Oncology Drugs Market By Indication (Breast Cancer, Prostate Cancer, Basal Cell Cancer, Skin Cancer (Non-Melanoma), Colorectal Cancer, Lung Cancer, Renal Cancer, Bladder Cancer, Lymphoma and Others), By Treatment (Drug Therapies (Antimetabolites, Antitumor Antibiotics, Asparagine-Specific Enzymes, Bisphosphonates and Biosimilars, DNA-Damaging Agents (Antineoplastics) and Alkylating Agents, Inhibitors(DNA-Repair Enzyme Inhibitors, Histone Deacetylase Inhibitors, Janus-Associated Kinase (JAK) Inhibitors, Proteasome Inhibitors, Tyrosine Kinase Inhibitors, PD-1 and PD-L1 Inhibitors and Others)), Surgery (Open Surgery and Minimally Invasive Surgery), Radiation Therapy, Chemotherapy, Immunotherapy (Chimeric Antigen Receptor (CAR) T-Cell Therapy, Cytokine Treatment, Donor Lymphocyte Infusion, Monoclonal Antibody Treatment and Others), Targeted Therapy, Hormone Therapy, Stem Cell Transplant (Autologous Stem Cell Transplantation, Allogeneic Stem Cell Transplantation, Reduced-Intensity Allogeneic Stem Cell Transplantation, Graft-Versus-Host Disease), Precision Medicine and Others), By Region (U.S., Canada, Mexico, Rest of North America, France, UK, Germany, Spain, Italy, Nordic Countries, Benelux Union, Rest of Europe, China, Japan, India, New Zealand, Australia, South Korea, Southeast Asia, Rest of Asia Pacific, Saudi Arabia, UAE, Egypt, Kuwait, South Africa, Rest of MEA, Brazil, Argentina, Rest of Latin America) – Global Insights, Growth, Size, Comparative Analysis, Trends and Forecast, 2019 – 2027
Industry Trends
Global oncology drugs market was estimated to be US$ 119 billion in 2018 is expected to reach US$ 350.01 billion by 2027, growing at an estimated CAGR of 12.7% over the forecast period. The geriatric population in the United States possess certain behaviors such as smoking, not being physically active and eating an unhealthy diet. Rising number of the same has aided the overall cancer drugs market. Development of new anti-cancer molecules coupled with favorable government initiatives has assisted the overall growth of the industry.
Amongst the drug type, the usage of immune-oncology PD-1 and PD-L1 inhibitors have recently witnessed a rapid growth owing to its remarkable clinical profile and their approval for various cancer types. The same is having a larger penetration as a new treatment option amongst patient suffering from metastatic melanoma. There has been an increased focus on personalized medicine that is in turn, leading to patient segmentation based biomarker status. There were 14 New Active Substance cancer therapeutics that was launched in 2017. They solely catered to targeted therapies. Out of which, 11 therapies got their approval from FDA. However, adverse effects associated with cancer drugs and high cost of the cancer drugs are some of factors that is anticipated to critically impact the global oncology drugs market.
Breast cancer has the highest share in 2018 and is expected to continue the same trend over the next eight years. One of the product named Neulasta, offered by Amgen, Inc. is being used by women with stage 4 breast cancer. The drug reported to reduce the risk of febrile neutropenia during chemotherapy by 97% compared to a placebo. It has been duly reported that in most cases adjuvant treat .i.e., chemotherapy with combination of drugs is considered a more favorable option compared to single combination. Some of the adjuvant drugs that are used are anthracyclines, taxanes, 5-fluorouracil, cyclophosphamide and carboplatin are the most demanded drug of Oncology Drugs Market.
In terms of revenue, stem cell transplants is expected to witness a considerable CAGR over the forecast period. This procedure restores blood-forming stem cells in people who have had their destroyed in chemotherapy or radiation therapy to treat cancers. There are two main types of stem cell transplants which are autologous and allogeneic. This treatment primarily help people who suffer from lymphoma and leukemia. They may be also used for multiple myeloma and neuroblastoma.
North America region held more than 45% of the overall Oncology Drugs Market and is anticipated to continue the same trend over the forecast period. In terms of penetration, the European Union hold the second position. The highest number of agents which are newly launched cancer drugs from 2011 to 2015 period are available in the United States and Germany. The reimbursement process differs by geography and the reimbursement ranges from 100% to 61% across countries. Also, owing to advances in cancer treatment, the mortality rates has steadily declined across the major developed countries in the recent past. This decline was the highest in France, followed by the United States and Japan. The improvement in the mortality rates is also due to factors such as early diagnosis, rise in screening services along with the introduction of new drugs (agents) that incorporates the diverse action mechanism.
A drug named Avelumab, approved in March 2017 is introduced for metastatic Merkel cell carcinoma which is a rare aggressive skin cancer type. Other therapies such as MEK inhibitors, BRAF inhibitors and anti-CTLA4 agents for melanoma application have been launched in the recent past.
Thus, the introduction of new drugs along with therapies associated with cancer can diminish the prevalence of the disease in various regions. Also, preventive measures are being increasingly taken by people wherein, they adopt healthy lifestyle and diet. Such factors are expected to reduce the incidence of these diseases in future years.
Global, Oncology Drugs Market Revenue & Forecast, (US$ Million), 2015 – 2027
Competitive Landscape
The report provides both, qualitative and quantitative research of Oncology Drugs Market, as well as provides comprehensive insights and favored development methods adopted by the key contenders. The report also offers extensive research on the key players in this market and details on the competitiveness of these players. Key business strategies such as mergers and acquisitions (M&A), affiliations, collaborations, and contracts adopted by these major market participants are also recognized and analyzed in the report. For each company, the report studies their global presence, competitors, service offerings, specification, pricing, and gross margin.
Some of the players operating in the oncology drugs market are F. Hoffmann-La Roche Ltd, Johnson & Johnson Services, Inc., Eli Lilly and Company, Novartis AG, Pfizer Inc., Bayer AG, AstraZeneca, Merck & Co., Inc.,CELGENE CORPORATION and Polaris Pharmaceuticals amongst others
- By Indication
- Breast Cancer
- Prostate Cancer
- Basal Cell Cancer
- Skin Cancer (Non-Melanoma)
- Colorectal cancer
- Lung Cancer
- Renal Cancer
- Bladder Cancer
- Lymphoma
- Others
- By Treatment
- Drug Therapies
- Antimetabolites
- Antitumor Antibiotics
- Asparagine-Specific Enzymes
- Bisphosphonates & Biosimilars
- DNA-Damaging Agents (Antineoplastics) and Alkylating Agents
- Inhibitors
- DNA-Repair Enzyme Inhibitors
- Histone Deacetylase Inhibitors
- Janus-Associated Kinase (JAK) Inhibitors
- Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
- Proteasome Inhibitors
- Tyrosine Kinase Inhibitors
- PD-1 and PD-L1 inhibitors
- Others
- Monoclonal Antibodies
- Others
- Surgery
- Open Surgery
- Minimally Invasive Surgery
- Radiation Therapy
- Chemotherapy
- Immunotherapy
- Chimeric antigen receptor (CAR) T-cell therapy
- Cytokine Treatment
- Donor lymphocyte infusion
- Monoclonal Antibody Treatment
- Others
- Targeted Therapy
- Hormone Therapy
- Stem Cell Transplant
- Autologous stem cell transplantation
- Allogeneic stem cell transplantation
- Reduced-Intensity Allogeneic Stem Cell Transplantation
- Graft-Versus-Host Disease
- Precision Medicine
- Others
- Drug Therapies
- By Geography
- North America
- U.S.
- Canada
- Mexico
- Rest of North America
- Europe
- France
- The UK
- Spain
- Germany
- Italy
- Nordic Countries
- Denmark
