18.9.1.3. Saudi Arabia mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Vaccine Modality
18.9.1.3.1.
Therapeutic
18.9.1.3.2.
TME
Modulation
18.9.1.3.3.
Prophylactic
18.9.1.3.4.
DC
Vaccine
18.9.1.3.5.
T Cell
regulation
18.9.1.4. Saudi Arabia mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Route of Administration
18.9.1.4.1.
Intravenous/Intradermal
18.9.1.4.2.
Intratumoral
18.9.1.4.3.
Intramuscular
18.9.1.5. Saudi Arabia mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Indication
18.9.1.5.1.
Oncology
18.9.1.5.1.1. Solid Tumors
18.9.1.5.1.2. Melanoma
18.9.1.5.1.3. Prostate cancer
18.9.1.5.1.4. Ovarian cancer
18.9.1.5.1.5. Glioblastoma
18.9.1.5.1.6. HPV + Cancer
18.9.1.5.1.7. Renal Cancer
18.9.1.5.1.8. Others
18.9.1.5.2.
Infectious
Diseases
18.9.1.5.2.1. COVID-19 vaccine
18.9.1.5.2.2. Rabies
18.9.1.5.2.3. Lassa, yellow fever
18.9.1.5.2.4. Respirational syncytial virus
18.9.1.5.2.5. Rota
18.9.1.5.2.6. Malaria
18.9.1.5.2.7. Universal influenza
18.9.1.5.2.8. Zika virus
18.9.1.5.2.9. Multiple sclerosis (MS)
18.9.1.5.2.10. Epstein-Barr virus (EBV)
18.9.1.5.2.11. HIV vaccine
18.9.1.5.2.12. Others
18.9.1.5.3.
Rare
Diseases
18.9.1.6. Saudi Arabia mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Applications
18.9.1.6.1.
Adolescents
and Pediatric
18.9.1.6.2.
Adults
18.9.1.7. Saudi Arabia mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By End-User
18.9.1.7.1.
Healthcare
Facilities
18.9.1.7.2.
Government
Vaccination Campaigns
18.9.1.7.3.
Research
Facilities
18.9.1.7.4.
Others
18.9.2. UAE
18.9.2.1. UAE mRNA Vaccines Market Revenue (US$ Mn) and
Forecasts, By Clinical Trial Phase
18.9.2.1.1.
Preclinical
18.9.2.1.2.
Phase I
18.9.2.1.3.
Phase II
18.9.2.1.4.
Phase
III
18.9.2.1.5.
Commercial
18.9.2.2. UAE mRNA Vaccines Market Revenue (US$ Mn) and
Forecasts, By Type
18.9.2.2.1.
Non-replicating
mRNA
18.9.2.2.2.
In vivo
self-replicating mRNA
18.9.2.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
18.9.2.3. UAE mRNA Vaccines Market Revenue (US$ Mn) and
Forecasts, By Vaccine Modality
18.9.2.3.1.
Therapeutic
18.9.2.3.2.
TME
Modulation
18.9.2.3.3.
Prophylactic
18.9.2.3.4.
DC
Vaccine
18.9.2.3.5.
T Cell
regulation
18.9.2.4. UAE mRNA Vaccines Market Revenue (US$ Mn) and
Forecasts, By Route of Administration
18.9.2.4.1.
Intravenous/Intradermal
18.9.2.4.2.
Intratumoral
18.9.2.4.3.
Intramuscular
18.9.2.5. UAE mRNA Vaccines Market Revenue (US$ Mn) and
Forecasts, By Indication
18.9.2.5.1.
Oncology
18.9.2.5.1.1. Solid Tumors
18.9.2.5.1.2. Melanoma
18.9.2.5.1.3. Prostate cancer
18.9.2.5.1.4. Ovarian cancer
18.9.2.5.1.5. Glioblastoma
18.9.2.5.1.6. HPV + Cancer
18.9.2.5.1.7. Renal Cancer
18.9.2.5.1.8. Others
18.9.2.5.2.
Infectious
Diseases
18.9.2.5.2.1. COVID-19 vaccine
18.9.2.5.2.2. Rabies
18.9.2.5.2.3. Lassa, yellow fever
18.9.2.5.2.4. Respirational syncytial virus
18.9.2.5.2.5. Rota
18.9.2.5.2.6. Malaria
18.9.2.5.2.7. Universal influenza
18.9.2.5.2.8. Zika virus
18.9.2.5.2.9. Multiple sclerosis (MS)
18.9.2.5.2.10. Epstein-Barr virus (EBV)
18.9.2.5.2.11. HIV vaccine
18.9.2.5.2.12. Others
18.9.2.5.3.
