Japan IVD Quality Control Market By Offerings (Quality Control Products (Serum based ,Whole Blood ,Urine) Quality Assurance Services, Data Management Solutions) By Control (Analytes) (Immunochemistry ,Clinical Chemistry ,Microbiology ,Hematology ,Coagulation ,Infectious disease ,Diabetics/ Hemoglobin ,Cardiology ,Others) By Manufacturer Type (Dependent/First-Party Controls, Independent/Third Party Controls) By End Users (Hospitals and Clinics ,Laboratories, Research Institutes, Others); By Region (Hokkaido, Tohoku, Kanto, Chubu, Kinki/Kansai, Chugoku, Shikoku, Kyushu (incl. Okinawa)) – Japan Insights, Growth, Size, Comparative Analysis, Trends and Forecast, 2023 – 2031
In vitro quality control (IVQC) is a crucial component of quality assurance in the pharmaceutical, biotechnology, and medical device industries. It involves a series of tests and procedures conducted outside the living organism (in vitro) to assess the quality, safety, and efficacy of products before they are introduced to the market or used in clinical trials. Japan IVD quality control market is expected to witness CAGR ~4.5% during 2023 – 2031.
Increasing Prevalence of Chronic Diseases: Japan IVD Quality Control Market
The increasing prevalence of chronic diseases such as cancer, diabetes, and cardiovascular diseases is one of the significant drivers of the IVD quality control market in Japan. According to the World Health Organization (WHO), chronic diseases are responsible for 71% of all deaths globally. Japan has one of the world's oldest populations, with a significant proportion of elderly individuals. With increasing age comes a higher risk of developing chronic diseases such as cardiovascular diseases, diabetes, cancer, and neurodegenerative disorders. Furthermore, rapid urbanization and modernization have brought about changes in lifestyle habits in Japan. A shift toward a more sedentary lifestyle, unhealthy diets, and smoking have contributed to the rise in chronic diseases.
Growing Demand for Diagnostic Testing Services: Japan IVD Quality Control Market
As the demand for healthcare services increases, there has been a growing need for diagnostic testing services worldwide, including Japan. The rising prevalence of chronic diseases, an aging population, and increased health awareness are some of the factors driving this demand. Diagnostic testing is critical for early detection, accurate diagnosis, and effective disease management, leading to better patient outcomes and reduced healthcare costs in the long run.
In Japan, the demand for diagnostic testing services has led to a parallel growth in the In Vitro Diagnostics (IVD) industry. IVD refers to medical devices, reagents, and accessories used to perform tests on samples taken from the human body (e.g., blood, urine, tissue) to diagnose diseases or monitor health conditions. It plays a pivotal role in supporting healthcare professionals' decisions by providing them with essential information to deliver precise and targeted treatments. The IVD quality control market in Japan has become increasingly important to ensure the accuracy, reliability, and safety of diagnostic tests. Quality control measures are crucial for verifying the performance of IVD products, minimizing errors, and maintaining consistency in results across different testing facilities.
Japan IVD Quality Control Market Revenue & Forecast, (US$ Million), 2015 – 2031
Quality control products accounted for a substantial market share in the IVD quality control market in 2022. Quality control products are essential components in ensuring the accuracy, reliability, and consistency of diagnostic testing in clinical laboratories and other healthcare settings. The healthcare industry in Japan emphasizes continuous improvement methodologies, such as Lean Six Sigma, to optimize processes and enhance overall quality. Quality control products support such initiatives by providing data to monitor and improve testing procedures. Additionally, advancements in quality control products, including the development of multi-analyte controls and third-party quality control materials, have contributed to their widespread adoption in the IVD market.
In the dynamic IVD quality control market in Japan, hospitals and clinics have consistently accounted for a lion's share of the market. These healthcare facilities play a crucial role in diagnostic testing, offering a wide range of medical services and requiring a robust quality control system to ensure accurate and reliable results. Hospitals and clinics serve as primary points of care for patients, diagnosing and treating various medical conditions. As a result, they heavily rely on IVD tests to aid in the diagnosis and monitoring of diseases. To maintain high standards of care, these facilities invest significantly in quality control products, such as control materials, calibrators, and proficiency testing materials.
The stringent regulatory environment in Japan emphasizes the importance of quality control in diagnostic testing. Hospitals and clinics, being responsible for patient outcomes, prioritize the implementation of effective quality control measures. This includes adherence to regulatory standards, proficiency testing programs, and ongoing performance evaluations to ensure accurate and reliable test results. The lion's share of the IVD quality control market held by hospitals and clinics is also attributed to the volume of testing conducted at these facilities. As major healthcare providers in the country, they handle a vast number of patient samples, necessitating a significant demand for quality control products to maintain accuracy and precision across their testing processes.
Furthermore, hospitals and clinics actively participate in accreditation programs and seek certifications from recognized bodies, further highlighting their commitment to quality and accuracy in diagnostic testing. These efforts reinforce the importance of quality control in their daily operations, driving the demand for IVD quality control market.
In conclusion, hospitals and clinics in Japan play a dominant role in the IVD quality control market due to their substantial testing volume, commitment to regulatory compliance, and focus on delivering accurate and reliable diagnostic services to their patients. As the demand for high-quality healthcare services continues to grow, the importance of quality control in these facilities will remain central to their operations and position in the IVD market.
Competitive Landscape
The report provides both, qualitative and quantitative research of Japan IVD quality control market, as well as provides comprehensive insights and development methods adopted by the key contenders. The report also offers extensive research on the key players in this market and details on the competitiveness of these players. Key business strategies such as mergers and acquisitions (M&A), affiliations, collaborations, and contracts adopted by these major market participants are also recognized and analyzed in the report. For each company, the report studies their Japan presence, competitors, service offerings and specification amongst others.
Some of the players operating in the Japan IVD quality control market are:
- Abbott Diagnostics
- Beckman Coulter, Inc.
- Bio-Rad Laboratories, Inc.
- F. Hoffmann-La Roche Ltd
- Fortress Diagnostics
- Fujirebio Inc.
- Horiba Medical Japan Co., Ltd.
- Randox Laboratories
- Sekisui Medical Co., Ltd.
- Sysmex Corporation
- Thermo Fisher Scientific Inc.