- Finland
- Iceland
- Norway
- Sweden
- Benelux Union
- Belgium
- The Netherlands
- Luxemburg
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- New Zealand
- Australia
- South Korea
- Southeast Asia
- Indonesia
- Thailand
- Malaysia
- Singapore
- Rest of Southeast Asia
- Rest of Asia Pacific
- Middle East & Africa
- Saudi Arabia
- UAE
- Egypt
- Kuwait
- South Africa
- Rest of Middle East & Africa
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- North America
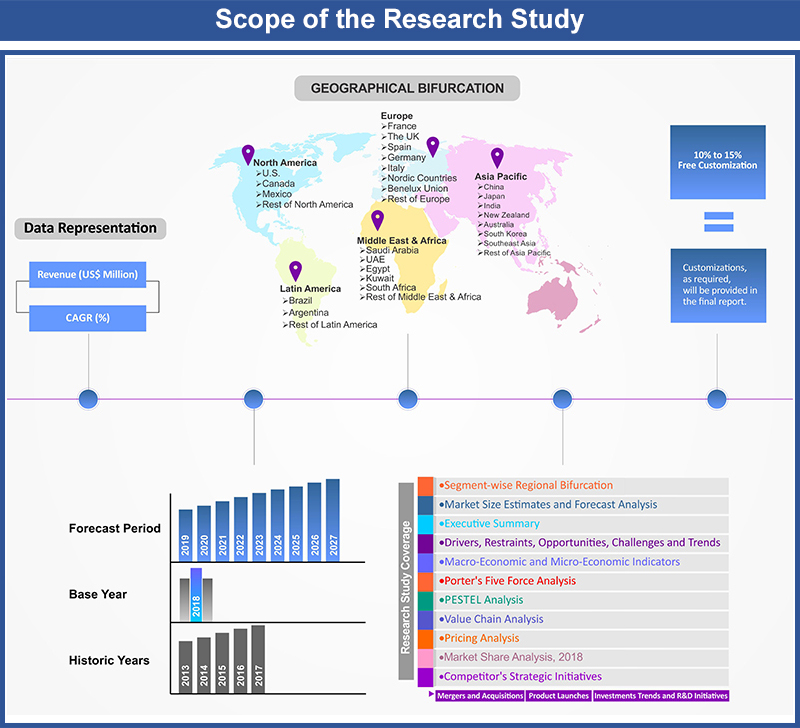
1. Market Scope
1.1. Market
Segmentation
1.2. Years
Considered
1.2.1. Historic
Years: 2013 - 2017
1.2.2. Base
Year: 2018
1.2.3. Forecast
Years: 2019 – 2027
2. Key Target Audiences
3. Research Methodology
3.1. Primary
Research
3.1.1. Research
Questionnaire
3.1.2. Global
Percentage Breakdown
3.1.3. Primary
Interviews: Key Opinion Leaders (KOLs)
3.2. Secondary
Research
3.2.1. Paid
Databases
3.2.2. Secondary
Sources
3.3. Market
Size Estimates
3.3.1. Top-Down
Approach
3.3.2. Bottom-Up
Approach
3.4. Data
Triangulation Methodology
3.5. Research
Assumptions
4. Recommendations and Insights from AMI’s Perspective**
5. Holistic Overview of Oncology Drugs Market
6. Market Synopsis:
Oncology Drugs Market
7. Oncology Drugs Market Analysis: Qualitative Perspective
7.1. Introduction
7.1.1. Product
Definition
7.1.2. Industry
Development
7.2. Market
Dynamics
7.2.1. Drivers
7.2.2. Restraints
7.2.3. Opportunities
7.2.4. Challenges
7.3. Trends in
Oncology Drugs Market
7.4. Market
Determinants Radar Chart
7.5. Macro-Economic
and Micro-Economic Indicators: Oncology Drugs Market
7.6. Porter’s
Five Force Analysis
8. Global Oncology Drugs Market Analysis and Forecasts, 2019 –
2027
8.1. Overview
8.1.1. Global
Oncology Drugs Market Revenue (US$ Mn)
8.2. Global
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
8.2.1. Breast
Cancer
8.2.1.1. Definition
8.2.1.2. Market
Penetration, 2018
8.2.1.3. Market
Estimation, 2013 – 2018
8.2.1.4. Market
Forecast, 2019 – 2027
8.2.1.5. Compound
Annual Growth Rate (CAGR)
8.2.1.6. Regional
Bifurcation
8.2.1.6.1. North
America
8.2.1.6.1.1. Market
Estimation, 2013 – 2018
8.2.1.6.1.2. Market
Forecast, 2019 – 2027
8.2.1.6.2. Europe
8.2.1.6.2.1. Market
Estimation, 2013 – 2018
8.2.1.6.2.2. Market
Forecast, 2019 – 2027
8.2.1.6.3. Asia
Pacific
8.2.1.6.3.1. Market
Estimation, 2013 – 2018
8.2.1.6.3.2. Market
Forecast, 2019 – 2027
8.2.1.6.4. Middle
East and Africa
8.2.1.6.4.1. Market
Estimation, 2013 – 2018
8.2.1.6.4.2. Market
Forecast, 2019 – 2027
8.2.1.6.5. Latin
America
8.2.1.6.5.1. Market
Estimation, 2013 – 2018
8.2.1.6.5.2. Market
Forecast, 2019 – 2027
8.2.2. Prostate
Cancer
8.2.2.1. Definition
8.2.2.2. Market
Penetration, 2018
8.2.2.3. Market
Estimation, 2013 – 2018
8.2.2.4. Market
Forecast, 2019 – 2027
8.2.2.5. Compound
Annual Growth Rate (CAGR)
8.2.2.6. Regional
Bifurcation
8.2.2.6.1. North
America
8.2.2.6.1.1. Market
Estimation, 2013 – 2018
8.2.2.6.1.2. Market
Forecast, 2019 – 2027
8.2.2.6.2. Europe
8.2.2.6.2.1. Market
Estimation, 2013 – 2018
8.2.2.6.2.2. Market
Forecast, 2019 – 2027
8.2.2.6.3. Asia
Pacific
8.2.2.6.3.1. Market
Estimation, 2013 – 2018
8.2.2.6.3.2. Market
Forecast, 2019 – 2027
8.2.2.6.4. Middle
East and Africa
8.2.2.6.4.1. Market
Estimation, 2013 – 2018
8.2.2.6.4.2. Market
Forecast, 2019 – 2027
8.2.2.6.5. Latin
America
8.2.2.6.5.1. Market
Estimation, 2013 – 2018
8.2.2.6.5.2. Market
Forecast, 2019 – 2027
8.2.3. Basal
Cell Cancer
8.2.3.1. Definition
8.2.3.2. Market
Penetration, 2018
8.2.3.3. Market
Estimation, 2013 – 2018
8.2.3.4. Market
Forecast, 2019 – 2027
8.2.3.5. Compound
Annual Growth Rate (CAGR)
8.2.3.6. Regional
Bifurcation
8.2.3.6.1. North
America
8.2.3.6.1.1. Market
Estimation, 2013 – 2018
8.2.3.6.1.2. Market
Forecast, 2019 – 2027
8.2.3.6.2. Europe
8.2.3.6.2.1. Market
Estimation, 2013 – 2018
8.2.3.6.2.2. Market
Forecast, 2019 – 2027
8.2.3.6.3. Asia
Pacific
8.2.3.6.3.1. Market
Estimation, 2013 – 2018
8.2.3.6.3.2. Market
Forecast, 2019 – 2027
8.2.3.6.4. Middle
East and Africa
8.2.3.6.4.1. Market
Estimation, 2013 – 2018
8.2.3.6.4.2. Market
Forecast, 2019 – 2027
8.2.3.6.5. Latin
America
8.2.3.6.5.1. Market
Estimation, 2013 – 2018
8.2.3.6.5.2. Market
Forecast, 2019 – 2027
8.2.4. Skin
Cancer (Non-Melanoma)
8.2.4.1. Definition
8.2.4.2. Market
Penetration, 2018
8.2.4.3. Market
Estimation, 2013 – 2018
8.2.4.4. Market
Forecast, 2019 – 2027
8.2.4.5. Compound
Annual Growth Rate (CAGR)
8.2.4.6. Regional
Bifurcation
8.2.4.6.1. North
America
8.2.4.6.1.1. Market
Estimation, 2013 – 2018
8.2.4.6.1.2. Market
Forecast, 2019 – 2027
8.2.4.6.2. Europe
8.2.4.6.2.1. Market
Estimation, 2013 – 2018
8.2.4.6.2.2. Market
Forecast, 2019 – 2027
8.2.4.6.3. Asia
Pacific
8.2.4.6.3.1. Market
Estimation, 2013 – 2018
8.2.4.6.3.2. Market
Forecast, 2019 – 2027
8.2.4.6.4. Middle
East and Africa
8.2.4.6.4.1. Market
Estimation, 2013 – 2018
8.2.4.6.4.2. Market
Forecast, 2019 – 2027
8.2.4.6.5. Latin
America
8.2.4.6.5.1. Market
Estimation, 2013 – 2018
8.2.4.6.5.2. Market
Forecast, 2019 – 2027
8.2.5. Colorectal
cancer
8.2.5.1. Definition
8.2.5.2. Market
Penetration, 2018
8.2.5.3. Market
Estimation, 2013 – 2018
8.2.5.4. Market
Forecast, 2019 – 2027
8.2.5.5. Compound
Annual Growth Rate (CAGR)
8.2.5.6. Regional
Bifurcation
8.2.5.6.1. North
America
8.2.5.6.1.1. Market
Estimation, 2013 – 2018
8.2.5.6.1.2. Market
Forecast, 2019 – 2027
8.2.5.6.2. Europe
8.2.5.6.2.1. Market
Estimation, 2013 – 2018
8.2.5.6.2.2. Market
Forecast, 2019 – 2027
8.2.5.6.3. Asia
Pacific
8.2.5.6.3.1. Market
Estimation, 2013 – 2018
8.2.5.6.3.2. Market
Forecast, 2019 – 2027
8.2.5.6.4. Middle
East and Africa
8.2.5.6.4.1. Market
Estimation, 2013 – 2018
8.2.5.6.4.2. Market
Forecast, 2019 – 2027
8.2.5.6.5. Latin
America
8.2.5.6.5.1. Market
Estimation, 2013 – 2018
8.2.5.6.5.2. Market
Forecast, 2019 – 2027
8.2.6. Lung
Cancer
8.2.6.1. Definition
8.2.6.2. Market
Penetration, 2018
8.2.6.3. Market
Estimation, 2013 – 2018
8.2.6.4. Market
Forecast, 2019 – 2027
8.2.6.5. Compound
Annual Growth Rate (CAGR)
8.2.6.6. Regional
Bifurcation
8.2.6.6.1. North
America
8.2.6.6.1.1. Market
Estimation, 2013 – 2018
8.2.6.6.1.2. Market
Forecast, 2019 – 2027
8.2.6.6.2. Europe
8.2.6.6.2.1. Market
Estimation, 2013 – 2018
8.2.6.6.2.2. Market
Forecast, 2019 – 2027
8.2.6.6.3. Asia
Pacific
8.2.6.6.3.1. Market
Estimation, 2013 – 2018
8.2.6.6.3.2. Market
Forecast, 2019 – 2027
8.2.6.6.4. Middle
East and Africa
8.2.6.6.4.1. Market
Estimation, 2013 – 2018
8.2.6.6.4.2. Market
Forecast, 2019 – 2027
8.2.6.6.5. Latin
America
8.2.6.6.5.1. Market
Estimation, 2013 – 2018
8.2.6.6.5.2. Market
Forecast, 2019 – 2027
8.2.7. Renal
Cancer
8.2.7.1. Definition
8.2.7.2. Market
Penetration, 2018
8.2.7.3. Market
Estimation, 2013 – 2018
8.2.7.4. Market
Forecast, 2019 – 2027
8.2.7.5. Compound
Annual Growth Rate (CAGR)
8.2.7.6. Regional
Bifurcation
8.2.7.6.1. North
America
8.2.7.6.1.1. Market
Estimation, 2013 – 2018
8.2.7.6.1.2. Market
Forecast, 2019 – 2027
8.2.7.6.2. Europe
8.2.7.6.2.1. Market
Estimation, 2013 – 2018
8.2.7.6.2.2. Market
Forecast, 2019 – 2027
8.2.7.6.3. Asia
Pacific
8.2.7.6.3.1. Market
Estimation, 2013 – 2018
8.2.7.6.3.2. Market
Forecast, 2019 – 2027
8.2.7.6.4. Middle
East and Africa
8.2.7.6.4.1. Market
Estimation, 2013 – 2018
8.2.7.6.4.2. Market
Forecast, 2019 – 2027
8.2.7.6.5. Latin
America
8.2.7.6.5.1. Market
Estimation, 2013 – 2018
8.2.7.6.5.2. Market
Forecast, 2019 – 2027
8.2.8. Bladder
Cancer
8.2.8.1. Definition
8.2.8.2. Market
Penetration, 2018
8.2.8.3. Market
Estimation, 2013 – 2018
8.2.8.4. Market
Forecast, 2019 – 2027
8.2.8.5. Compound
Annual Growth Rate (CAGR)
8.2.8.6. Regional
Bifurcation
8.2.8.6.1. North
America
8.2.8.6.1.1. Market
Estimation, 2013 – 2018
8.2.8.6.1.2. Market
Forecast, 2019 – 2027
8.2.8.6.2. Europe
8.2.8.6.2.1. Market
Estimation, 2013 – 2018
8.2.8.6.2.2. Market
Forecast, 2019 – 2027
8.2.8.6.3. Asia
Pacific
8.2.8.6.3.1. Market
Estimation, 2013 – 2018
8.2.8.6.3.2. Market
Forecast, 2019 – 2027
8.2.8.6.4. Middle
East and Africa
8.2.8.6.4.1. Market
Estimation, 2013 – 2018