Rare
Diseases
18.9.2.6. UAE mRNA Vaccines Market Revenue (US$ Mn) and
Forecasts, By Applications
18.9.2.6.1.
Adolescents
and Pediatric
18.9.2.6.2.
Adults
18.9.2.7. UAE mRNA Vaccines Market Revenue (US$ Mn) and
Forecasts, By End-User
18.9.2.7.1.
Healthcare
Facilities
18.9.2.7.2.
Government
Vaccination Campaigns
18.9.2.7.3.
Research
Facilities
18.9.2.7.4.
Others
18.9.3. Egypt
18.9.3.1. Egypt mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Clinical Trial Phase
18.9.3.1.1.
Preclinical
18.9.3.1.2.
Phase I
18.9.3.1.3.
Phase II
18.9.3.1.4.
Phase
III
18.9.3.1.5.
Commercial
18.9.3.2. Egypt mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Type
18.9.3.2.1.
Non-replicating
mRNA
18.9.3.2.2.
In vivo
self-replicating mRNA
18.9.3.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
18.9.3.3. Egypt mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Vaccine Modality
18.9.3.3.1.
Therapeutic
18.9.3.3.2.
TME
Modulation
18.9.3.3.3.
Prophylactic
18.9.3.3.4.
DC
Vaccine
18.9.3.3.5.
T Cell
regulation
18.9.3.4. Egypt mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Route of Administration
18.9.3.4.1.
Intravenous/Intradermal
18.9.3.4.2.
Intratumoral
18.9.3.4.3.
Intramuscular
18.9.3.5. Egypt mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Indication
18.9.3.5.1.
Oncology
18.9.3.5.1.1. Solid Tumors
18.9.3.5.1.2. Melanoma
18.9.3.5.1.3. Prostate cancer
18.9.3.5.1.4. Ovarian cancer
18.9.3.5.1.5. Glioblastoma
18.9.3.5.1.6. HPV + Cancer
18.9.3.5.1.7. Renal Cancer
18.9.3.5.1.8. Others
18.9.3.5.2.
Infectious
Diseases
18.9.3.5.2.1. COVID-19 vaccine
18.9.3.5.2.2. Rabies
18.9.3.5.2.3. Lassa, yellow fever
18.9.3.5.2.4. Respirational syncytial virus
18.9.3.5.2.5. Rota
18.9.3.5.2.6. Malaria
18.9.3.5.2.7. Universal influenza
18.9.3.5.2.8. Zika virus
18.9.3.5.2.9. Multiple sclerosis (MS)
18.9.3.5.2.10. Epstein-Barr virus (EBV)
18.9.3.5.2.11. HIV vaccine
18.9.3.5.2.12. Others
18.9.3.5.3.
Rare
Diseases
18.9.3.6. Egypt mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Applications
18.9.3.6.1.
Adolescents
and Pediatric
18.9.3.6.2.
Adults
18.9.3.7. Egypt mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By End-User
18.9.3.7.1.
Healthcare
Facilities
18.9.3.7.2.
Government
Vaccination Campaigns
18.9.3.7.3.
Research
Facilities
18.9.3.7.4.
Others
18.9.4. Kuwait
18.9.4.1. Kuwait mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Clinical Trial Phase
18.9.4.1.1.
Preclinical
18.9.4.1.2.
Phase I
18.9.4.1.3.
Phase II
18.9.4.1.4.
Phase
III
18.9.4.1.5.
Commercial
18.9.4.2. Kuwait mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Type
18.9.4.2.1.
Non-replicating
mRNA
18.9.4.2.2.
In vivo self-replicating
mRNA
18.9.4.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
18.9.4.3. Kuwait mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Vaccine Modality
18.9.4.3.1.
Therapeutic
18.9.4.3.2.
TME
Modulation
18.9.4.3.3.
Prophylactic
18.9.4.3.4.
DC
Vaccine
18.9.4.3.5.
T Cell
regulation
18.9.4.4. Kuwait mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Route of Administration
18.9.4.4.1.
Intravenous/Intradermal
18.9.4.4.2.
Intratumoral
18.9.4.4.3.
Intramuscular
18.9.4.5. Kuwait mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Indication
18.9.4.5.1.