- Other Market Participants
Japan IVD Quality Control Market
- Quality control Products
- Serum based
- Whole Blood
- Urine
- Quality Assurance Services
- Data Management Solutions
By Control (Analytes)
- Immunochemistry
- Clinical Chemistry
- Microbiology
- Hematology
- Coagulation
- Infectious disease
- Diabetics/ Hemoglobin
- Cardiology
- Others
By Manufacturer Type
- Dependent/first-party controls
- Independent/third party controls
By End Users
- Hospitals and Clinics
- Laboratories
- Research Institutes
- Others
By Region
- Hokkaido
- Tohoku
- Kanto
- Chubu
- Kinki/Kansai
- Chugoku
- Shikoku
- Kyushu (incl. Okinawa)
Table of Contents
1. Market Scope
1.1. Market
Segmentation
1.2. Years
Considered
1.2.1. Historic
Years: 2015 - 2021
1.2.2. Base
Year: 2022
1.2.3. Forecast
Years: 2023 – 2031
2. Key Target Audiences
3. Research Methodology
3.1. Primary
Research
3.1.1. Research
Questionnaire
3.1.2. Japan
Percentage Breakdown
3.1.3. Primary
Interviews: Key Opinion Leaders (KOLs)
3.2. Secondary
Research
3.2.1. Paid
Databases
3.2.2. Secondary
Sources
3.3. Market
Size Estimates
3.3.1. Top-Down
Approach
3.3.2. Bottom-Up
Approach
3.4. Data
Triangulation Methodology
3.5. Research
Assumptions
4. Recommendations and Insights from AMI’s Perspective**
5. Holistic Overview of IVD Quality Control Market
6. Market Synopsis: IVD
Quality Control Market
7. IVD Quality Control Market Analysis: Qualitative Perspective
7.1. Introduction
7.1.1. Product
Definition
7.1.2. Industry
Development
7.2. Market
Dynamics
7.2.1. Drivers
7.2.2. Restraints
7.2.3. Opportunities
7.3. Trends in
IVD Quality Control Market
7.4. Market
Determinants Radar Chart
7.5. Macro-Economic
and Micro-Economic Indicators: IVD Quality Control Market
7.6. Porter’s
Five Force Analysis
7.7. Impact of
Covid-19 on IVD Quality Control Market
8. Japan IVD Quality Control Market Analysis and Forecasts, 2023
- 2031
8.1. Overview
8.1.1. Japan
IVD Quality Control Market Revenue (US$ Mn)
8.2. Japan IVD
Quality Control Market Revenue (US$ Mn) and
Forecasts, By Offerings
8.2.1. Quality
Control Products (Definition, Market Penetration (2019), Market Estimation
(2015 - 2022), Market Forecast (2023 - 2031), Compound Annual Growth Rate
(CAGR), Regional Bifurcation (Hokkaido, Tohoku, Kanto, Chubu, Kinki/Kansai, Chugoku,
Shikoku, Kyushu (incl. Okinawa)) and Information on Serum Based, Whole Blood,
Urine)
8.2.1.1. Serum Based
8.2.1.2. Whole Blood
8.2.1.3. Urine
8.2.2. Data
Management Solutions
8.2.2.1. Definition
8.2.2.2. Market Estimation and Penetration, 2015 - 2022
8.2.2.3. Market Forecast, 2023 - 2031
8.2.2.4.
Compound Annual Growth
Rate (CAGR)
8.2.2.5.
Regional Bifurcation
8.2.2.5.1. Hokkaido
8.2.2.5.1.1. Market
Estimation, 2015 – 2022
8.2.2.5.1.2. Market
Forecast, 2023 – 2031
8.2.2.5.2. Tohoku
8.2.2.5.2.1. Market
Estimation, 2015 – 2022
8.2.2.5.2.2. Market
Forecast, 2023 – 2031
8.2.2.5.3. Kanto
8.2.2.5.3.1. Market
Estimation, 2015 – 2022
8.2.2.5.3.2. Market
Forecast, 2023 – 2031
8.2.2.5.4. Chubu
8.2.2.5.4.1. Market
Estimation, 2015 – 2022
8.2.2.5.4.2. Market
Forecast, 2023 – 2031
8.2.2.5.5. Kinki/Kansai
8.2.2.5.5.1. Market
Estimation, 2015 – 2022
8.2.2.5.5.2. Market
Forecast, 2023 – 2031
8.2.2.5.6. Chugoku
8.2.2.5.6.1. Market
Estimation, 2015 – 2022
8.2.2.5.6.2. Market
Forecast, 2023 – 2031
8.2.2.5.7. Shikoku
8.2.2.5.7.1. Market
Estimation, 2015 – 2022
8.2.2.5.7.2. Market
Forecast, 2023 – 2031
8.2.2.5.8. Kyushu
(incl. Okinawa)
8.2.2.5.8.1. Market
Estimation, 2015 – 2022
8.2.2.5.8.2. Market
Forecast, 2023 – 2031
8.2.3. Quality
Assurance Services
8.2.3.1. Definition
8.2.3.2. Market Estimation and Penetration, 2015 - 2022
8.2.3.3. Market Forecast, 2023 - 2031
8.2.3.4.
Compound Annual Growth Rate
(CAGR)
8.2.3.5.
Regional Bifurcation
8.2.3.5.1. Hokkaido
8.2.3.5.1.1. Market
Estimation, 2015 – 2022
8.2.3.5.1.2. Market
Forecast, 2023 – 2031
8.2.3.5.2. Tohoku
8.2.3.5.2.1. Market
Estimation, 2015 – 2022
8.2.3.5.2.2. Market
Forecast, 2023 – 2031
8.2.3.5.3. Kanto
8.2.3.5.3.1. Market
Estimation, 2015 – 2022
8.2.3.5.3.2. Market
Forecast, 2023 – 2031
8.2.3.5.4. Chubu
8.2.3.5.4.1. Market
Estimation, 2015 – 2022
8.2.3.5.4.2. Market
Forecast, 2023 – 2031
8.2.3.5.5. Kinki/Kansai
8.2.3.5.5.1. Market
Estimation, 2015 – 2022
8.2.3.5.5.2. Market
Forecast, 2023 – 2031
8.2.3.5.6. Chugoku
8.2.3.5.6.1. Market
Estimation, 2015 – 2022
8.2.3.5.6.2. Market
Forecast, 2023 – 2031
8.2.3.5.7. Shikoku
8.2.3.5.7.1. Market
Estimation, 2015 – 2022
8.2.3.5.7.2. Market
Forecast, 2023 – 2031
8.2.3.5.8. Kyushu
(incl. Okinawa)
8.2.3.5.8.1. Market
Estimation, 2015 – 2022
8.2.3.5.8.2. Market
Forecast, 2023 – 2031
8.3. Key
Segment for Channeling Investments
8.3.1. By
Offerings
9. Japan IVD Quality Control Market Analysis and Forecasts, 2023
- 2031
9.1. Overview
9.2. Japan IVD
Quality Control Market Revenue (US$ Mn) and
Forecasts, By Control (Analytes)
9.2.1. Immunochemistry
9.2.1.1. Definition
9.2.1.2. Market Estimation and Penetration, 2015 - 2022
9.2.1.3. Market Forecast, 2023 - 2031
9.2.1.4.
Compound Annual Growth
Rate (CAGR)
9.2.1.5.