8.2.8.6.4.2. Market
Forecast, 2019 – 2027
8.2.8.6.5. Latin
America
8.2.8.6.5.1. Market
Estimation, 2013 – 2018
8.2.8.6.5.2. Market
Forecast, 2019 – 2027
8.2.9. Lymphoma
8.2.9.1. Definition
8.2.9.2. Market
Penetration, 2018
8.2.9.3. Market
Estimation, 2013 – 2018
8.2.9.4. Market
Forecast, 2019 – 2027
8.2.9.5. Compound
Annual Growth Rate (CAGR)
8.2.9.6. Regional
Bifurcation
8.2.9.6.1. North
America
8.2.9.6.1.1. Market
Estimation, 2013 – 2018
8.2.9.6.1.2. Market
Forecast, 2019 – 2027
8.2.9.6.2. Europe
8.2.9.6.2.1. Market
Estimation, 2013 – 2018
8.2.9.6.2.2. Market
Forecast, 2019 – 2027
8.2.9.6.3. Asia
Pacific
8.2.9.6.3.1. Market
Estimation, 2013 – 2018
8.2.9.6.3.2. Market
Forecast, 2019 – 2027
8.2.9.6.4. Middle
East and Africa
8.2.9.6.4.1. Market
Estimation, 2013 – 2018
8.2.9.6.4.2. Market
Forecast, 2019 – 2027
8.2.9.6.5. Latin
America
8.2.9.6.5.1. Market
Estimation, 2013 – 2018
8.2.9.6.5.2. Market
Forecast, 2019 – 2027
8.2.10. Others
8.2.10.1. Definition
8.2.10.2. Market
Penetration, 2018
8.2.10.3. Market
Estimation, 2013 – 2018
8.2.10.4. Market
Forecast, 2019 – 2027
8.2.10.5. Compound
Annual Growth Rate (CAGR)
8.2.10.6. Regional
Bifurcation
8.2.10.6.1. North
America
8.2.10.6.1.1. Market
Estimation, 2013 – 2018
8.2.10.6.1.2. Market
Forecast, 2019 – 2027
8.2.10.6.2. Europe
8.2.10.6.2.1. Market
Estimation, 2013 – 2018
8.2.10.6.2.2. Market
Forecast, 2019 – 2027
8.2.10.6.3. Asia
Pacific
8.2.10.6.3.1. Market
Estimation, 2013 – 2018
8.2.10.6.3.2. Market
Forecast, 2019 – 2027
8.2.10.6.4. Middle
East and Africa
8.2.10.6.4.1. Market
Estimation, 2013 – 2018
8.2.10.6.4.2. Market
Forecast, 2019 – 2027
8.2.10.6.5. Latin
America
8.2.10.6.5.1. Market
Estimation, 2013 – 2018
8.2.10.6.5.2. Market
Forecast, 2019 – 2027
8.3. Key
Segment for Channeling Investments
8.3.1. By
Indication
9. Global Oncology Drugs Market Analysis and Forecasts, 2019 –
2027
9.1. Overview
9.2. Global
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
9.2.1. Drug
Therapies (Definition, Market Penetration (2018), Market Estimation (2013 –
2018), Market Forecast (2019 – 2027), Compound Annual Growth Rate (CAGR),
Regional Bifurcation (North America, Europe, Asia Pacific, Middle East and
Africa, Latin America) and Information on Antimetabolites, Antitumor
Antibiotics, Asparagine-Specific Enzymes, Bisphosphonates and Biosimilars,
DNA-Damaging Agents (Antineoplastics) and Alkylating Agents, Inhibitors,
Monoclonal Antibodies, Others)
9.2.1.1. Antimetabolites
9.2.1.2. Antitumor
Antibiotics
9.2.1.3. Asparagine-Specific
Enzymes
9.2.1.4. Bisphosphonates
and Biosimilars
9.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
9.2.1.6. Inhibitors
9.2.1.6.1. DNA-Repair
Enzyme Inhibitors
9.2.1.6.2. Histone
Deacetylase Inhibitors
9.2.1.6.3. Janus-Associated
Kinase (JAK) Inhibitors
9.2.1.6.4. Phosphoinositide
3-kinase inhibitors (PI3K inhibitors)
9.2.1.6.5. Proteasome
Inhibitors
9.2.1.6.6. Tyrosine
Kinase Inhibitors
9.2.1.6.7. PD-1
and PD-L1 inhibitors
9.2.1.6.8. Others
9.2.1.7. Monoclonal
Antibodies
9.2.1.8. Others
9.2.2. Surgery
(Definition, Market Penetration (2018), Market Estimation (2013 – 2018), Market
Forecast (2019 – 2027), Compound Annual Growth Rate (CAGR), Regional
Bifurcation (North America, Europe, Asia Pacific, Middle East and Africa, Latin
America) and Information on Open Surgery, Minimally Invasive Surgery)
9.2.2.1. Open
Surgery
9.2.2.2. Minimally
Invasive Surgery
9.2.3. Radiation
Therapy
9.2.3.1. Definition
9.2.3.2. Market
Penetration, 2018
9.2.3.3. Market
Estimation, 2013 – 2018
9.2.3.4. Market
Forecast, 2019 – 2027
9.2.3.5. Compound
Annual Growth Rate (CAGR)
9.2.3.6. Regional
Bifurcation
9.2.3.6.1. North
America
9.2.3.6.1.1. Market
Estimation, 2013 – 2018
9.2.3.6.1.2. Market
Forecast, 2019 – 2027
9.2.3.6.2. Europe
9.2.3.6.2.1. Market
Estimation, 2013 – 2018
9.2.3.6.2.2. Market
Forecast, 2019 – 2027
9.2.3.6.3. Asia
Pacific
9.2.3.6.3.1. Market
Estimation, 2013 – 2018
9.2.3.6.3.2. Market
Forecast, 2019 – 2027
9.2.3.6.4. Middle
East and Africa
9.2.3.6.4.1. Market
Estimation, 2013 – 2018
9.2.3.6.4.2. Market
Forecast, 2019 – 2027
9.2.3.6.5. Latin
America
9.2.3.6.5.1. Market
Estimation, 2013 – 2018
9.2.3.6.5.2. Market
Forecast, 2019 – 2027
9.2.4. Chemotherapy
9.2.4.1. Definition
9.2.4.2. Market
Penetration, 2018
9.2.4.3. Market
Estimation, 2013 – 2018
9.2.4.4. Market
Forecast, 2019 – 2027
9.2.4.5. Compound
Annual Growth Rate (CAGR)
9.2.4.6. Regional
Bifurcation
9.2.4.6.1. North
America
9.2.4.6.1.1. Market
Estimation, 2013 – 2018
9.2.4.6.1.2. Market
Forecast, 2019 – 2027
9.2.4.6.2. Europe
9.2.4.6.2.1. Market
Estimation, 2013 – 2018
9.2.4.6.2.2. Market
Forecast, 2019 – 2027
9.2.4.6.3. Asia
Pacific
9.2.4.6.3.1. Market
Estimation, 2013 – 2018
9.2.4.6.3.2. Market
Forecast, 2019 – 2027
9.2.4.6.4. Middle
East and Africa
9.2.4.6.4.1. Market
Estimation, 2013 – 2018
9.2.4.6.4.2. Market
Forecast, 2019 – 2027
9.2.4.6.5. Latin
America
9.2.4.6.5.1. Market
Estimation, 2013 – 2018
9.2.4.6.5.2. Market
Forecast, 2019 – 2027
9.2.5. Immunotherapy
(Definition, Market Penetration (2018), Market Estimation (2013 – 2018), Market
Forecast (2019 – 2027), Compound Annual Growth Rate (CAGR), Regional
Bifurcation (North America, Europe, Asia Pacific, Middle East and Africa, Latin
America) and Information on Chimeric antigen receptor (CAR) T-cell therapy,
Cytokine Treatment, Donor lymphocyte infusion, Monoclonal Antibody Treatment,
Others)
9.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
9.2.5.2. Cytokine
Treatment
9.2.5.3. Donor
lymphocyte infusion
9.2.5.4. Monoclonal
Antibody Treatment
9.2.5.5. Others
9.2.6. Targeted
Therapy
9.2.6.1. Definition
9.2.6.2. Market
Penetration, 2018
9.2.6.3. Market
Estimation, 2013 – 2018
9.2.6.4. Market
Forecast, 2019 – 2027
9.2.6.5. Compound
Annual Growth Rate (CAGR)
9.2.6.6. Regional
Bifurcation
9.2.6.6.1. North
America
9.2.6.6.1.1. Market
Estimation, 2013 – 2018
9.2.6.6.1.2. Market
Forecast, 2019 – 2027
9.2.6.6.2. Europe
9.2.6.6.2.1. Market
Estimation, 2013 – 2018
9.2.6.6.2.2. Market
Forecast, 2019 – 2027
9.2.6.6.3. Asia
Pacific
9.2.6.6.3.1. Market
Estimation, 2013 – 2018
9.2.6.6.3.2. Market
Forecast, 2019 – 2027
9.2.6.6.4. Middle
East and Africa
9.2.6.6.4.1. Market
Estimation, 2013 – 2018
9.2.6.6.4.2. Market
Forecast, 2019 – 2027
9.2.6.6.5. Latin
America
9.2.6.6.5.1. Market
Estimation, 2013 – 2018
9.2.6.6.5.2. Market
Forecast, 2019 – 2027
9.2.7. Hormone
Therapy
9.2.7.1. Definition
9.2.7.2. Market
Penetration, 2018
9.2.7.3. Market
Estimation, 2013 – 2018
9.2.7.4. Market
Forecast, 2019 – 2027
9.2.7.5. Compound
Annual Growth Rate (CAGR)
9.2.7.6. Regional
Bifurcation
9.2.7.6.1. North
America
9.2.7.6.1.1. Market
Estimation, 2013 – 2018
9.2.7.6.1.2. Market
Forecast, 2019 – 2027
9.2.7.6.2. Europe
9.2.7.6.2.1. Market
Estimation, 2013 – 2018
9.2.7.6.2.2. Market
Forecast, 2019 – 2027
9.2.7.6.3. Asia
Pacific
9.2.7.6.3.1. Market
Estimation, 2013 – 2018
9.2.7.6.3.2. Market
Forecast, 2019 – 2027
9.2.7.6.4. Middle
East and Africa
9.2.7.6.4.1. Market
Estimation, 2013 – 2018
9.2.7.6.4.2. Market
Forecast, 2019 – 2027
9.2.7.6.5. Latin
America
9.2.7.6.5.1. Market
Estimation, 2013 – 2018
9.2.7.6.5.2. Market
Forecast, 2019 – 2027
9.2.8. Stem
Cell Therapy (Definition, Market Penetration (2018), Market Estimation (2013 –
2018), Market Forecast (2019 – 2027), Compound Annual Growth Rate (CAGR),
Regional Bifurcation (North America, Europe, Asia Pacific, Middle East and
Africa, Latin America) and Information on Autologous stem cell transplantation,
Allogeneic stem cell transplantation, Reduced-Intensity Allogeneic Stem Cell
Transplantation, Graft-Versus-Host Disease)
9.2.8.1. Autologous
stem cell transplantation
9.2.8.2. Allogeneic
stem cell transplantation
9.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
9.2.8.4. Graft-Versus-Host
Disease
9.2.9. Precision
Medicine
9.2.9.1. Definition
9.2.9.2. Market
Penetration, 2018
9.2.9.3. Market
Estimation, 2013 – 2018
9.2.9.4. Market
Forecast, 2019 – 2027
9.2.9.5. Compound
Annual Growth Rate (CAGR)
9.2.9.6. Regional
Bifurcation
9.2.9.6.1. North