Oncology
18.9.4.5.1.1. Solid Tumors
18.9.4.5.1.2. Melanoma
18.9.4.5.1.3. Prostate cancer
18.9.4.5.1.4. Ovarian cancer
18.9.4.5.1.5. Glioblastoma
18.9.4.5.1.6. HPV + Cancer
18.9.4.5.1.7. Renal Cancer
18.9.4.5.1.8. Others
18.9.4.5.2.
Infectious
Diseases
18.9.4.5.2.1. COVID-19 vaccine
18.9.4.5.2.2. Rabies
18.9.4.5.2.3. Lassa, yellow fever
18.9.4.5.2.4. Respirational syncytial virus
18.9.4.5.2.5. Rota
18.9.4.5.2.6. Malaria
18.9.4.5.2.7. Universal influenza
18.9.4.5.2.8. Zika virus
18.9.4.5.2.9. Multiple sclerosis (MS)
18.9.4.5.2.10. Epstein-Barr virus (EBV)
18.9.4.5.2.11. HIV vaccine
18.9.4.5.2.12. Others
18.9.4.5.3.
Rare
Diseases
18.9.4.6. Kuwait mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Applications
18.9.4.6.1.
Adolescents
and Pediatric
18.9.4.6.2.
Adults
18.9.4.7. Kuwait mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By End-User
18.9.4.7.1.
Healthcare
Facilities
18.9.4.7.2.
Government
Vaccination Campaigns
18.9.4.7.3.
Research
Facilities
18.9.4.7.4.
Others
18.9.5. South Africa
18.9.5.1. South Africa mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Clinical Trial Phase
18.9.5.1.1.
Preclinical
18.9.5.1.2.
Phase I
18.9.5.1.3.
Phase II
18.9.5.1.4.
Phase
III
18.9.5.1.5.
Commercial
18.9.5.2. South Africa mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Type
18.9.5.2.1.
Non-replicating
mRNA
18.9.5.2.2.
In vivo
self-replicating mRNA
18.9.5.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
18.9.5.3. South Africa mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Vaccine Modality
18.9.5.3.1.
Therapeutic
18.9.5.3.2.
TME
Modulation
18.9.5.3.3.
Prophylactic
18.9.5.3.4.
DC
Vaccine
18.9.5.3.5.
T Cell
regulation
18.9.5.4. South Africa mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Route of Administration
18.9.5.4.1.
Intravenous/Intradermal
18.9.5.4.2.
Intratumoral
18.9.5.4.3.
Intramuscular
18.9.5.5. South Africa mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Indication
18.9.5.5.1.
Oncology
18.9.5.5.1.1. Solid Tumors
18.9.5.5.1.2. Melanoma
18.9.5.5.1.3. Prostate cancer
18.9.5.5.1.4. Ovarian cancer
18.9.5.5.1.5. Glioblastoma
18.9.5.5.1.6. HPV + Cancer
18.9.5.5.1.7. Renal Cancer
18.9.5.5.1.8. Others
18.9.5.5.2.
Infectious
Diseases
18.9.5.5.2.1. COVID-19 vaccine
18.9.5.5.2.2. Rabies
18.9.5.5.2.3. Lassa, yellow fever
18.9.5.5.2.4. Respirational syncytial virus
18.9.5.5.2.5. Rota
18.9.5.5.2.6. Malaria
18.9.5.5.2.7. Universal influenza
18.9.5.5.2.8. Zika virus
18.9.5.5.2.9. Multiple sclerosis (MS)
18.9.5.5.2.10. Epstein-Barr virus (EBV)
18.9.5.5.2.11. HIV vaccine
18.9.5.5.2.12. Others
18.9.5.5.3.
Rare
Diseases
18.9.5.6. South Africa mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Applications
18.9.5.6.1.
Adolescents
and Pediatric
18.9.5.6.2.
Adults
18.9.5.7. South Africa mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By End-User
18.9.5.7.1.
Healthcare
Facilities
18.9.5.7.2.
Government
Vaccination Campaigns
18.9.5.7.3.
Research
Facilities
18.9.5.7.4.
Others
18.9.6. Rest of Middle East & Africa
18.9.6.1. Rest of Middle East & Africa mRNA
Vaccines Market Revenue (US$ Mn) and Forecasts, By Clinical Trial Phase
18.9.6.1.1.
Preclinical
18.9.6.1.2.
Phase I
18.9.6.1.3.
Phase II
18.9.6.1.4.
Phase
III
18.9.6.1.5.