Regional Bifurcation
9.2.1.5.1. Hokkaido
9.2.1.5.1.1. Market
Estimation, 2015 – 2022
9.2.1.5.1.2. Market
Forecast, 2023 – 2031
9.2.1.5.2. Tohoku
9.2.1.5.2.1. Market
Estimation, 2015 – 2022
9.2.1.5.2.2. Market
Forecast, 2023 – 2031
9.2.1.5.3. Kanto
9.2.1.5.3.1. Market
Estimation, 2015 – 2022
9.2.1.5.3.2. Market
Forecast, 2023 – 2031
9.2.1.5.4. Chubu
9.2.1.5.4.1. Market
Estimation, 2015 – 2022
9.2.1.5.4.2. Market
Forecast, 2023 – 2031
9.2.1.5.5. Kinki/Kansai
9.2.1.5.5.1. Market
Estimation, 2015 – 2022
9.2.1.5.5.2. Market
Forecast, 2023 – 2031
9.2.1.5.6. Chugoku
9.2.1.5.6.1. Market
Estimation, 2015 – 2022
9.2.1.5.6.2. Market
Forecast, 2023 – 2031
9.2.1.5.7. Shikoku
9.2.1.5.7.1. Market
Estimation, 2015 – 2022
9.2.1.5.7.2. Market
Forecast, 2023 – 2031
9.2.1.5.8. Kyushu
(incl. Okinawa)
9.2.1.5.8.1. Market
Estimation, 2015 – 2022
9.2.1.5.8.2. Market
Forecast, 2023 – 2031
9.2.2. Clinical
Chemistry
9.2.2.1. Definition
9.2.2.2. Market Estimation and Penetration, 2015 - 2022
9.2.2.3. Market Forecast, 2023 - 2031
9.2.2.4.
Compound Annual Growth
Rate (CAGR)
9.2.2.5.
Regional Bifurcation
9.2.2.5.1. Hokkaido
9.2.2.5.1.1. Market
Estimation, 2015 – 2022
9.2.2.5.1.2. Market
Forecast, 2023 – 2031
9.2.2.5.2. Tohoku
9.2.2.5.2.1. Market
Estimation, 2015 – 2022
9.2.2.5.2.2. Market
Forecast, 2023 – 2031
9.2.2.5.3. Kanto
9.2.2.5.3.1. Market
Estimation, 2015 – 2022
9.2.2.5.3.2. Market
Forecast, 2023 – 2031
9.2.2.5.4. Chubu
9.2.2.5.4.1. Market
Estimation, 2015 – 2022
9.2.2.5.4.2. Market
Forecast, 2023 – 2031
9.2.2.5.5. Kinki/Kansai
9.2.2.5.5.1. Market
Estimation, 2015 – 2022
9.2.2.5.5.2. Market
Forecast, 2023 – 2031
9.2.2.5.6. Chugoku
9.2.2.5.6.1. Market
Estimation, 2015 – 2022
9.2.2.5.6.2. Market
Forecast, 2023 – 2031
9.2.2.5.7. Shikoku
9.2.2.5.7.1. Market
Estimation, 2015 – 2022
9.2.2.5.7.2. Market
Forecast, 2023 – 2031
9.2.2.5.8. Kyushu
(incl. Okinawa)
9.2.2.5.8.1. Market
Estimation, 2015 – 2022
9.2.2.5.8.2. Market
Forecast, 2023 – 2031
9.2.3. Microbiology
9.2.3.1. Definition
9.2.3.2. Market Estimation and Penetration, 2015 - 2022
9.2.3.3. Market Forecast, 2023 - 2031
9.2.3.4.
Compound Annual Growth
Rate (CAGR)
9.2.3.5.
Regional Bifurcation
9.2.3.5.1. Hokkaido
9.2.3.5.1.1. Market
Estimation, 2015 – 2022
9.2.3.5.1.2. Market
Forecast, 2023 – 2031
9.2.3.5.2. Tohoku
9.2.3.5.2.1. Market
Estimation, 2015 – 2022
9.2.3.5.2.2. Market
Forecast, 2023 – 2031
9.2.3.5.3. Kanto
9.2.3.5.3.1. Market
Estimation, 2015 – 2022
9.2.3.5.3.2. Market
Forecast, 2023 – 2031
9.2.3.5.4. Chubu
9.2.3.5.4.1. Market
Estimation, 2015 – 2022
9.2.3.5.4.2. Market
Forecast, 2023 – 2031
9.2.3.5.5. Kinki/Kansai
9.2.3.5.5.1. Market
Estimation, 2015 – 2022
9.2.3.5.5.2. Market
Forecast, 2023 – 2031
9.2.3.5.6. Chugoku
9.2.3.5.6.1. Market
Estimation, 2015 – 2022
9.2.3.5.6.2. Market
Forecast, 2023 – 2031
9.2.3.5.7. Shikoku
9.2.3.5.7.1. Market
Estimation, 2015 – 2022
9.2.3.5.7.2. Market
Forecast, 2023 – 2031
9.2.3.5.8. Kyushu
(incl. Okinawa)
9.2.3.5.8.1. Market
Estimation, 2015 – 2022
9.2.3.5.8.2. Market
Forecast, 2023 – 2031
9.2.4. Hematology
9.2.4.1. Definition
9.2.4.2. Market Estimation and Penetration, 2015 - 2022
9.2.4.3. Market Forecast, 2023 - 2031
9.2.4.4.
Compound Annual Growth
Rate (CAGR)
9.2.4.5.
Regional Bifurcation
9.2.4.5.1. Hokkaido
9.2.4.5.1.1. Market
Estimation, 2015 – 2022
9.2.4.5.1.2. Market
Forecast, 2023 – 2031
9.2.4.5.2. Tohoku
9.2.4.5.2.1. Market
Estimation, 2015 – 2022
9.2.4.5.2.2. Market
Forecast, 2023 – 2031
9.2.4.5.3. Kanto
9.2.4.5.3.1. Market
Estimation, 2015 – 2022
9.2.4.5.3.2. Market
Forecast, 2023 – 2031
9.2.4.5.4. Chubu
9.2.4.5.4.1. Market
Estimation, 2015 – 2022
9.2.4.5.4.2. Market
Forecast, 2023 – 2031
9.2.4.5.5. Kinki/Kansai
9.2.4.5.5.1. Market
Estimation, 2015 – 2022
9.2.4.5.5.2. Market
Forecast, 2023 – 2031
9.2.4.5.6. Chugoku
9.2.4.5.6.1. Market
Estimation, 2015 – 2022
9.2.4.5.6.2. Market
Forecast, 2023 – 2031
9.2.4.5.7. Shikoku
9.2.4.5.7.1. Market
Estimation, 2015 – 2022
9.2.4.5.7.2. Market
Forecast, 2023 – 2031
9.2.4.5.8. Kyushu
(incl. Okinawa)
9.2.4.5.8.1. Market
Estimation, 2015 – 2022
9.2.4.5.8.2. Market
Forecast, 2023 – 2031
9.2.5. Coagulation
9.2.5.1. Definition
9.2.5.2. Market Estimation and Penetration, 2015 - 2022
9.2.5.3. Market Forecast, 2023 - 2031
9.2.5.4.
Compound Annual Growth
Rate (CAGR)
9.2.5.5.