America
9.2.9.6.1.1. Market
Estimation, 2013 – 2018
9.2.9.6.1.2. Market
Forecast, 2019 – 2027
9.2.9.6.2. Europe
9.2.9.6.2.1. Market
Estimation, 2013 – 2018
9.2.9.6.2.2. Market
Forecast, 2019 – 2027
9.2.9.6.3. Asia
Pacific
9.2.9.6.3.1. Market
Estimation, 2013 – 2018
9.2.9.6.3.2. Market
Forecast, 2019 – 2027
9.2.9.6.4. Middle
East and Africa
9.2.9.6.4.1. Market
Estimation, 2013 – 2018
9.2.9.6.4.2. Market
Forecast, 2019 – 2027
9.2.9.6.5. Latin
America
9.2.9.6.5.1. Market
Estimation, 2013 – 2018
9.2.9.6.5.2. Market
Forecast, 2019 – 2027
9.2.10. Others
9.2.10.1. Definition
9.2.10.2. Market
Penetration, 2018
9.2.10.3. Market
Estimation, 2013 – 2018
9.2.10.4. Market
Forecast, 2019 – 2027
9.2.10.5. Compound
Annual Growth Rate (CAGR)
9.2.10.6. Regional
Bifurcation
9.2.10.6.1. North
America
9.2.10.6.1.1. Market
Estimation, 2013 – 2018
9.2.10.6.1.2. Market
Forecast, 2019 – 2027
9.2.10.6.2. Europe
9.2.10.6.2.1. Market
Estimation, 2013 – 2018
9.2.10.6.2.2. Market
Forecast, 2019 – 2027
9.2.10.6.3. Asia
Pacific
9.2.10.6.3.1. Market
Estimation, 2013 – 2018
9.2.10.6.3.2. Market
Forecast, 2019 – 2027
9.2.10.6.4. Middle
East and Africa
9.2.10.6.4.1. Market
Estimation, 2013 – 2018
9.2.10.6.4.2. Market
Forecast, 2019 – 2027
9.2.10.6.5. Latin
America
9.2.10.6.5.1. Market
Estimation, 2013 – 2018
9.2.10.6.5.2. Market
Forecast, 2019 – 2027
9.3. Key
Segment for Channeling Investments
9.3.1. By
Treatment
10. North America Oncology Drugs Market Analysis and Forecasts,
2019 - 2027
10.1. Overview
10.1.1. North
America Oncology Drugs Market Revenue (US$ Mn)
10.2. North
America Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
10.2.1. Breast
Cancer
10.2.2. Prostate
Cancer
10.2.3. Basal
Cell Cancer
10.2.4. Skin
Cancer (Non-Melanoma)
10.2.5. Colorectal
cancer
10.2.6. Lung
Cancer
10.2.7. Renal
Cancer
10.2.8. Bladder
Cancer
10.2.9. Lymphoma
10.2.10. Others
10.3. North
America Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
10.3.1. Drug
Therapies
10.3.1.1. Antimetabolites
10.3.1.2. Antitumor
Antibiotics
10.3.1.3. Asparagine-Specific
Enzymes
10.3.1.4. Bisphosphonates
and Biosimilars
10.3.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
10.3.1.6. Inhibitors
10.3.1.6.1. DNA-Repair
Enzyme Inhibitors
10.3.1.6.2. Histone
Deacetylase Inhibitors
10.3.1.6.3. Janus-Associated
Kinase (JAK) Inhibitors
10.3.1.6.4. Phosphoinositide
3-kinase inhibitors (PI3K inhibitors)
10.3.1.6.5. Proteasome
Inhibitors
10.3.1.6.6. Tyrosine
Kinase Inhibitors
10.3.1.6.7. PD-1
and PD-L1 inhibitors
10.3.1.6.8. Others
10.3.1.7. Monoclonal
Antibodies
10.3.1.8. Others
10.3.2. Surgery
10.3.2.1. Open
Surgery
10.3.2.2. Minimally
Invasive Surgery
10.3.3. Radiation
Therapy
10.3.4. Chemotherapy
10.3.5. Immunotherapy
10.3.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
10.3.5.2. Cytokine
Treatment
10.3.5.3. Donor
lymphocyte infusion
10.3.5.4. Monoclonal
Antibody Treatment
10.3.5.5. Others
10.3.6. Targeted
Therapy
10.3.7. Hormone
Therapy
10.3.8. Stem
Cell Therapy
10.3.8.1. Autologous
stem cell transplantation
10.3.8.2. Allogeneic
stem cell transplantation
10.3.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
10.3.8.4. Graft-Versus-Host
Disease
10.3.9. Precision
Medicine
10.3.10. Others
10.4. North
America Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Country
10.4.1. U.S
10.4.1.1. U.S
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
10.4.1.1.1. Breast
Cancer
10.4.1.1.2. Prostate
Cancer
10.4.1.1.3. Basal
Cell Cancer
10.4.1.1.4. Skin
Cancer (Non-Melanoma)
10.4.1.1.5. Colorectal
cancer
10.4.1.1.6. Lung
Cancer
10.4.1.1.7. Renal
Cancer
10.4.1.1.8. Bladder
Cancer
10.4.1.1.9. Lymphoma
10.4.1.1.10. Others
10.4.1.2. U.S
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
10.4.1.2.1. Drug
Therapies
10.4.1.2.1.1. Antimetabolites
10.4.1.2.1.2. Antitumor
Antibiotics
10.4.1.2.1.3. Asparagine-Specific
Enzymes
10.4.1.2.1.4. Bisphosphonates
and Biosimilars
10.4.1.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
10.4.1.2.1.6. Inhibitors
10.4.1.2.1.6.1. DNA-Repair Enzyme Inhibitors
10.4.1.2.1.6.2. Histone Deacetylase Inhibitors
10.4.1.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
10.4.1.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
10.4.1.2.1.6.5. Proteasome Inhibitors
10.4.1.2.1.6.6. Tyrosine Kinase Inhibitors
10.4.1.2.1.6.7. PD-1 and PD-L1 inhibitors
10.4.1.2.1.6.8. Others
10.4.1.2.1.7. Monoclonal
Antibodies
10.4.1.2.1.8. Others
10.4.1.2.2. Surgery
10.4.1.2.2.1. Open
Surgery
10.4.1.2.2.2. Minimally
Invasive Surgery
10.4.1.2.3. Radiation
Therapy
10.4.1.2.4. Chemotherapy
10.4.1.2.5. Immunotherapy
10.4.1.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
10.4.1.2.5.2. Cytokine
Treatment
10.4.1.2.5.3. Donor
lymphocyte infusion
10.4.1.2.5.4. Monoclonal
Antibody Treatment
10.4.1.2.5.5. Others
10.4.1.2.6. Targeted
Therapy
10.4.1.2.7. Hormone
Therapy
10.4.1.2.8. Stem
Cell Therapy
10.4.1.2.8.1. Autologous
stem cell transplantation
10.4.1.2.8.2. Allogeneic
stem cell transplantation
10.4.1.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
10.4.1.2.8.4. Graft-Versus-Host
Disease
10.4.1.2.9. Precision
Medicine
10.4.1.2.10. Others
10.4.2. Canada
10.4.2.1. Canada
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
10.4.2.1.1. Breast
Cancer
10.4.2.1.2. Prostate
Cancer
10.4.2.1.3. Basal
Cell Cancer
10.4.2.1.4. Skin
Cancer (Non-Melanoma)
10.4.2.1.5. Colorectal
cancer
10.4.2.1.6. Lung
Cancer
10.4.2.1.7. Renal
Cancer
10.4.2.1.8. Bladder
Cancer
10.4.2.1.9. Lymphoma
10.4.2.1.10. Others
10.4.2.2. Canada
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
10.4.2.2.1. Drug
Therapies
10.4.2.2.1.1. Antimetabolites
10.4.2.2.1.2. Antitumor
Antibiotics
10.4.2.2.1.3. Asparagine-Specific
Enzymes
10.4.2.2.1.4. Bisphosphonates
and Biosimilars
10.4.2.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
10.4.2.2.1.6. Inhibitors
10.4.2.2.1.6.1. DNA-Repair Enzyme Inhibitors
10.4.2.2.1.6.2. Histone Deacetylase Inhibitors
10.4.2.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
10.4.2.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
10.4.2.2.1.6.5. Proteasome Inhibitors
10.4.2.2.1.6.6. Tyrosine Kinase Inhibitors
10.4.2.2.1.6.7. PD-1 and PD-L1 inhibitors
10.4.2.2.1.6.8. Others
10.4.2.2.1.7. Monoclonal
Antibodies
10.4.2.2.1.8. Others
10.4.2.2.2. Surgery
10.4.2.2.2.1. Open
Surgery
10.4.2.2.2.2. Minimally
Invasive Surgery
10.4.2.2.3. Radiation
Therapy
10.4.2.2.4. Chemotherapy
10.4.2.2.5. Immunotherapy
10.4.2.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
10.4.2.2.5.2. Cytokine
Treatment
10.4.2.2.5.3. Donor
lymphocyte infusion
10.4.2.2.5.4. Monoclonal
Antibody Treatment
10.4.2.2.5.5. Others
10.4.2.2.6. Targeted
Therapy
10.4.2.2.7. Hormone
Therapy
10.4.2.2.8. Stem
Cell Therapy
10.4.2.2.8.1. Autologous
stem cell transplantation
10.4.2.2.8.2. Allogeneic
stem cell transplantation
10.4.2.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
10.4.2.2.8.4. Graft-Versus-Host
Disease
10.4.2.2.9. Precision
Medicine
10.4.2.2.10. Others
10.4.3. Mexico
10.4.3.1. Mexico
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
10.4.3.1.1. Breast
Cancer
10.4.3.1.2. Prostate
Cancer
10.4.3.1.3. Basal
Cell Cancer
10.4.3.1.4. Skin
Cancer (Non-Melanoma)
10.4.3.1.5. Colorectal
cancer
10.4.3.1.6. Lung
Cancer
10.4.3.1.7. Renal
Cancer
10.4.3.1.8. Bladder
Cancer
10.4.3.1.9. Lymphoma
10.4.3.1.10. Others
10.4.3.2. Mexico
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
10.4.3.2.1. Drug
Therapies
10.4.3.2.1.1. Antimetabolites
10.4.3.2.1.2. Antitumor
Antibiotics
10.4.3.2.1.3. Asparagine-Specific
Enzymes
10.4.3.2.1.4. Bisphosphonates
and Biosimilars
10.4.3.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
10.4.3.2.1.6. Inhibitors
10.4.3.2.1.6.1. DNA-Repair Enzyme Inhibitors
10.4.3.2.1.6.2. Histone Deacetylase Inhibitors
10.4.3.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
10.4.3.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
10.4.3.2.1.6.5. Proteasome Inhibitors
10.4.3.2.1.6.6. Tyrosine Kinase Inhibitors
10.4.3.2.1.6.7. PD-1 and PD-L1 inhibitors
10.4.3.2.1.6.8. Others