Commercial
18.9.6.2. Rest of Middle East & Africa mRNA
Vaccines Market Revenue (US$ Mn) and Forecasts, By Type
18.9.6.2.1.
Non-replicating
mRNA
18.9.6.2.2.
In vivo
self-replicating mRNA
18.9.6.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
18.9.6.3. Rest of Middle East & Africa mRNA
Vaccines Market Revenue (US$ Mn) and Forecasts, By Vaccine Modality
18.9.6.3.1.
Therapeutic
18.9.6.3.2.
TME
Modulation
18.9.6.3.3.
Prophylactic
18.9.6.3.4.
DC
Vaccine
18.9.6.3.5.
T Cell
regulation
18.9.6.4. Rest of Middle East & Africa mRNA
Vaccines Market Revenue (US$ Mn) and Forecasts, By Route of Administration
18.9.6.4.1.
Intravenous/Intradermal
18.9.6.4.2.
Intratumoral
18.9.6.4.3.
Intramuscular
18.9.6.5. Rest of Middle East & Africa mRNA
Vaccines Market Revenue (US$ Mn) and Forecasts, By Indication
18.9.6.5.1.
Oncology
18.9.6.5.1.1. Solid Tumors
18.9.6.5.1.2. Melanoma
18.9.6.5.1.3. Prostate cancer
18.9.6.5.1.4. Ovarian cancer
18.9.6.5.1.5. Glioblastoma
18.9.6.5.1.6. HPV + Cancer
18.9.6.5.1.7. Renal Cancer
18.9.6.5.1.8. Others
18.9.6.5.2.
Infectious
Diseases
18.9.6.5.2.1. COVID-19 vaccine
18.9.6.5.2.2. Rabies
18.9.6.5.2.3. Lassa, yellow fever
18.9.6.5.2.4. Respirational syncytial virus
18.9.6.5.2.5. Rota
18.9.6.5.2.6. Malaria
18.9.6.5.2.7. Universal influenza
18.9.6.5.2.8. Zika virus
18.9.6.5.2.9. Multiple sclerosis (MS)
18.9.6.5.2.10. Epstein-Barr virus (EBV)
18.9.6.5.2.11. HIV vaccine
18.9.6.5.2.12. Others
18.9.6.5.3.
Rare
Diseases
18.9.6.6. Rest of Middle East & Africa mRNA
Vaccines Market Revenue (US$ Mn) and Forecasts, By Applications
18.9.6.6.1.
Adolescents
and Pediatric
18.9.6.6.2.
Adults
18.9.6.7. Rest of Middle East & Africa mRNA
Vaccines Market Revenue (US$ Mn) and Forecasts, By End-User
18.9.6.7.1.
Healthcare
Facilities
18.9.6.7.2.
Government
Vaccination Campaigns
18.9.6.7.3.
Research
Facilities
18.9.6.7.4.
Others
18.10. Key Segment for Channeling Investments
18.10.1. By Country
18.10.2. By Clinical Trial Phase
18.10.3. By Type
18.10.4. By Vaccine Modality
18.10.5. By Route of Administration
18.10.6. By Indication
18.10.7. By Applications
18.10.8. By End-User
19. Latin America mRNA Vaccines Market Analysis
and Forecasts, 2021 - 2029
19.1. Overview
19.1.1. Latin America mRNA Vaccines Market Revenue
(US$ Mn)
19.2. Latin America mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Clinical Trial Phase
19.2.1. Preclinical
19.2.2. Phase
I
19.2.3. Phase II
19.2.4. Phase III
19.2.5. Commercial
19.3. Latin America mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Type
19.3.1. Non-replicating mRNA
19.3.2. In vivo self-replicating mRNA
19.3.3. In vitro dendritic cell non-replicating mRNA
vaccine
19.4. Latin America mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Vaccine Modality
19.4.1. Therapeutic
19.4.2. TME Modulation
19.4.3. Prophylactic
19.4.4. DC Vaccine
19.4.5. T Cell regulation
19.5. Latin America mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Route of Administration
19.5.1. Intravenous/Intradermal
19.5.2. Intratumoral
19.5.3. Intramuscular
19.6. Latin America mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Indication
19.6.1. Oncology
19.6.1.1. Solid Tumors
19.6.1.2. Melanoma
19.6.1.3. Prostate cancer
19.6.1.4. Ovarian cancer
19.6.1.5. Glioblastoma
19.6.1.6. HPV + Cancer
19.6.1.7. Renal Cancer
19.6.1.8. Others
19.6.2. Infectious Diseases
19.6.2.1. COVID-19 vaccine
19.6.2.2. Rabies
19.6.2.3. Lassa, yellow fever
19.6.2.4. Respirational syncytial virus
19.6.2.5. Rota
19.6.2.6. Malaria
19.6.2.7. Universal influenza
19.6.2.8. Zika virus
19.6.2.9. Multiple sclerosis (MS)
19.6.2.10. Epstein-Barr virus (EBV)
19.6.2.11. HIV vaccine
19.6.2.12. Others
19.6.3. Rare Diseases
19.7. Latin America mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Applications
19.7.1. Adolescents and Pediatric
19.7.2. Adults
19.8. Latin America mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By End-User
19.8.1. Healthcare Facilities
19.8.2. Government Vaccination Campaigns
19.8.3. Research Facilities
19.8.4. Others
19.9. Latin America mRNA Vaccines Market Revenue
(US$ Mn) and Forecasts, By Country
19.9.1. Brazil
19.9.1.1. Brazil mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Clinical Trial Phase
19.9.1.1.1.