Regional Bifurcation
9.2.5.5.1. Hokkaido
9.2.5.5.1.1. Market
Estimation, 2015 – 2022
9.2.5.5.1.2. Market
Forecast, 2023 – 2031
9.2.5.5.2. Tohoku
9.2.5.5.2.1. Market
Estimation, 2015 – 2022
9.2.5.5.2.2. Market
Forecast, 2023 – 2031
9.2.5.5.3. Kanto
9.2.5.5.3.1. Market
Estimation, 2015 – 2022
9.2.5.5.3.2. Market
Forecast, 2023 – 2031
9.2.5.5.4. Chubu
9.2.5.5.4.1. Market
Estimation, 2015 – 2022
9.2.5.5.4.2. Market
Forecast, 2023 – 2031
9.2.5.5.5. Kinki/Kansai
9.2.5.5.5.1. Market
Estimation, 2015 – 2022
9.2.5.5.5.2. Market
Forecast, 2023 – 2031
9.2.5.5.6. Chugoku
9.2.5.5.6.1. Market
Estimation, 2015 – 2022
9.2.5.5.6.2. Market
Forecast, 2023 – 2031
9.2.5.5.7. Shikoku
9.2.5.5.7.1. Market
Estimation, 2015 – 2022
9.2.5.5.7.2. Market
Forecast, 2023 – 2031
9.2.5.5.8. Kyushu
(incl. Okinawa)
9.2.5.5.8.1. Market
Estimation, 2015 – 2022
9.2.5.5.8.2. Market
Forecast, 2023 – 2031
9.2.6. Infectious
Disease
9.2.6.1. Definition
9.2.6.2. Market Estimation and Penetration, 2015 - 2022
9.2.6.3. Market Forecast, 2023 - 2031
9.2.6.4.
Compound Annual Growth
Rate (CAGR)
9.2.6.5.
Regional Bifurcation
9.2.6.5.1. Hokkaido
9.2.6.5.1.1. Market
Estimation, 2015 – 2022
9.2.6.5.1.2. Market
Forecast, 2023 – 2031
9.2.6.5.2. Tohoku
9.2.6.5.2.1. Market
Estimation, 2015 – 2022
9.2.6.5.2.2. Market
Forecast, 2023 – 2031
9.2.6.5.3. Kanto
9.2.6.5.3.1. Market
Estimation, 2015 – 2022
9.2.6.5.3.2. Market
Forecast, 2023 – 2031
9.2.6.5.4. Chubu
9.2.6.5.4.1. Market
Estimation, 2015 – 2022
9.2.6.5.4.2. Market
Forecast, 2023 – 2031
9.2.6.5.5. Kinki/Kansai
9.2.6.5.5.1. Market
Estimation, 2015 – 2022
9.2.6.5.5.2. Market
Forecast, 2023 – 2031
9.2.6.5.6. Chugoku
9.2.6.5.6.1. Market
Estimation, 2015 – 2022
9.2.6.5.6.2. Market
Forecast, 2023 – 2031
9.2.6.5.7. Shikoku
9.2.6.5.7.1. Market
Estimation, 2015 – 2022
9.2.6.5.7.2. Market
Forecast, 2023 – 2031
9.2.6.5.8. Kyushu
(incl. Okinawa)
9.2.6.5.8.1. Market
Estimation, 2015 – 2022
9.2.6.5.8.2. Market
Forecast, 2023 – 2031
9.2.7. Diabetics/
Hemoglobin
9.2.7.1. Definition
9.2.7.2. Market Estimation and Penetration, 2015 - 2022
9.2.7.3. Market Forecast, 2023 - 2031
9.2.7.4.
Compound Annual Growth
Rate (CAGR)
9.2.7.5.
Regional Bifurcation
9.2.7.5.1. Hokkaido
9.2.7.5.1.1. Market
Estimation, 2015 – 2022
9.2.7.5.1.2. Market
Forecast, 2023 – 2031
9.2.7.5.2. Tohoku
9.2.7.5.2.1. Market
Estimation, 2015 – 2022
9.2.7.5.2.2. Market
Forecast, 2023 – 2031
9.2.7.5.3. Kanto
9.2.7.5.3.1. Market
Estimation, 2015 – 2022
9.2.7.5.3.2. Market
Forecast, 2023 – 2031
9.2.7.5.4. Chubu
9.2.7.5.4.1. Market
Estimation, 2015 – 2022
9.2.7.5.4.2. Market
Forecast, 2023 – 2031
9.2.7.5.5. Kinki/Kansai
9.2.7.5.5.1. Market
Estimation, 2015 – 2022
9.2.7.5.5.2. Market
Forecast, 2023 – 2031
9.2.7.5.6. Chugoku
9.2.7.5.6.1. Market
Estimation, 2015 – 2022
9.2.7.5.6.2. Market
Forecast, 2023 – 2031
9.2.7.5.7. Shikoku
9.2.7.5.7.1. Market
Estimation, 2015 – 2022
9.2.7.5.7.2. Market
Forecast, 2023 – 2031
9.2.7.5.8. Kyushu
(incl. Okinawa)
9.2.7.5.8.1. Market
Estimation, 2015 – 2022
9.2.7.5.8.2. Market
Forecast, 2023 – 2031
9.2.8. Cardiology
9.2.8.1. Definition
9.2.8.2. Market Estimation and Penetration, 2015 - 2022
9.2.8.3. Market Forecast, 2023 - 2031
9.2.8.4.
Compound Annual Growth
Rate (CAGR)
9.2.8.5.
Regional Bifurcation
9.2.8.5.1. Hokkaido
9.2.8.5.1.1. Market
Estimation, 2015 – 2022
9.2.8.5.1.2. Market
Forecast, 2023 – 2031
9.2.8.5.2. Tohoku
9.2.8.5.2.1. Market
Estimation, 2015 – 2022
9.2.8.5.2.2. Market
Forecast, 2023 – 2031
9.2.8.5.3. Kanto
9.2.8.5.3.1. Market
Estimation, 2015 – 2022
9.2.8.5.3.2. Market
Forecast, 2023 – 2031
9.2.8.5.4. Chubu
9.2.8.5.4.1. Market
Estimation, 2015 – 2022
9.2.8.5.4.2. Market
Forecast, 2023 – 2031
9.2.8.5.5. Kinki/Kansai
9.2.8.5.5.1. Market
Estimation, 2015 – 2022
9.2.8.5.5.2. Market
Forecast, 2023 – 2031
9.2.8.5.6. Chugoku
9.2.8.5.6.1. Market
Estimation, 2015 – 2022
9.2.8.5.6.2. Market
Forecast, 2023 – 2031
9.2.8.5.7. Shikoku
9.2.8.5.7.1. Market
Estimation, 2015 – 2022
9.2.8.5.7.2. Market
Forecast, 2023 – 2031
9.2.8.5.8. Kyushu
(incl. Okinawa)
9.2.8.5.8.1. Market
Estimation, 2015 – 2022
9.2.8.5.8.2. Market
Forecast, 2023 – 2031
9.2.9. Others
9.2.9.1. Definition
9.2.9.2. Market Estimation and Penetration, 2015 - 2022
9.2.9.3. Market Forecast, 2023 - 2031
9.2.9.4. Compound Annual Growth Rate (CAGR)
9.3. Key
Segment for Channeling Investments
9.3.1. By
Control (Analytes)
10. Japan IVD Quality Control Market Analysis and Forecasts, 2023
- 2031
10.1. Overview
10.2. Japan IVD
Quality Control Market Revenue (US$ Mn) and
Forecasts, By Manufacturer Type
10.2.1. Dependent/first-party
controls
10.2.1.1. Definition
10.2.1.2. Market Estimation and Penetration, 2015 - 2022
10.2.1.3. Market Forecast, 2023 - 2031
10.2.1.4.