10.4.3.2.1.7. Monoclonal
Antibodies
10.4.3.2.1.8. Others
10.4.3.2.2. Surgery
10.4.3.2.2.1. Open
Surgery
10.4.3.2.2.2. Minimally
Invasive Surgery
10.4.3.2.3. Radiation
Therapy
10.4.3.2.4. Chemotherapy
10.4.3.2.5. Immunotherapy
10.4.3.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
10.4.3.2.5.2. Cytokine
Treatment
10.4.3.2.5.3. Donor
lymphocyte infusion
10.4.3.2.5.4. Monoclonal
Antibody Treatment
10.4.3.2.5.5. Others
10.4.3.2.6. Targeted
Therapy
10.4.3.2.7. Hormone
Therapy
10.4.3.2.8. Stem
Cell Therapy
10.4.3.2.8.1. Autologous
stem cell transplantation
10.4.3.2.8.2. Allogeneic
stem cell transplantation
10.4.3.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
10.4.3.2.8.4. Graft-Versus-Host
Disease
10.4.3.2.9. Precision
Medicine
10.4.3.2.10. Others
10.4.4. Rest of
North America
10.4.4.1. Rest
of North America Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By
Indication
10.4.4.1.1. Breast
Cancer
10.4.4.1.2. Prostate
Cancer
10.4.4.1.3. Basal
Cell Cancer
10.4.4.1.4. Skin
Cancer (Non-Melanoma)
10.4.4.1.5. Colorectal
cancer
10.4.4.1.6. Lung
Cancer
10.4.4.1.7. Renal
Cancer
10.4.4.1.8. Bladder
Cancer
10.4.4.1.9. Lymphoma
10.4.4.1.10. Others
10.4.4.2. Rest
of North America Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By
Treatment
10.4.4.2.1. Drug
Therapies
10.4.4.2.1.1. Antimetabolites
10.4.4.2.1.2. Antitumor
Antibiotics
10.4.4.2.1.3. Asparagine-Specific
Enzymes
10.4.4.2.1.4. Bisphosphonates
and Biosimilars
10.4.4.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
10.4.4.2.1.6. Inhibitors
10.4.4.2.1.6.1. DNA-Repair Enzyme Inhibitors
10.4.4.2.1.6.2. Histone Deacetylase Inhibitors
10.4.4.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
10.4.4.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
10.4.4.2.1.6.5. Proteasome Inhibitors
10.4.4.2.1.6.6. Tyrosine Kinase Inhibitors
10.4.4.2.1.6.7. PD-1 and PD-L1 inhibitors
10.4.4.2.1.6.8. Others
10.4.4.2.1.7. Monoclonal
Antibodies
10.4.4.2.1.8. Others
10.4.4.2.2. Surgery
10.4.4.2.2.1. Open
Surgery
10.4.4.2.2.2. Minimally
Invasive Surgery
10.4.4.2.3. Radiation
Therapy
10.4.4.2.4. Chemotherapy
10.4.4.2.5. Immunotherapy
10.4.4.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
10.4.4.2.5.2. Cytokine
Treatment
10.4.4.2.5.3. Donor
lymphocyte infusion
10.4.4.2.5.4. Monoclonal
Antibody Treatment
10.4.4.2.5.5. Others
10.4.4.2.6. Targeted
Therapy
10.4.4.2.7. Hormone
Therapy
10.4.4.2.8. Stem
Cell Therapy
10.4.4.2.8.1. Autologous
stem cell transplantation
10.4.4.2.8.2. Allogeneic
stem cell transplantation
10.4.4.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
10.4.4.2.8.4. Graft-Versus-Host
Disease
10.4.4.2.9. Precision
Medicine
10.4.4.2.10. Others
10.5. Key
Segment for Channeling Investments
10.5.1. By
Country
10.5.2. By
Indication
10.5.3. By
Treatment
11. Europe Oncology Drugs Market Analysis and Forecasts, 2019 -
2027
11.1. Overview
11.1.1. Europe
Oncology Drugs Market Revenue (US$ Mn)
11.2. Europe
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
11.2.1. Breast
Cancer
11.2.2. Prostate
Cancer
11.2.3. Basal
Cell Cancer
11.2.4. Skin
Cancer (Non-Melanoma)
11.2.5. Colorectal
cancer
11.2.6. Lung
Cancer
11.2.7. Renal
Cancer
11.2.8. Bladder
Cancer
11.2.9. Lymphoma
11.2.10. Others
11.3. Europe
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
11.3.1. Drug
Therapies
11.3.1.1. Antimetabolites
11.3.1.2. Antitumor
Antibiotics
11.3.1.3. Asparagine-Specific
Enzymes
11.3.1.4. Bisphosphonates
and Biosimilars
11.3.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
11.3.1.6. Inhibitors
11.3.1.6.1. DNA-Repair
Enzyme Inhibitors
11.3.1.6.2. Histone
Deacetylase Inhibitors
11.3.1.6.3. Janus-Associated
Kinase (JAK) Inhibitors
11.3.1.6.4. Phosphoinositide
3-kinase inhibitors (PI3K inhibitors)
11.3.1.6.5. Proteasome
Inhibitors
11.3.1.6.6. Tyrosine
Kinase Inhibitors
11.3.1.6.7. PD-1
and PD-L1 inhibitors
11.3.1.6.8. Others
11.3.1.7. Monoclonal
Antibodies
11.3.1.8. Others
11.3.2. Surgery
11.3.2.1. Open
Surgery
11.3.2.2. Minimally
Invasive Surgery
11.3.3. Radiation
Therapy
11.3.4. Chemotherapy
11.3.5. Immunotherapy
11.3.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
11.3.5.2. Cytokine
Treatment
11.3.5.3. Donor
lymphocyte infusion
11.3.5.4. Monoclonal
Antibody Treatment
11.3.5.5. Others
11.3.6. Targeted
Therapy
11.3.7. Hormone
Therapy
11.3.8. Stem
Cell Therapy
11.3.8.1. Autologous
stem cell transplantation
11.3.8.2. Allogeneic
stem cell transplantation
11.3.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
11.3.8.4. Graft-Versus-Host
Disease
11.3.9. Precision
Medicine
11.3.10. Others
11.4. Europe
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Country
11.4.1. France
11.4.1.1. France
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
11.4.1.1.1. Breast
Cancer
11.4.1.1.2. Prostate
Cancer
11.4.1.1.3. Basal
Cell Cancer
11.4.1.1.4. Skin
Cancer (Non-Melanoma)
11.4.1.1.5. Colorectal
cancer
11.4.1.1.6. Lung
Cancer
11.4.1.1.7. Renal
Cancer
11.4.1.1.8. Bladder
Cancer
11.4.1.1.9. Lymphoma
11.4.1.1.10. Others
11.4.1.2. France
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
11.4.1.2.1. Drug
Therapies
11.4.1.2.1.1. Antimetabolites
11.4.1.2.1.2. Antitumor
Antibiotics
11.4.1.2.1.3. Asparagine-Specific
Enzymes
11.4.1.2.1.4. Bisphosphonates
and Biosimilars
11.4.1.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
11.4.1.2.1.6. Inhibitors
11.4.1.2.1.6.1. DNA-Repair Enzyme Inhibitors
11.4.1.2.1.6.2. Histone Deacetylase Inhibitors
11.4.1.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
11.4.1.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
11.4.1.2.1.6.5. Proteasome Inhibitors
11.4.1.2.1.6.6. Tyrosine Kinase Inhibitors
11.4.1.2.1.6.7. PD-1 and PD-L1 inhibitors
11.4.1.2.1.6.8. Others
11.4.1.2.1.7. Monoclonal
Antibodies
11.4.1.2.1.8. Others
11.4.1.2.2. Surgery
11.4.1.2.2.1. Open
Surgery
11.4.1.2.2.2. Minimally
Invasive Surgery
11.4.1.2.3. Radiation
Therapy
11.4.1.2.4. Chemotherapy
11.4.1.2.5. Immunotherapy
11.4.1.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
11.4.1.2.5.2. Cytokine
Treatment
11.4.1.2.5.3. Donor
lymphocyte infusion
11.4.1.2.5.4. Monoclonal
Antibody Treatment
11.4.1.2.5.5. Others
11.4.1.2.6. Targeted
Therapy
11.4.1.2.7. Hormone
Therapy
11.4.1.2.8. Stem
Cell Therapy
11.4.1.2.8.1. Autologous
stem cell transplantation
11.4.1.2.8.2. Allogeneic
stem cell transplantation
11.4.1.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
11.4.1.2.8.4. Graft-Versus-Host
Disease
11.4.1.2.9. Precision
Medicine
11.4.1.2.10. Others
11.4.2. The UK
11.4.2.1. The
UK Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
11.4.2.1.1. Breast
Cancer
11.4.2.1.2. Prostate
Cancer
11.4.2.1.3. Basal
Cell Cancer
11.4.2.1.4. Skin
Cancer (Non-Melanoma)
11.4.2.1.5. Colorectal
cancer
11.4.2.1.6. Lung
Cancer
11.4.2.1.7. Renal
Cancer
11.4.2.1.8. Bladder
Cancer
11.4.2.1.9. Lymphoma
11.4.2.1.10. Others
11.4.2.2. The
UK Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
11.4.2.2.1. Drug
Therapies
11.4.2.2.1.1. Antimetabolites
11.4.2.2.1.2. Antitumor
Antibiotics
11.4.2.2.1.3. Asparagine-Specific
Enzymes
11.4.2.2.1.4. Bisphosphonates
and Biosimilars
11.4.2.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
11.4.2.2.1.6. Inhibitors
11.4.2.2.1.6.1. DNA-Repair Enzyme Inhibitors
11.4.2.2.1.6.2. Histone Deacetylase Inhibitors
11.4.2.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
11.4.2.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
11.4.2.2.1.6.5. Proteasome Inhibitors
11.4.2.2.1.6.6. Tyrosine Kinase Inhibitors
11.4.2.2.1.6.7. PD-1 and PD-L1 inhibitors
11.4.2.2.1.6.8. Others
11.4.2.2.1.7. Monoclonal
Antibodies
11.4.2.2.1.8. Others
11.4.2.2.2. Surgery
11.4.2.2.2.1. Open
Surgery
11.4.2.2.2.2. Minimally
Invasive Surgery
11.4.2.2.3. Radiation
Therapy
11.4.2.2.4. Chemotherapy
11.4.2.2.5. Immunotherapy
11.4.2.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
11.4.2.2.5.2. Cytokine
Treatment
11.4.2.2.5.3. Donor
lymphocyte infusion
11.4.2.2.5.4. Monoclonal
Antibody Treatment
11.4.2.2.5.5. Others
11.4.2.2.6. Targeted
Therapy
11.4.2.2.7. Hormone
Therapy
11.4.2.2.8. Stem
Cell Therapy