Preclinical
19.9.1.1.2.
Phase I
19.9.1.1.3.
Phase II
19.9.1.1.4.
Phase
III
19.9.1.1.5.
Commercial
19.9.1.2. Brazil mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Type
19.9.1.2.1.
Non-replicating
mRNA
19.9.1.2.2.
In vivo
self-replicating mRNA
19.9.1.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
19.9.1.3. Brazil mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Vaccine Modality
19.9.1.3.1.
Therapeutic
19.9.1.3.2.
TME
Modulation
19.9.1.3.3.
Prophylactic
19.9.1.3.4.
DC
Vaccine
19.9.1.3.5.
T Cell
regulation
19.9.1.4. Brazil mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Route of Administration
19.9.1.4.1.
Intravenous/Intradermal
19.9.1.4.2.
Intratumoral
19.9.1.4.3.
Intramuscular
19.9.1.5. Brazil mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Indication
19.9.1.5.1.
Oncology
19.9.1.5.1.1. Solid Tumors
19.9.1.5.1.2. Melanoma
19.9.1.5.1.3. Prostate cancer
19.9.1.5.1.4. Ovarian cancer
19.9.1.5.1.5. Glioblastoma
19.9.1.5.1.6. HPV + Cancer
19.9.1.5.1.7. Renal Cancer
19.9.1.5.1.8. Others
19.9.1.5.2.
Infectious
Diseases
19.9.1.5.2.1. COVID-19 vaccine
19.9.1.5.2.2. Rabies
19.9.1.5.2.3. Lassa, yellow fever
19.9.1.5.2.4. Respirational syncytial virus
19.9.1.5.2.5. Rota
19.9.1.5.2.6. Malaria
19.9.1.5.2.7. Universal influenza
19.9.1.5.2.8. Zika virus
19.9.1.5.2.9. Multiple sclerosis (MS)
19.9.1.5.2.10. Epstein-Barr virus (EBV)
19.9.1.5.2.11. HIV vaccine
19.9.1.5.2.12. Others
19.9.1.5.3.
Rare
Diseases
19.9.1.6. Brazil mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By Applications
19.9.1.6.1.
Adolescents
and Pediatric
19.9.1.6.2.
Adults
19.9.1.7. Brazil mRNA Vaccines Market Revenue (US$ Mn)
and Forecasts, By End-User
19.9.1.7.1.
Healthcare
Facilities
19.9.1.7.2.
Government
Vaccination Campaigns
19.9.1.7.3.
Research
Facilities
19.9.1.7.4.
Others
19.9.2. Argentina
19.9.2.1. Argentina mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Clinical Trial Phase
19.9.2.1.1.
Preclinical
19.9.2.1.2.
Phase I
19.9.2.1.3.
Phase II
19.9.2.1.4.
Phase
III
19.9.2.1.5.
Commercial
19.9.2.2. Argentina mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Type
19.9.2.2.1.
Non-replicating
mRNA
19.9.2.2.2.
In vivo
self-replicating mRNA
19.9.2.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
19.9.2.3. Argentina mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Vaccine Modality
19.9.2.3.1.
Therapeutic
19.9.2.3.2.
TME
Modulation
19.9.2.3.3.
Prophylactic
19.9.2.3.4.
DC
Vaccine
19.9.2.3.5.
T Cell
regulation
19.9.2.4. Argentina mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Route of Administration
19.9.2.4.1.