Compound Annual Growth
Rate (CAGR)
10.2.1.5.
Regional Bifurcation
10.2.1.5.1. Hokkaido
10.2.1.5.1.1. Market
Estimation, 2015 – 2022
10.2.1.5.1.2. Market
Forecast, 2023 – 2031
10.2.1.5.2. Tohoku
10.2.1.5.2.1. Market
Estimation, 2015 – 2022
10.2.1.5.2.2. Market
Forecast, 2023 – 2031
10.2.1.5.3. Kanto
10.2.1.5.3.1. Market
Estimation, 2015 – 2022
10.2.1.5.3.2. Market
Forecast, 2023 – 2031
10.2.1.5.4. Chubu
10.2.1.5.4.1. Market
Estimation, 2015 – 2022
10.2.1.5.4.2. Market
Forecast, 2023 – 2031
10.2.1.5.5. Kinki/Kansai
10.2.1.5.5.1. Market
Estimation, 2015 – 2022
10.2.1.5.5.2. Market
Forecast, 2023 – 2031
10.2.1.5.6. Chugoku
10.2.1.5.6.1. Market
Estimation, 2015 – 2022
10.2.1.5.6.2. Market
Forecast, 2023 – 2031
10.2.1.5.7. Shikoku
10.2.1.5.7.1. Market
Estimation, 2015 – 2022
10.2.1.5.7.2. Market
Forecast, 2023 – 2031
10.2.1.5.8. Kyushu
(incl. Okinawa)
10.2.1.5.8.1. Market
Estimation, 2015 – 2022
10.2.1.5.8.2. Market
Forecast, 2023 – 2031
10.2.2. Independent/third
party controls
10.2.2.1. Definition
10.2.2.2. Market Estimation and Penetration, 2015 - 2022
10.2.2.3. Market Forecast, 2023 - 2031
10.2.2.4.
Compound Annual Growth
Rate (CAGR)
10.2.2.5.
Regional Bifurcation
10.2.2.5.1. Hokkaido
10.2.2.5.1.1. Market
Estimation, 2015 – 2022
10.2.2.5.1.2. Market
Forecast, 2023 – 2031
10.2.2.5.2. Tohoku
10.2.2.5.2.1. Market
Estimation, 2015 – 2022
10.2.2.5.2.2. Market
Forecast, 2023 – 2031
10.2.2.5.3. Kanto
10.2.2.5.3.1. Market
Estimation, 2015 – 2022
10.2.2.5.3.2. Market
Forecast, 2023 – 2031
10.2.2.5.4. Chubu
10.2.2.5.4.1. Market
Estimation, 2015 – 2022
10.2.2.5.4.2. Market
Forecast, 2023 – 2031
10.2.2.5.5. Kinki/Kansai
10.2.2.5.5.1. Market
Estimation, 2015 – 2022
10.2.2.5.5.2. Market
Forecast, 2023 – 2031
10.2.2.5.6. Chugoku
10.2.2.5.6.1. Market
Estimation, 2015 – 2022
10.2.2.5.6.2. Market
Forecast, 2023 – 2031
10.2.2.5.7. Shikoku
10.2.2.5.7.1. Market
Estimation, 2015 – 2022
10.2.2.5.7.2. Market
Forecast, 2023 – 2031
10.2.2.5.8. Kyushu
(incl. Okinawa)
10.2.2.5.8.1. Market
Estimation, 2015 – 2022
10.2.2.5.8.2. Market
Forecast, 2023 – 2031
10.3. Key
Segment for Channeling Investments
10.3.1. By
Manufacturer Type
11. Japan IVD Quality Control Market Analysis and Forecasts, 2023
- 2031
11.1. Overview
11.2. Japan IVD
Quality Control Market Revenue (US$ Mn) and
Forecasts, By End Users
11.2.1. Hospitals
and Clinics
11.2.1.1. Definition
11.2.1.2. Market Estimation and Penetration, 2015 - 2022
11.2.1.3. Market Forecast, 2023 - 2031
11.2.1.4.
Compound Annual Growth
Rate (CAGR)
11.2.1.5.
Regional Bifurcation
11.2.1.5.1. Hokkaido
11.2.1.5.1.1.
Market Estimation, 2015 – 2022
11.2.1.5.1.2.
Market Forecast, 2023 – 2031
11.2.1.5.2. Tohoku
11.2.1.5.2.1.
Market Estimation, 2015 – 2022
11.2.1.5.2.2.
Market Forecast, 2023 – 2031
11.2.1.5.3. Kanto
11.2.1.5.3.1.
Market Estimation, 2015 – 2022
11.2.1.5.3.2.
Market Forecast, 2023 – 2031
11.2.1.5.4. Chubu
11.2.1.5.4.1.
Market Estimation, 2015 – 2022
11.2.1.5.4.2.
Market Forecast, 2023 – 2031
11.2.1.5.5. Kinki/Kansai
11.2.1.5.5.1.
Market Estimation, 2015 – 2022
11.2.1.5.5.2.
Market Forecast, 2023 – 2031
11.2.1.5.6. Chugoku
11.2.1.5.6.1.
Market Estimation, 2015 – 2022
11.2.1.5.6.2.
Market Forecast, 2023 – 2031
11.2.1.5.7. Shikoku
11.2.1.5.7.1.
Market Estimation, 2015 – 2022
11.2.1.5.7.2.
Market Forecast, 2023 – 2031
11.2.1.5.8. Kyushu
(incl. Okinawa)
11.2.1.5.8.1.
Market Estimation, 2015 – 2022
11.2.1.5.8.2.
Market Forecast, 2023 – 2031
11.2.2. Laboratories
11.2.2.1. Definition
11.2.2.2. Market Estimation and Penetration, 2015 - 2022
11.2.2.3. Market Forecast, 2023 - 2031
11.2.2.4.
Compound Annual Growth
Rate (CAGR)
11.2.2.5.
Regional Bifurcation
11.2.2.5.1. Hokkaido
11.2.2.5.1.1.
Market Estimation, 2015 – 2022
11.2.2.5.1.2.
Market Forecast, 2023 – 2031
11.2.2.5.2. Tohoku
11.2.2.5.2.1.
Market Estimation, 2015 – 2022
11.2.2.5.2.2.
Market Forecast, 2023 – 2031
11.2.2.5.3. Kanto
11.2.2.5.3.1.
Market Estimation, 2015 – 2022
11.2.2.5.3.2.
Market Forecast, 2023 – 2031
11.2.2.5.4. Chubu
11.2.2.5.4.1.
Market Estimation, 2015 – 2022
11.2.2.5.4.2.
Market Forecast, 2023 – 2031
11.2.2.5.5. Kinki/Kansai
11.2.2.5.5.1.
Market Estimation, 2015 – 2022
11.2.2.5.5.2.
Market Forecast, 2023 – 2031
11.2.2.5.6. Chugoku
11.2.2.5.6.1.
Market Estimation, 2015 – 2022
11.2.2.5.6.2.