11.4.2.2.8.1. Autologous
stem cell transplantation
11.4.2.2.8.2. Allogeneic
stem cell transplantation
11.4.2.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
11.4.2.2.8.4. Graft-Versus-Host
Disease
11.4.2.2.9. Precision
Medicine
11.4.2.2.10. Others
11.4.3. Spain
11.4.3.1. Spain
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
11.4.3.1.1. Breast
Cancer
11.4.3.1.2. Prostate
Cancer
11.4.3.1.3. Basal
Cell Cancer
11.4.3.1.4. Skin
Cancer (Non-Melanoma)
11.4.3.1.5. Colorectal
cancer
11.4.3.1.6. Lung
Cancer
11.4.3.1.7. Renal
Cancer
11.4.3.1.8. Bladder
Cancer
11.4.3.1.9. Lymphoma
11.4.3.1.10. Others
11.4.3.2. Spain
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
11.4.3.2.1. Drug
Therapies
11.4.3.2.1.1. Antimetabolites
11.4.3.2.1.2. Antitumor
Antibiotics
11.4.3.2.1.3. Asparagine-Specific
Enzymes
11.4.3.2.1.4. Bisphosphonates
and Biosimilars
11.4.3.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
11.4.3.2.1.6. Inhibitors
11.4.3.2.1.6.1. DNA-Repair Enzyme Inhibitors
11.4.3.2.1.6.2. Histone Deacetylase Inhibitors
11.4.3.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
11.4.3.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
11.4.3.2.1.6.5. Proteasome Inhibitors
11.4.3.2.1.6.6. Tyrosine Kinase Inhibitors
11.4.3.2.1.6.7. PD-1 and PD-L1 inhibitors
11.4.3.2.1.6.8. Others
11.4.3.2.1.7. Monoclonal
Antibodies
11.4.3.2.1.8. Others
11.4.3.2.2. Surgery
11.4.3.2.2.1. Open
Surgery
11.4.3.2.2.2. Minimally
Invasive Surgery
11.4.3.2.3. Radiation
Therapy
11.4.3.2.4. Chemotherapy
11.4.3.2.5. Immunotherapy
11.4.3.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
11.4.3.2.5.2. Cytokine
Treatment
11.4.3.2.5.3. Donor
lymphocyte infusion
11.4.3.2.5.4. Monoclonal
Antibody Treatment
11.4.3.2.5.5. Others
11.4.3.2.6. Targeted
Therapy
11.4.3.2.7. Hormone
Therapy
11.4.3.2.8. Stem
Cell Therapy
11.4.3.2.8.1. Autologous
stem cell transplantation
11.4.3.2.8.2. Allogeneic
stem cell transplantation
11.4.3.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
11.4.3.2.8.4. Graft-Versus-Host
Disease
11.4.3.2.9. Precision
Medicine
11.4.3.2.10. Others
11.4.4. Germany
11.4.4.1. Germany
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
11.4.4.1.1. Breast
Cancer
11.4.4.1.2. Prostate
Cancer
11.4.4.1.3. Basal
Cell Cancer
11.4.4.1.4. Skin
Cancer (Non-Melanoma)
11.4.4.1.5. Colorectal
cancer
11.4.4.1.6. Lung
Cancer
11.4.4.1.7. Renal
Cancer
11.4.4.1.8. Bladder
Cancer
11.4.4.1.9. Lymphoma
11.4.4.1.10. Others
11.4.4.2. Germany
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
11.4.4.2.1. Drug
Therapies
11.4.4.2.1.1. Antimetabolites
11.4.4.2.1.2. Antitumor
Antibiotics
11.4.4.2.1.3. Asparagine-Specific
Enzymes
11.4.4.2.1.4. Bisphosphonates
and Biosimilars
11.4.4.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
11.4.4.2.1.6. Inhibitors
11.4.4.2.1.6.1. DNA-Repair Enzyme Inhibitors
11.4.4.2.1.6.2. Histone Deacetylase Inhibitors
11.4.4.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
11.4.4.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
11.4.4.2.1.6.5. Proteasome Inhibitors
11.4.4.2.1.6.6. Tyrosine Kinase Inhibitors
11.4.4.2.1.6.7. PD-1 and PD-L1 inhibitors
11.4.4.2.1.6.8. Others
11.4.4.2.1.7. Monoclonal
Antibodies
11.4.4.2.1.8. Others
11.4.4.2.2. Surgery
11.4.4.2.2.1. Open
Surgery
11.4.4.2.2.2. Minimally
Invasive Surgery
11.4.4.2.3. Radiation
Therapy
11.4.4.2.4. Chemotherapy
11.4.4.2.5. Immunotherapy
11.4.4.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
11.4.4.2.5.2. Cytokine
Treatment
11.4.4.2.5.3. Donor
lymphocyte infusion
11.4.4.2.5.4. Monoclonal
Antibody Treatment
11.4.4.2.5.5. Others
11.4.4.2.6. Targeted
Therapy
11.4.4.2.7. Hormone
Therapy
11.4.4.2.8. Stem
Cell Therapy
11.4.4.2.8.1. Autologous
stem cell transplantation
11.4.4.2.8.2. Allogeneic
stem cell transplantation
11.4.4.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
11.4.4.2.8.4. Graft-Versus-Host
Disease
11.4.4.2.9. Precision
Medicine
11.4.4.2.10. Others
11.4.5. Italy
11.4.5.1. Italy
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
11.4.5.1.1. Breast
Cancer
11.4.5.1.2. Prostate
Cancer
11.4.5.1.3. Basal
Cell Cancer
11.4.5.1.4. Skin
Cancer (Non-Melanoma)
11.4.5.1.5. Colorectal
cancer
11.4.5.1.6. Lung
Cancer
11.4.5.1.7. Renal
Cancer
11.4.5.1.8. Bladder
Cancer
11.4.5.1.9. Lymphoma
11.4.5.1.10. Others
11.4.5.2. Italy
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
11.4.5.2.1. Drug
Therapies
11.4.5.2.1.1. Antimetabolites
11.4.5.2.1.2. Antitumor
Antibiotics
11.4.5.2.1.3. Asparagine-Specific
Enzymes
11.4.5.2.1.4. Bisphosphonates
and Biosimilars
11.4.5.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
11.4.5.2.1.6. Inhibitors
11.4.5.2.1.6.1. DNA-Repair Enzyme Inhibitors
11.4.5.2.1.6.2. Histone Deacetylase Inhibitors
11.4.5.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
11.4.5.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
11.4.5.2.1.6.5. Proteasome Inhibitors
11.4.5.2.1.6.6. Tyrosine Kinase Inhibitors
11.4.5.2.1.6.7. PD-1 and PD-L1 inhibitors
11.4.5.2.1.6.8. Others
11.4.5.2.1.7. Monoclonal
Antibodies
11.4.5.2.1.8. Others
11.4.5.2.2. Surgery
11.4.5.2.2.1. Open
Surgery
11.4.5.2.2.2. Minimally
Invasive Surgery
11.4.5.2.3. Radiation
Therapy
11.4.5.2.4. Chemotherapy
11.4.5.2.5. Immunotherapy
11.4.5.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
11.4.5.2.5.2. Cytokine
Treatment
11.4.5.2.5.3. Donor
lymphocyte infusion
11.4.5.2.5.4. Monoclonal
Antibody Treatment
11.4.5.2.5.5. Others
11.4.5.2.6. Targeted
Therapy
11.4.5.2.7. Hormone
Therapy
11.4.5.2.8. Stem
Cell Therapy
11.4.5.2.8.1. Autologous
stem cell transplantation
11.4.5.2.8.2. Allogeneic
stem cell transplantation
11.4.5.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
11.4.5.2.8.4. Graft-Versus-Host
Disease
11.4.5.2.9. Precision
Medicine
11.4.5.2.10. Others
11.4.6. Nordic
Countries
11.4.6.1. Nordic
Countries Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
11.4.6.1.1. Breast
Cancer
11.4.6.1.2. Prostate
Cancer
11.4.6.1.3. Basal
Cell Cancer
11.4.6.1.4. Skin
Cancer (Non-Melanoma)
11.4.6.1.5. Colorectal
cancer
11.4.6.1.6. Lung
Cancer
11.4.6.1.7. Renal
Cancer
11.4.6.1.8. Bladder
Cancer
11.4.6.1.9. Lymphoma
11.4.6.1.10. Others
11.4.6.2. Nordic
Countries Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
11.4.6.2.1. Drug
Therapies
11.4.6.2.1.1. Antimetabolites
11.4.6.2.1.2. Antitumor
Antibiotics
11.4.6.2.1.3. Asparagine-Specific
Enzymes
11.4.6.2.1.4. Bisphosphonates
and Biosimilars
11.4.6.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
11.4.6.2.1.6. Inhibitors
11.4.6.2.1.6.1. DNA-Repair Enzyme Inhibitors
11.4.6.2.1.6.2. Histone Deacetylase Inhibitors
11.4.6.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
11.4.6.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
11.4.6.2.1.6.5. Proteasome Inhibitors
11.4.6.2.1.6.6. Tyrosine Kinase Inhibitors
11.4.6.2.1.6.7. PD-1 and PD-L1 inhibitors
11.4.6.2.1.6.8. Others
11.4.6.2.1.7. Monoclonal
Antibodies
11.4.6.2.1.8. Others
11.4.6.2.2. Surgery
11.4.6.2.2.1. Open
Surgery
11.4.6.2.2.2. Minimally
Invasive Surgery
11.4.6.2.3. Radiation
Therapy
11.4.6.2.4. Chemotherapy
11.4.6.2.5. Immunotherapy
11.4.6.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
11.4.6.2.5.2. Cytokine
Treatment
11.4.6.2.5.3. Donor
lymphocyte infusion
11.4.6.2.5.4. Monoclonal
Antibody Treatment
11.4.6.2.5.5. Others
11.4.6.2.6. Targeted
Therapy
11.4.6.2.7. Hormone
Therapy
11.4.6.2.8. Stem
Cell Therapy
11.4.6.2.8.1. Autologous
stem cell transplantation
11.4.6.2.8.2. Allogeneic
stem cell transplantation
11.4.6.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
11.4.6.2.8.4. Graft-Versus-Host
Disease
11.4.6.2.9. Precision
Medicine
11.4.6.2.10. Others
11.4.6.3. Nordic
Countries Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Country
11.4.6.3.1. Denmark
11.4.6.3.2. Finland
11.4.6.3.3. Iceland
11.4.6.3.4. Sweden
11.4.6.3.5. Norway
11.4.7. Benelux
Union
11.4.7.1. Benelux
Union Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
11.4.7.1.1. Breast
Cancer
11.4.7.1.2. Prostate
Cancer
11.4.7.1.3. Basal
Cell Cancer
11.4.7.1.4. Skin
Cancer (Non-Melanoma)