Intravenous/Intradermal
19.9.2.4.2.
Intratumoral
19.9.2.4.3.
Intramuscular
19.9.2.5. Argentina mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Indication
19.9.2.5.1.
Oncology
19.9.2.5.1.1. Solid Tumors
19.9.2.5.1.2. Melanoma
19.9.2.5.1.3. Prostate cancer
19.9.2.5.1.4. Ovarian cancer
19.9.2.5.1.5. Glioblastoma
19.9.2.5.1.6. HPV + Cancer
19.9.2.5.1.7. Renal Cancer
19.9.2.5.1.8. Others
19.9.2.5.2.
Infectious
Diseases
19.9.2.5.2.1. COVID-19 vaccine
19.9.2.5.2.2. Rabies
19.9.2.5.2.3. Lassa, yellow fever
19.9.2.5.2.4. Respirational syncytial virus
19.9.2.5.2.5. Rota
19.9.2.5.2.6. Malaria
19.9.2.5.2.7. Universal influenza
19.9.2.5.2.8. Zika virus
19.9.2.5.2.9. Multiple sclerosis (MS)
19.9.2.5.2.10. Epstein-Barr virus (EBV)
19.9.2.5.2.11. HIV vaccine
19.9.2.5.2.12. Others
19.9.2.5.3.
Rare
Diseases
19.9.2.6. Argentina mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By Applications
19.9.2.6.1.
Adolescents
and Pediatric
19.9.2.6.2.
Adults
19.9.2.7. Argentina mRNA Vaccines Market Revenue (US$
Mn) and Forecasts, By End-User
19.9.2.7.1.
Healthcare
Facilities
19.9.2.7.2.
Government
Vaccination Campaigns
19.9.2.7.3.
Research
Facilities
19.9.2.7.4.
Others
19.9.3. Rest of Latin America
19.9.3.1. Rest of Latin America mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Clinical Trial Phase
19.9.3.1.1.
Preclinical
19.9.3.1.2.
Phase I
19.9.3.1.3.
Phase II
19.9.3.1.4.
Phase
III
19.9.3.1.5.
Commercial
19.9.3.2. Rest of Latin America mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Type
19.9.3.2.1.
Non-replicating
mRNA
19.9.3.2.2.
In vivo
self-replicating mRNA
19.9.3.2.3.
In vitro
dendritic cell non-replicating mRNA vaccine
19.9.3.3. Rest of Latin America mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Vaccine Modality
19.9.3.3.1.
Therapeutic
19.9.3.3.2.
TME
Modulation
19.9.3.3.3.
Prophylactic
19.9.3.3.4.
DC
Vaccine
19.9.3.3.5.
T Cell
regulation
19.9.3.4. Rest of Latin America mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Route of Administration
19.9.3.4.1.
Intravenous/Intradermal
19.9.3.4.2.
Intratumoral
19.9.3.4.3.
Intramuscular
19.9.3.5. Rest of Latin America mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Indication
19.9.3.5.1.
Oncology
19.9.3.5.1.1. Solid Tumors
19.9.3.5.1.2. Melanoma
19.9.3.5.1.3. Prostate cancer
19.9.3.5.1.4. Ovarian cancer
19.9.3.5.1.5. Glioblastoma
19.9.3.5.1.6. HPV + Cancer
19.9.3.5.1.7. Renal Cancer
19.9.3.5.1.8. Others
19.9.3.5.2.
Infectious
Diseases
19.9.3.5.2.1. COVID-19 vaccine
19.9.3.5.2.2. Rabies
19.9.3.5.2.3. Lassa, yellow fever
19.9.3.5.2.4. Respirational syncytial virus
19.9.3.5.2.5. Rota
19.9.3.5.2.6. Malaria
19.9.3.5.2.7. Universal influenza
19.9.3.5.2.8. Zika virus
19.9.3.5.2.9. Multiple sclerosis (MS)
19.9.3.5.2.10. Epstein-Barr virus (EBV)
19.9.3.5.2.11. HIV vaccine
19.9.3.5.2.12. Others
19.9.3.5.3.
Rare
Diseases
19.9.3.6. Rest of Latin America mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By Applications
19.9.3.6.1.
Adolescents
and Pediatric
19.9.3.6.2.
Adults
19.9.3.7. Rest of Latin America mRNA Vaccines Market
Revenue (US$ Mn) and Forecasts, By End-User
19.9.3.7.1.
Healthcare
Facilities
19.9.3.7.2.