Market Forecast, 2023 – 2031
11.2.2.5.7. Shikoku
11.2.2.5.7.1.
Market Estimation, 2015 – 2022
11.2.2.5.7.2.
Market Forecast, 2023 – 2031
11.2.2.5.8. Kyushu
(incl. Okinawa)
11.2.2.5.8.1.
Market Estimation, 2015 – 2022
11.2.2.5.8.2.
Market Forecast, 2023 – 2031
11.2.3. Research
Institutes
11.2.3.1. Definition
11.2.3.2. Market Estimation and Penetration, 2015 - 2022
11.2.3.3. Market Forecast, 2023 - 2031
11.2.3.4.
Compound Annual Growth
Rate (CAGR)
11.2.3.5.
Regional Bifurcation
11.2.3.5.1. Hokkaido
11.2.3.5.1.1.
Market Estimation, 2015 – 2022
11.2.3.5.1.2.
Market Forecast, 2023 – 2031
11.2.3.5.2. Tohoku
11.2.3.5.2.1.
Market Estimation, 2015 – 2022
11.2.3.5.2.2.
Market Forecast, 2023 – 2031
11.2.3.5.3. Kanto
11.2.3.5.3.1.
Market Estimation, 2015 – 2022
11.2.3.5.3.2.
Market Forecast, 2023 – 2031
11.2.3.5.4. Chubu
11.2.3.5.4.1.
Market Estimation, 2015 – 2022
11.2.3.5.4.2.
Market Forecast, 2023 – 2031
11.2.3.5.5. Kinki/Kansai
11.2.3.5.5.1.
Market Estimation, 2015 – 2022
11.2.3.5.5.2.
Market Forecast, 2023 – 2031
11.2.3.5.6. Chugoku
11.2.3.5.6.1.
Market Estimation, 2015 – 2022
11.2.3.5.6.2.
Market Forecast, 2023 – 2031
11.2.3.5.7. Shikoku
11.2.3.5.7.1.
Market Estimation, 2015 – 2022
11.2.3.5.7.2.
Market Forecast, 2023 – 2031
11.2.3.5.8. Kyushu
(incl. Okinawa)
11.2.3.5.8.1.
Market Estimation, 2015 – 2022
11.2.3.5.8.2.
Market Forecast, 2023 – 2031
11.2.4. Others
11.2.4.1. Definition
11.2.4.2. Market Estimation and Penetration, 2015 - 2022
11.2.4.3. Market Forecast, 2023 - 2031
11.2.4.4.
Compound Annual Growth
Rate (CAGR)
11.2.4.5.
Regional Bifurcation
11.2.4.5.1. Hokkaido
11.2.4.5.1.1.
Market Estimation, 2015 – 2022
11.2.4.5.1.2.
Market Forecast, 2023 – 2031
11.2.4.5.2. Tohoku
11.2.4.5.2.1.
Market Estimation, 2015 – 2022
11.2.4.5.2.2.
Market Forecast, 2023 – 2031
11.2.4.5.3. Kanto
11.2.4.5.3.1.
Market Estimation, 2015 – 2022
11.2.4.5.3.2.
Market Forecast, 2023 – 2031
11.2.4.5.4. Chubu
11.2.4.5.4.1.
Market Estimation, 2015 – 2022
11.2.4.5.4.2.
Market Forecast, 2023 – 2031
11.2.4.5.5. Kinki/Kansai
11.2.4.5.5.1.
Market Estimation, 2015 – 2022
11.2.4.5.5.2.
Market Forecast, 2023 – 2031
11.2.4.5.6. Chugoku
11.2.4.5.6.1.
Market Estimation, 2015 – 2022
11.2.4.5.6.2.
Market Forecast, 2023 – 2031
11.2.4.5.7. Shikoku
11.2.4.5.7.1.
Market Estimation, 2015 – 2022
11.2.4.5.7.2.
Market Forecast, 2023 – 2031
11.2.4.5.8. Kyushu
(incl. Okinawa)
11.2.4.5.8.1.
Market Estimation, 2015 – 2022
11.2.4.5.8.2.
Market Forecast, 2023 – 2031
11.3. Key
Segment for Channeling Investments
11.3.1. By End
Users
12.
Japan IVD Quality Control
Market Analysis and Forecasts, 2023 - 2031
12.1. Overview
12.2.
Japan IVD Quality Control Market Analysis and Forecasts, by Region
12.2.1. Hokkaido
12.2.1.1. Hokkaido IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Offerings
12.2.1.1.1. Quality Control Products
12.2.1.1.1.1. Serum
Based
12.2.1.1.1.2. Whole
Blood
12.2.1.1.1.3. Urine
12.2.1.1.2. Data Management Solutions
12.2.1.1.3. Quality Assurance Services
12.2.1.2. Hokkaido IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Control (Analytes)
12.2.1.2.1. Immunochemistry
12.2.1.2.2. Clinical Chemistry
12.2.1.2.3. Microbiology
12.2.1.2.4. Hematology
12.2.1.2.5. Coagulation
12.2.1.2.6. Infectious Disease
12.2.1.2.7. Diabetics/ Hemoglobin
12.2.1.2.8. Cardiology
12.2.1.2.9. Others
12.2.1.3. Hokkaido IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Manufacturer Type
12.2.1.3.1. Dependent/first-party controls
12.2.1.3.2. Independent/third party controls
12.2.1.4. Hokkaido IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By End Users
12.2.1.4.1. Hospitals and Clinics
12.2.1.4.2. Laboratories
12.2.1.4.3. Research Institutes
12.2.1.4.4.
Others
12.2.2. Tohoku
12.2.2.1. Tohoku IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Offerings
12.2.2.1.1. Quality Control Products
12.2.2.1.1.1. Serum
Based
12.2.2.1.1.2. Whole
Blood
12.2.2.1.1.3. Urine
12.2.2.1.2. Data Management Solutions
12.2.2.1.3. Quality Assurance Services
12.2.2.2. Tohoku IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Control (Analytes)
12.2.2.2.1. Immunochemistry
12.2.2.2.2. Clinical Chemistry
12.2.2.2.3. Microbiology
12.2.2.2.4. Hematology
12.2.2.2.5. Coagulation
12.2.2.2.6. Infectious Disease
12.2.2.2.7. Diabetics/ Hemoglobin
12.2.2.2.8. Cardiology
12.2.2.2.9. Others
12.2.2.3. Tohoku IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Manufacturer Type
12.2.2.3.1. Dependent/first-party controls
12.2.2.3.2. Independent/third party controls
12.2.2.4. Tohoku IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By End Users
12.2.2.4.1. Hospitals and Clinics
12.2.2.4.2. Laboratories
12.2.2.4.3. Research Institutes
12.2.2.4.4.