11.4.7.1.5. Colorectal
cancer
11.4.7.1.6. Lung
Cancer
11.4.7.1.7. Renal
Cancer
11.4.7.1.8. Bladder
Cancer
11.4.7.1.9. Lymphoma
11.4.7.1.10. Others
11.4.7.2. Benelux
Union Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
11.4.7.2.1. Drug
Therapies
11.4.7.2.1.1. Antimetabolites
11.4.7.2.1.2. Antitumor
Antibiotics
11.4.7.2.1.3. Asparagine-Specific
Enzymes
11.4.7.2.1.4. Bisphosphonates
and Biosimilars
11.4.7.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
11.4.7.2.1.6. Inhibitors
11.4.7.2.1.6.1. DNA-Repair Enzyme Inhibitors
11.4.7.2.1.6.2. Histone Deacetylase Inhibitors
11.4.7.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
11.4.7.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
11.4.7.2.1.6.5. Proteasome Inhibitors
11.4.7.2.1.6.6. Tyrosine Kinase Inhibitors
11.4.7.2.1.6.7. PD-1 and PD-L1 inhibitors
11.4.7.2.1.6.8. Others
11.4.7.2.1.7. Monoclonal
Antibodies
11.4.7.2.1.8. Others
11.4.7.2.2. Surgery
11.4.7.2.2.1. Open
Surgery
11.4.7.2.2.2. Minimally
Invasive Surgery
11.4.7.2.3. Radiation
Therapy
11.4.7.2.4. Chemotherapy
11.4.7.2.5. Immunotherapy
11.4.7.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
11.4.7.2.5.2. Cytokine
Treatment
11.4.7.2.5.3. Donor
lymphocyte infusion
11.4.7.2.5.4. Monoclonal
Antibody Treatment
11.4.7.2.5.5. Others
11.4.7.2.6. Targeted
Therapy
11.4.7.2.7. Hormone
Therapy
11.4.7.2.8. Stem
Cell Therapy
11.4.7.2.8.1. Autologous
stem cell transplantation
11.4.7.2.8.2. Allogeneic
stem cell transplantation
11.4.7.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
11.4.7.2.8.4. Graft-Versus-Host
Disease
11.4.7.2.9. Precision
Medicine
11.4.7.2.10. Others
11.4.7.3. Benelux
Union Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Country
11.4.7.3.1. Belgium
11.4.7.3.2. The
Netherlands
11.4.7.3.3. Luxembourg
11.4.8. Rest of
Europe
11.4.8.1. Rest
of Europe Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
11.4.8.1.1. Breast
Cancer
11.4.8.1.2. Prostate
Cancer
11.4.8.1.3. Basal
Cell Cancer
11.4.8.1.4. Skin
Cancer (Non-Melanoma)
11.4.8.1.5. Colorectal
cancer
11.4.8.1.6. Lung
Cancer
11.4.8.1.7. Renal
Cancer
11.4.8.1.8. Bladder
Cancer
11.4.8.1.9. Lymphoma
11.4.8.1.10. Others
11.4.8.2. Rest
of Europe Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
11.4.8.2.1. Drug
Therapies
11.4.8.2.1.1. Antimetabolites
11.4.8.2.1.2. Antitumor
Antibiotics
11.4.8.2.1.3. Asparagine-Specific
Enzymes
11.4.8.2.1.4. Bisphosphonates
and Biosimilars
11.4.8.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
11.4.8.2.1.6. Inhibitors
11.4.8.2.1.6.1. DNA-Repair Enzyme Inhibitors
11.4.8.2.1.6.2. Histone Deacetylase Inhibitors
11.4.8.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
11.4.8.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
11.4.8.2.1.6.5. Proteasome Inhibitors
11.4.8.2.1.6.6. Tyrosine Kinase Inhibitors
11.4.8.2.1.6.7. PD-1 and PD-L1 inhibitors
11.4.8.2.1.6.8. Others
11.4.8.2.1.7. Monoclonal
Antibodies
11.4.8.2.1.8. Others
11.4.8.2.2. Surgery
11.4.8.2.2.1. Open
Surgery
11.4.8.2.2.2. Minimally
Invasive Surgery
11.4.8.2.3. Radiation
Therapy
11.4.8.2.4. Chemotherapy
11.4.8.2.5. Immunotherapy
11.4.8.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
11.4.8.2.5.2. Cytokine
Treatment
11.4.8.2.5.3. Donor
lymphocyte infusion
11.4.8.2.5.4. Monoclonal
Antibody Treatment
11.4.8.2.5.5. Others
11.4.8.2.6. Targeted
Therapy
11.4.8.2.7. Hormone
Therapy
11.4.8.2.8. Stem
Cell Therapy
11.4.8.2.8.1. Autologous
stem cell transplantation
11.4.8.2.8.2. Allogeneic
stem cell transplantation
11.4.8.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
11.4.8.2.8.4. Graft-Versus-Host
Disease
11.4.8.2.9. Precision
Medicine
11.4.8.2.10. Others
11.5. Key
Segment for Channeling Investments
11.5.1. By
Country
11.5.2. By
Indication
11.5.3. By
Treatment
12. Asia Pacific Oncology Drugs Market Analysis and Forecasts,
2019 - 2027
12.1. Overview
12.1.1. Asia
Pacific Oncology Drugs Market Revenue (US$ Mn)
12.2. Asia
Pacific Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
12.2.1. Breast
Cancer
12.2.2. Prostate
Cancer
12.2.3. Basal
Cell Cancer
12.2.4. Skin
Cancer (Non-Melanoma)
12.2.5. Colorectal
cancer
12.2.6. Lung
Cancer
12.2.7. Renal
Cancer
12.2.8. Bladder
Cancer
12.2.9. Lymphoma
12.2.10. Others
12.3. Asia Pacific
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
12.3.1. Drug
Therapies
12.3.1.1. Antimetabolites
12.3.1.2. Antitumor
Antibiotics
12.3.1.3. Asparagine-Specific
Enzymes
12.3.1.4. Bisphosphonates
and Biosimilars
12.3.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
12.3.1.6. Inhibitors
12.3.1.6.1. DNA-Repair
Enzyme Inhibitors
12.3.1.6.2. Histone
Deacetylase Inhibitors
12.3.1.6.3. Janus-Associated
Kinase (JAK) Inhibitors
12.3.1.6.4. Phosphoinositide
3-kinase inhibitors (PI3K inhibitors)
12.3.1.6.5. Proteasome
Inhibitors
12.3.1.6.6. Tyrosine
Kinase Inhibitors
12.3.1.6.7. PD-1
and PD-L1 inhibitors
12.3.1.6.8. Others
12.3.1.7. Monoclonal
Antibodies
12.3.1.8. Others
12.3.2. Surgery
12.3.2.1. Open
Surgery
12.3.2.2. Minimally
Invasive Surgery
12.3.3. Radiation
Therapy
12.3.4. Chemotherapy
12.3.5. Immunotherapy
12.3.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
12.3.5.2. Cytokine
Treatment
12.3.5.3. Donor
lymphocyte infusion
12.3.5.4. Monoclonal
Antibody Treatment
12.3.5.5. Others
12.3.6. Targeted
Therapy
12.3.7. Hormone
Therapy
12.3.8. Stem
Cell Therapy
12.3.8.1. Autologous
stem cell transplantation
12.3.8.2. Allogeneic
stem cell transplantation
12.3.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
12.3.8.4. Graft-Versus-Host
Disease
12.3.9. Precision
Medicine
12.3.10. Others
12.4. Asia
Pacific Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Country
12.4.1. China
12.4.1.1. China
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
12.4.1.1.1. Breast
Cancer
12.4.1.1.2. Prostate
Cancer
12.4.1.1.3. Basal
Cell Cancer
12.4.1.1.4. Skin
Cancer (Non-Melanoma)
12.4.1.1.5. Colorectal
cancer
12.4.1.1.6. Lung
Cancer
12.4.1.1.7. Renal
Cancer
12.4.1.1.8. Bladder
Cancer
12.4.1.1.9. Lymphoma
12.4.1.1.10. Others
12.4.1.2. China
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
12.4.1.2.1. Drug
Therapies
12.4.1.2.1.1. Antimetabolites
12.4.1.2.1.2. Antitumor
Antibiotics
12.4.1.2.1.3. Asparagine-Specific
Enzymes
12.4.1.2.1.4. Bisphosphonates
and Biosimilars
12.4.1.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
12.4.1.2.1.6. Inhibitors
12.4.1.2.1.6.1. DNA-Repair Enzyme Inhibitors
12.4.1.2.1.6.2. Histone Deacetylase Inhibitors
12.4.1.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
12.4.1.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
12.4.1.2.1.6.5. Proteasome Inhibitors
12.4.1.2.1.6.6. Tyrosine Kinase Inhibitors
12.4.1.2.1.6.7. PD-1 and PD-L1 inhibitors
12.4.1.2.1.6.8. Others
12.4.1.2.1.7. Monoclonal
Antibodies
12.4.1.2.1.8. Others
12.4.1.2.2. Surgery
12.4.1.2.2.1. Open
Surgery
12.4.1.2.2.2. Minimally
Invasive Surgery
12.4.1.2.3. Radiation
Therapy
12.4.1.2.4. Chemotherapy
12.4.1.2.5. Immunotherapy
12.4.1.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
12.4.1.2.5.2. Cytokine
Treatment
12.4.1.2.5.3. Donor
lymphocyte infusion
12.4.1.2.5.4. Monoclonal
Antibody Treatment
12.4.1.2.5.5. Others
12.4.1.2.6. Targeted
Therapy
12.4.1.2.7. Hormone
Therapy
12.4.1.2.8. Stem
Cell Therapy
12.4.1.2.8.1. Autologous
stem cell transplantation
12.4.1.2.8.2. Allogeneic
stem cell transplantation
12.4.1.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
12.4.1.2.8.4. Graft-Versus-Host
Disease
12.4.1.2.9. Precision
Medicine
12.4.1.2.10. Others
12.4.2. Japan
12.4.2.1. Japan
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
12.4.2.1.1. Breast
Cancer
12.4.2.1.2. Prostate
Cancer
12.4.2.1.3. Basal
Cell Cancer
12.4.2.1.4. Skin
Cancer (Non-Melanoma)
12.4.2.1.5. Colorectal
cancer
12.4.2.1.6. Lung
Cancer
12.4.2.1.7. Renal
Cancer
12.4.2.1.8. Bladder
Cancer
12.4.2.1.9. Lymphoma
12.4.2.1.10. Others
12.4.2.2. Japan
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
12.4.2.2.1. Drug
Therapies
12.4.2.2.1.1. Antimetabolites
12.4.2.2.1.2. Antitumor
Antibiotics
12.4.2.2.1.3. Asparagine-Specific
Enzymes