Government
Vaccination Campaigns
19.9.3.7.3.
Research
Facilities
19.9.3.7.4.
Others
19.10. Key Segment for Channeling Investments
19.10.1. By Country
19.10.2. By Clinical Trial Phase
19.10.3. By Type
19.10.4. By Vaccine Modality
19.10.5. By Route of Administration
19.10.6. By Indication
19.10.7. By Applications
19.10.8. By End-User
20. Competitive Benchmarking
20.1. Brand Benchmarking
20.2. Market Share Analysis, 2020
20.3. Global Presence and Growth Strategies
20.3.1. Mergers and Acquisitions
20.3.2. Product Launches
20.3.3. Investments Trends
20.3.4. R&D Initiatives
21. Player Profiles
21.1. Argos Therapeutics
21.1.1. Company Details
21.1.2. Company Overview
21.1.3. Product Offerings
21.1.4. Key Developments
21.1.5. Financial Analysis
21.1.6. SWOT Analysis
21.1.7. Business Strategies
21.2. BioNTech SE
21.2.1. Company Details
21.2.2. Company Overview
21.2.3. Product Offerings
21.2.4. Key Developments
21.2.5. Financial Analysis
21.2.6. SWOT Analysis
21.2.7. Business Strategies
21.3. cōIMMUNE
21.3.1. Company Details
21.3.2. Company Overview
21.3.3. Product Offerings
21.3.4. Key Developments
21.3.5. Financial Analysis
21.3.6. SWOT Analysis
21.3.7. Business Strategies
21.4. CureVac AG
21.4.1. Company Details
21.4.2. Company Overview
21.4.3. Product Offerings
21.4.4. Key Developments
21.4.5. Financial Analysis
21.4.6. SWOT Analysis
21.4.7. Business Strategies
21.5. eTheRNA
21.5.1. Company Details
21.5.2. Company Overview
21.5.3. Product Offerings
21.5.4. Key Developments
21.5.5. Financial Analysis
21.5.6. SWOT Analysis
21.5.7. Business Strategies
21.6. Genentech, Inc
21.6.1. Company Details
21.6.2. Company Overview
21.6.3. Product Offerings
21.6.4. Key Developments
21.6.5. Financial Analysis
21.6.6. SWOT Analysis
21.6.7. Business Strategies
21.7. Immunomic Therapeutics, Inc.
21.7.1. Company Details
21.7.2. Company Overview
21.7.3. Product Offerings
21.7.4. Key Developments
21.7.5. Financial Analysis
21.7.6. SWOT Analysis
21.7.7. Business Strategies
21.8. Moderna, Inc.
21.8.1. Company Details
21.8.2. Company Overview
21.8.3. Product Offerings
21.8.4. Key Developments
21.8.5. Financial Analysis
21.8.6. SWOT Analysis
21.8.7. Business Strategies
21.9. Pfizer Inc.
21.9.1. Company Details
21.9.2. Company Overview
21.9.3. Product Offerings
21.9.4. Key Developments
21.9.5. Financial Analysis
21.9.6. SWOT Analysis
21.9.7. Business Strategies
21.10. Other Market Participants
22. Key Findings
**Exclusive for Multi-User and
Enterprise User.
At Absolute Markets Insights, we are engaged in building both global as well as country specific reports. As a result, the approach taken for deriving the estimation and forecast for a specific country is a bit unique and different in comparison to the global research studies. In this case, we not only study the concerned market factors & trends prevailing in a particular country (from secondary research) but we also tend to calculate the actual market size & forecast from the revenue generated from the market participants involved in manufacturing or distributing the any concerned product. These companies can also be service providers. For analyzing any country specifically, we do consider the growth factors prevailing under the states/cities/county for the same. For instance, if we are analyzing an industry specific to United States, we primarily need to study about the states present under the same(where the product/service has the highest growth). Similar analysis will be followed by other countries. Our scope of the report changes with different markets.
Our research study is mainly implement through a mix of both secondary and primary research. Various sources such as industry magazines, trade journals, and government websites and trade associations are reviewed for gathering precise data. Primary interviews are conducted to validate the market size derived from secondary research. Industry experts, major manufacturers and distributors are contacted for further validation purpose on the current market penetration and growth trends.