Others
12.2.3. Kanto
12.2.3.1. Kanto IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Offerings
12.2.3.1.1. Quality Control Products
12.2.3.1.1.1. Serum
Based
12.2.3.1.1.2. Whole
Blood
12.2.3.1.1.3. Urine
12.2.3.1.2. Data Management Solutions
12.2.3.1.3. Quality Assurance Services
12.2.3.2. Kanto IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Control (Analytes)
12.2.3.2.1. Immunochemistry
12.2.3.2.2. Clinical Chemistry
12.2.3.2.3. Microbiology
12.2.3.2.4. Hematology
12.2.3.2.5. Coagulation
12.2.3.2.6. Infectious Disease
12.2.3.2.7. Diabetics/ Hemoglobin
12.2.3.2.8. Cardiology
12.2.3.2.9. Others
12.2.3.3. Kanto IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Manufacturer Type
12.2.3.3.1. Dependent/first-party controls
12.2.3.3.2. Independent/third party controls
12.2.3.4. Kanto IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By End Users
12.2.3.4.1. Hospitals and Clinics
12.2.3.4.2. Laboratories
12.2.3.4.3. Research Institutes
12.2.3.4.4.
Others
12.2.4. Chubu
12.2.4.1. Chubu IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Offerings
12.2.4.1.1. Quality Control Products
12.2.4.1.1.1. Serum
Based
12.2.4.1.1.2. Whole
Blood
12.2.4.1.1.3. Urine
12.2.4.1.2. Data Management Solutions
12.2.4.1.3. Quality Assurance Services
12.2.4.2. Chubu IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Control (Analytes)
12.2.4.2.1. Immunochemistry
12.2.4.2.2. Clinical Chemistry
12.2.4.2.3. Microbiology
12.2.4.2.4. Hematology
12.2.4.2.5. Coagulation
12.2.4.2.6. Infectious Disease
12.2.4.2.7. Diabetics/ Hemoglobin
12.2.4.2.8. Cardiology
12.2.4.2.9. Others
12.2.4.3. Chubu IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Manufacturer Type
12.2.4.3.1. Dependent/first-party controls
12.2.4.3.2. Independent/third party controls
12.2.4.4. Chubu IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By End Users
12.2.4.4.1. Hospitals and Clinics
12.2.4.4.2. Laboratories
12.2.4.4.3. Research Institutes
12.2.4.4.4.
Others
12.2.5. Kinki/Kansai
12.2.5.1. Kinki/Kansai IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Offerings
12.2.5.1.1. Quality Control Products
12.2.5.1.1.1. Serum
Based
12.2.5.1.1.2. Whole
Blood
12.2.5.1.1.3. Urine
12.2.5.1.2. Data Management Solutions
12.2.5.1.3. Quality Assurance Services
12.2.5.2. Kinki/Kansai IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Control (Analytes)
12.2.5.2.1. Immunochemistry
12.2.5.2.2. Clinical Chemistry
12.2.5.2.3. Microbiology
12.2.5.2.4. Hematology
12.2.5.2.5. Coagulation
12.2.5.2.6. Infectious Disease
12.2.5.2.7. Diabetics/ Hemoglobin
12.2.5.2.8. Cardiology
12.2.5.2.9. Others
12.2.5.3. Kinki/Kansai IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Manufacturer Type
12.2.5.3.1. Dependent/first-party controls
12.2.5.3.2. Independent/third party controls
12.2.5.4. Kinki/Kansai IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By End Users
12.2.5.4.1. Hospitals and Clinics
12.2.5.4.2. Laboratories
12.2.5.4.3. Research Institutes
12.2.5.4.4.
Others
12.2.6. Chugoku
12.2.6.1. Chugoku IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Offerings
12.2.6.1.1. Quality Control Products
12.2.6.1.1.1. Serum
Based
12.2.6.1.1.2. Whole
Blood
12.2.6.1.1.3. Urine
12.2.6.1.2. Data Management Solutions
12.2.6.1.3. Quality Assurance Services
12.2.6.2. Chugoku IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Control (Analytes)
12.2.6.2.1. Immunochemistry
12.2.6.2.2. Clinical Chemistry
12.2.6.2.3. Microbiology
12.2.6.2.4. Hematology
12.2.6.2.5. Coagulation
12.2.6.2.6. Infectious Disease
12.2.6.2.7. Diabetics/ Hemoglobin
12.2.6.2.8. Cardiology
12.2.6.2.9. Others
12.2.6.3. Chugoku IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Manufacturer Type
12.2.6.3.1. Dependent/first-party controls
12.2.6.3.2. Independent/third party controls
12.2.6.4. Chugoku IVD Quality Control Market Revenue (US$ Mn) and Forecasts,
By End Users
12.2.6.4.1. Hospitals and Clinics
12.2.6.4.2. Laboratories
12.2.6.4.3. Research Institutes
12.2.6.4.4.
Others
12.2.7. Shikoku
12.2.7.1. Shikoku IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Offerings
12.2.7.1.1. Quality Control Products
12.2.7.1.1.1. Serum
Based
12.2.7.1.1.2. Whole
Blood
12.2.7.1.1.3. Urine
12.2.7.1.2. Data Management Solutions
12.2.7.1.3. Quality Assurance Services
12.2.7.2. Shikoku IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Control (Analytes)
12.2.7.2.1. Immunochemistry
12.2.7.2.2. Clinical Chemistry
12.2.7.2.3. Microbiology
12.2.7.2.4. Hematology
12.2.7.2.5. Coagulation
12.2.7.2.6. Infectious Disease
12.2.7.2.7. Diabetics/ Hemoglobin
12.2.7.2.8. Cardiology
12.2.7.2.9. Others
12.2.7.3. Shikoku IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By Manufacturer Type
12.2.7.3.1. Dependent/first-party controls
12.2.7.3.2. Independent/third party controls
12.2.7.4. Shikoku IVD Quality Control Market Revenue (US$ Mn) and
Forecasts, By End Users
12.2.7.4.1. Hospitals and Clinics
12.2.7.4.2. Laboratories
12.2.7.4.3. Research Institutes
12.2.7.4.4.
Others
12.2.8. Kyushu (incl. Okinawa)
12.2.8.1. Kyushu (incl. Okinawa) IVD Quality Control Market Revenue (US$
Mn) and Forecasts, By Offerings
12.2.8.1.1. Quality Control Products
12.2.8.1.1.1. Serum
Based
12.2.8.1.1.2. Whole
Blood
12.2.8.1.1.3. Urine
12.2.8.1.2. Data Management Solutions
12.2.8.1.3. Quality Assurance Services
12.2.8.2. Kyushu (incl. Okinawa) IVD Quality Control Market Revenue (US$
Mn) and Forecasts, By Control (Analytes)
12.2.8.2.1. Immunochemistry
12.2.8.2.2. Clinical Chemistry
12.2.8.2.3. Microbiology
12.2.8.2.4. Hematology
12.2.8.2.5. Coagulation
12.2.8.2.6. Infectious Disease
12.2.8.2.7. Diabetics/ Hemoglobin
12.2.8.2.8. Cardiology
12.2.8.2.9. Others
12.2.8.3. Kyushu (incl. Okinawa) IVD Quality Control Market Revenue (US$
Mn) and Forecasts, By Manufacturer Type
12.2.8.3.1. Dependent/first-party controls
12.2.8.3.2. Independent/third party controls
12.2.8.4. Kyushu (incl. Okinawa) IVD Quality Control Market Revenue (US$
Mn) and Forecasts, By End Users
12.2.8.4.1. Hospitals and Clinics
12.2.8.4.2. Laboratories
12.2.8.4.3. Research Institutes
12.2.8.4.4.