Prominent participants in our primary research process include:
- Key Opinion Leaders namely the CEOs, CSOs, VPs, purchasing managers, amongst others
- Research and development participants, distributors/suppliers and subject matter experts
Secondary Research includes data extracted from paid data sources:
- Reuters
- Factiva
- Bloomberg
- One Source
- Hoovers
Research Methodology
Key Inclusions

Reach to us
Call us on
+91-74002-42424
Drop us an email at
sales@absolutemarketsinsights.com
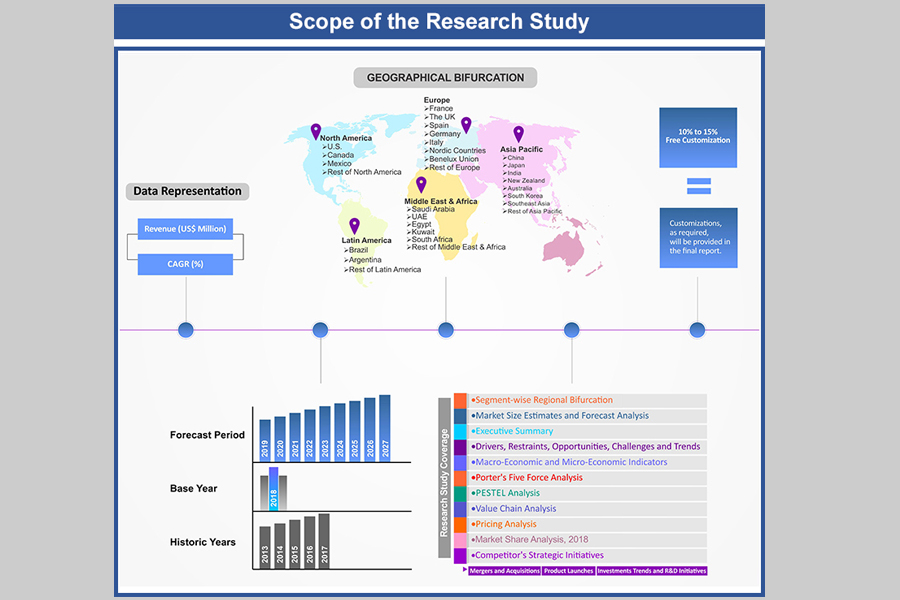
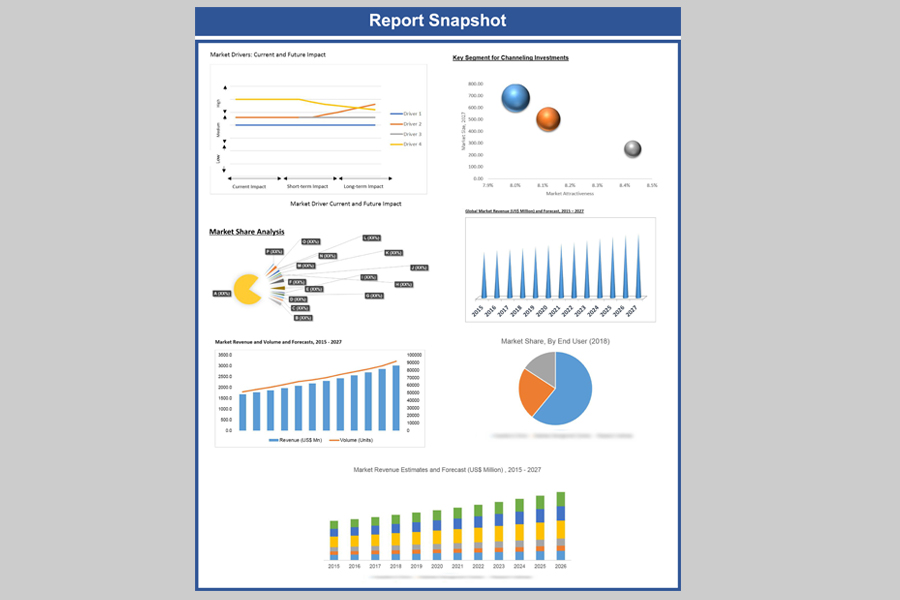
Why Absolute Markets Insights?
An effective strategy is the entity that influences a business to stand out of the crowd. An organization with a phenomenal strategy for success dependably has the edge over the rivals in the market. It offers the organizations a head start in planning their strategy. Absolute Market Insights is the new initiation in the industry that will furnish you with the lead your business needs. Absolute Market Insights is the best destination for your business intelligence and analytical solutions; essentially because our qualitative and quantitative sources of information are competent to give one-stop solutions. We inventively combine qualitative and quantitative research in accurate proportions to have the best report, which not only gives the most recent insights but also assists you to grow.