Others
12.3. Key
Segment for Channeling Investments
12.3.1. By Region
13. Competitive Benchmarking
13.1. Market
Share Analysis, 2022
13.2. Japan
Presence and Growth Strategies
13.2.1. Mergers
and Acquisitions
13.2.2. Product
Launches
13.2.3. Investments
Trends
13.2.4. R&D
Initiatives
14. Player Profiles
14.1. Abbott Diagnostics
14.1.1. Company
Details
14.1.2. Company
Overview
14.1.3. Product
Offerings
14.1.4. Key
Developments
14.1.5. Financial
Analysis
14.1.6. SWOT
Analysis
14.1.7. Business
Strategies
14.2. Beckman Coulter, Inc.
14.2.1. Company
Details
14.2.2. Company
Overview
14.2.3. Product
Offerings
14.2.4. Key
Developments
14.2.5. Financial
Analysis
14.2.6. SWOT
Analysis
14.2.7. Business
Strategies
14.3. Bio-Rad Laboratories, Inc.
14.3.1. Company
Details
14.3.2. Company
Overview
14.3.3. Product
Offerings
14.3.4. Key
Developments
14.3.5. Financial
Analysis
14.3.6. SWOT
Analysis
14.3.7. Business
Strategies
14.4. F. Hoffmann-La
Roche Ltd
14.4.1. Company
Details
14.4.2. Company
Overview
14.4.3. Product
Offerings
14.4.4. Key
Developments
14.4.5. Financial
Analysis
14.4.6. SWOT
Analysis
14.4.7. Business
Strategies
14.5. Fortress Diagnostics
14.5.1. Company
Details
14.5.2. Company
Overview
14.5.3. Product
Offerings
14.5.4. Key
Developments
14.5.5. Financial
Analysis
14.5.6. SWOT
Analysis
14.5.7. Business
Strategies
14.6. Fujirebio Inc.
14.6.1. Company
Details
14.6.2. Company
Overview
14.6.3. Product
Offerings
14.6.4. Key
Developments
14.6.5. Financial
Analysis
14.6.6. SWOT
Analysis
14.6.7. Business
Strategies
14.7. Horiba Medical Japan Co., Ltd.
14.7.1. Company
Details
14.7.2. Company
Overview
14.7.3. Product
Offerings
14.7.4. Key
Developments
14.7.5. Financial
Analysis
14.7.6. SWOT
Analysis
14.7.7. Business
Strategies
14.8. Randox Laboratories
14.8.1. Company
Details
14.8.2. Company
Overview
14.8.3. Product
Offerings
14.8.4. Key
Developments
14.8.5. Financial
Analysis
14.8.6. SWOT
Analysis
14.8.7. Business
Strategies
14.9. Sekisui Medical Co., Ltd.
14.9.1. Company
Details
14.9.2. Company
Overview
14.9.3. Product
Offerings
14.9.4. Key
Developments
14.9.5. Financial
Analysis
14.9.6. SWOT
Analysis
14.9.7. Business
Strategies
14.10. Sysmex Corporation
14.10.1. Company
Details
14.10.2. Company
Overview
14.10.3. Product
Offerings
14.10.4. Key
Developments
14.10.5. Financial
Analysis
14.10.6. SWOT
Analysis
14.10.7. Business
Strategies
14.11. Thermo Fisher Scientific Inc.
14.11.1. Company
Details
14.11.2. Company
Overview
14.11.3. Product
Offerings
14.11.4. Key
Developments
14.11.5. Financial
Analysis
14.11.6. SWOT
Analysis
14.11.7. Business
Strategies
14.12. Other
Market Participants
15. Key Findings
Note: This ToC is tentative
and can be changed according to the research study conducted during the course
of report completion.
**Exclusive for Multi-User
and Enterprise User.
At Absolute Markets Insights, we are engaged in building both global as well as country specific reports. As a result, the approach taken for deriving the estimation and forecast for a specific country is a bit unique and different in comparison to the global research studies. In this case, we not only study the concerned market factors & trends prevailing in a particular country (from secondary research) but we also tend to calculate the actual market size & forecast from the revenue generated from the market participants involved in manufacturing or distributing the any concerned product. These companies can also be service providers. For analyzing any country specifically, we do consider the growth factors prevailing under the states/cities/county for the same. For instance, if we are analyzing an industry specific to United States, we primarily need to study about the states present under the same(where the product/service has the highest growth). Similar analysis will be followed by other countries. Our scope of the report changes with different markets.
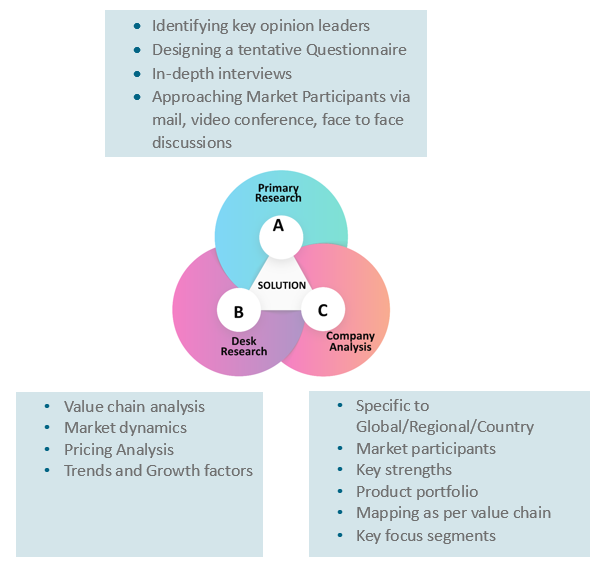
Our research study is mainly implement through a mix of both secondary and primary research. Various sources such as industry magazines, trade journals, and government websites and trade associations are reviewed for gathering precise data. Primary interviews are conducted to validate the market size derived from secondary research. Industry experts, major manufacturers and distributors are contacted for further validation purpose on the current market penetration and growth trends.
Prominent participants in our primary research process include:
- Key Opinion Leaders namely the CEOs, CSOs, VPs, purchasing managers, amongst others
- Research and development participants, distributors/suppliers and subject matter experts
Secondary Research includes data extracted from paid data sources:
- Reuters
- Factiva
- Bloomberg
- One Source
- Hoovers
Research Methodology
Key Inclusions

Reach to us
Call us on
+91-74002-42424
Drop us an email at
sales@absolutemarketsinsights.com
Why Absolute Markets Insights?
An effective strategy is the entity that influences a business to stand out of the crowd. An organization with a phenomenal strategy for success dependably has the edge over the rivals in the market. It offers the organizations a head start in planning their strategy. Absolute Market Insights is the new initiation in the industry that will furnish you with the lead your business needs. Absolute Market Insights is the best destination for your business intelligence and analytical solutions; essentially because our qualitative and quantitative sources of information are competent to give one-stop solutions. We inventively combine qualitative and quantitative research in accurate proportions to have the best report, which not only gives the most recent insights but also assists you to grow.