12.4.2.2.1.4. Bisphosphonates
and Biosimilars
12.4.2.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
12.4.2.2.1.6. Inhibitors
12.4.2.2.1.6.1. DNA-Repair Enzyme Inhibitors
12.4.2.2.1.6.2. Histone Deacetylase Inhibitors
12.4.2.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
12.4.2.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
12.4.2.2.1.6.5. Proteasome Inhibitors
12.4.2.2.1.6.6. Tyrosine Kinase Inhibitors
12.4.2.2.1.6.7. PD-1 and PD-L1 inhibitors
12.4.2.2.1.6.8. Others
12.4.2.2.1.7. Monoclonal
Antibodies
12.4.2.2.1.8. Others
12.4.2.2.2. Surgery
12.4.2.2.2.1. Open
Surgery
12.4.2.2.2.2. Minimally
Invasive Surgery
12.4.2.2.3. Radiation
Therapy
12.4.2.2.4. Chemotherapy
12.4.2.2.5. Immunotherapy
12.4.2.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
12.4.2.2.5.2. Cytokine
Treatment
12.4.2.2.5.3. Donor
lymphocyte infusion
12.4.2.2.5.4. Monoclonal
Antibody Treatment
12.4.2.2.5.5. Others
12.4.2.2.6. Targeted
Therapy
12.4.2.2.7. Hormone
Therapy
12.4.2.2.8. Stem
Cell Therapy
12.4.2.2.8.1. Autologous
stem cell transplantation
12.4.2.2.8.2. Allogeneic
stem cell transplantation
12.4.2.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
12.4.2.2.8.4. Graft-Versus-Host
Disease
12.4.2.2.9. Precision
Medicine
12.4.2.2.10. Others
12.4.3. India
12.4.3.1. India
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
12.4.3.1.1. Breast
Cancer
12.4.3.1.2. Prostate
Cancer
12.4.3.1.3. Basal
Cell Cancer
12.4.3.1.4. Skin
Cancer (Non-Melanoma)
12.4.3.1.5. Colorectal
cancer
12.4.3.1.6. Lung
Cancer
12.4.3.1.7. Renal
Cancer
12.4.3.1.8. Bladder
Cancer
12.4.3.1.9. Lymphoma
12.4.3.1.10. Others
12.4.3.2. India
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
12.4.3.2.1. Drug
Therapies
12.4.3.2.1.1. Antimetabolites
12.4.3.2.1.2. Antitumor
Antibiotics
12.4.3.2.1.3. Asparagine-Specific
Enzymes
12.4.3.2.1.4. Bisphosphonates
and Biosimilars
12.4.3.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
12.4.3.2.1.6. Inhibitors
12.4.3.2.1.6.1. DNA-Repair Enzyme Inhibitors
12.4.3.2.1.6.2. Histone Deacetylase Inhibitors
12.4.3.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
12.4.3.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
12.4.3.2.1.6.5. Proteasome Inhibitors
12.4.3.2.1.6.6. Tyrosine Kinase Inhibitors
12.4.3.2.1.6.7. PD-1 and PD-L1 inhibitors
12.4.3.2.1.6.8. Others
12.4.3.2.1.7. Monoclonal
Antibodies
12.4.3.2.1.8. Others
12.4.3.2.2. Surgery
12.4.3.2.2.1. Open
Surgery
12.4.3.2.2.2. Minimally
Invasive Surgery
12.4.3.2.3. Radiation
Therapy
12.4.3.2.4. Chemotherapy
12.4.3.2.5. Immunotherapy
12.4.3.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
12.4.3.2.5.2. Cytokine
Treatment
12.4.3.2.5.3. Donor
lymphocyte infusion
12.4.3.2.5.4. Monoclonal
Antibody Treatment
12.4.3.2.5.5. Others
12.4.3.2.6. Targeted
Therapy
12.4.3.2.7. Hormone
Therapy
12.4.3.2.8. Stem
Cell Therapy
12.4.3.2.8.1. Autologous
stem cell transplantation
12.4.3.2.8.2. Allogeneic
stem cell transplantation
12.4.3.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
12.4.3.2.8.4. Graft-Versus-Host
Disease
12.4.3.2.9. Precision
Medicine
12.4.3.2.10. Others
12.4.4. New
Zealand
12.4.4.1. New
Zealand Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
12.4.4.1.1. Breast
Cancer
12.4.4.1.2. Prostate
Cancer
12.4.4.1.3. Basal
Cell Cancer
12.4.4.1.4. Skin
Cancer (Non-Melanoma)
12.4.4.1.5. Colorectal
cancer
12.4.4.1.6. Lung
Cancer
12.4.4.1.7. Renal
Cancer
12.4.4.1.8. Bladder
Cancer
12.4.4.1.9. Lymphoma
12.4.4.1.10. Others
12.4.4.2. New
Zealand Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
12.4.4.2.1. Drug
Therapies
12.4.4.2.1.1. Antimetabolites
12.4.4.2.1.2. Antitumor
Antibiotics
12.4.4.2.1.3. Asparagine-Specific
Enzymes
12.4.4.2.1.4. Bisphosphonates
and Biosimilars
12.4.4.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
12.4.4.2.1.6. Inhibitors
12.4.4.2.1.6.1. DNA-Repair Enzyme Inhibitors
12.4.4.2.1.6.2. Histone Deacetylase Inhibitors
12.4.4.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
12.4.4.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
12.4.4.2.1.6.5. Proteasome Inhibitors
12.4.4.2.1.6.6. Tyrosine Kinase Inhibitors
12.4.4.2.1.6.7. PD-1 and PD-L1 inhibitors
12.4.4.2.1.6.8. Others
12.4.4.2.1.7. Monoclonal
Antibodies
12.4.4.2.1.8. Others
12.4.4.2.2. Surgery
12.4.4.2.2.1. Open
Surgery
12.4.4.2.2.2. Minimally
Invasive Surgery
12.4.4.2.3. Radiation
Therapy
12.4.4.2.4. Chemotherapy
12.4.4.2.5. Immunotherapy
12.4.4.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
12.4.4.2.5.2. Cytokine
Treatment
12.4.4.2.5.3. Donor
lymphocyte infusion
12.4.4.2.5.4. Monoclonal
Antibody Treatment
12.4.4.2.5.5. Others
12.4.4.2.6. Targeted
Therapy
12.4.4.2.7. Hormone
Therapy
12.4.4.2.8. Stem
Cell Therapy
12.4.4.2.8.1. Autologous
stem cell transplantation
12.4.4.2.8.2. Allogeneic
stem cell transplantation
12.4.4.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
12.4.4.2.8.4. Graft-Versus-Host
Disease
12.4.4.2.9. Precision
Medicine
12.4.4.2.10. Others
12.4.5. Australia
12.4.5.1. Australia
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
12.4.5.1.1. Breast
Cancer
12.4.5.1.2. Prostate
Cancer
12.4.5.1.3. Basal
Cell Cancer
12.4.5.1.4. Skin
Cancer (Non-Melanoma)
12.4.5.1.5. Colorectal
cancer
12.4.5.1.6. Lung
Cancer
12.4.5.1.7. Renal
Cancer
12.4.5.1.8. Bladder
Cancer
12.4.5.1.9. Lymphoma
12.4.5.1.10. Others
12.4.5.2. Australia
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
12.4.5.2.1. Drug
Therapies
12.4.5.2.1.1. Antimetabolites
12.4.5.2.1.2. Antitumor
Antibiotics
12.4.5.2.1.3. Asparagine-Specific
Enzymes
12.4.5.2.1.4. Bisphosphonates
and Biosimilars
12.4.5.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
12.4.5.2.1.6. Inhibitors
12.4.5.2.1.6.1. DNA-Repair Enzyme Inhibitors
12.4.5.2.1.6.2. Histone Deacetylase Inhibitors
12.4.5.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
12.4.5.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
12.4.5.2.1.6.5. Proteasome Inhibitors
12.4.5.2.1.6.6. Tyrosine Kinase Inhibitors
12.4.5.2.1.6.7. PD-1 and PD-L1 inhibitors
12.4.5.2.1.6.8. Others
12.4.5.2.1.7. Monoclonal
Antibodies
12.4.5.2.1.8. Others
12.4.5.2.2. Surgery
12.4.5.2.2.1. Open
Surgery
12.4.5.2.2.2. Minimally
Invasive Surgery
12.4.5.2.3. Radiation
Therapy
12.4.5.2.4. Chemotherapy
12.4.5.2.5. Immunotherapy
12.4.5.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
12.4.5.2.5.2. Cytokine
Treatment
12.4.5.2.5.3. Donor
lymphocyte infusion
12.4.5.2.5.4. Monoclonal
Antibody Treatment
12.4.5.2.5.5. Others
12.4.5.2.6. Targeted
Therapy
12.4.5.2.7. Hormone
Therapy
12.4.5.2.8. Stem
Cell Therapy
12.4.5.2.8.1. Autologous
stem cell transplantation
12.4.5.2.8.2. Allogeneic
stem cell transplantation
12.4.5.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
12.4.5.2.8.4. Graft-Versus-Host
Disease
12.4.5.2.9. Precision
Medicine
12.4.5.2.10. Others
12.4.6. South
Korea
12.4.6.1. South
Korea Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
12.4.6.1.1. Breast
Cancer
12.4.6.1.2. Prostate
Cancer
12.4.6.1.3. Basal
Cell Cancer
12.4.6.1.4. Skin
Cancer (Non-Melanoma)
12.4.6.1.5. Colorectal
cancer
12.4.6.1.6. Lung
Cancer
12.4.6.1.7. Renal
Cancer
12.4.6.1.8. Bladder
Cancer
12.4.6.1.9. Lymphoma
12.4.6.1.10. Others
12.4.6.2. South
Korea Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
12.4.6.2.1. Drug
Therapies
12.4.6.2.1.1. Antimetabolites
12.4.6.2.1.2. Antitumor
Antibiotics
12.4.6.2.1.3. Asparagine-Specific
Enzymes
12.4.6.2.1.4. Bisphosphonates
and Biosimilars
12.4.6.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
12.4.6.2.1.6. Inhibitors
12.4.6.2.1.6.1. DNA-Repair Enzyme Inhibitors
12.4.6.2.1.6.2. Histone Deacetylase Inhibitors
12.4.6.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
12.4.6.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
12.4.6.2.1.6.5. Proteasome Inhibitors
12.4.6.2.1.6.6. Tyrosine Kinase Inhibitors
12.4.6.2.1.6.7. PD-1 and PD-L1 inhibitors
12.4.6.2.1.6.8. Others
12.4.6.2.1.7. Monoclonal
Antibodies
12.4.6.2.1.8. Others
12.4.6.2.2. Surgery
12.4.6.2.2.1. Open
Surgery
12.4.6.2.2.2. Minimally
Invasive Surgery
12.4.6.2.3. Radiation
Therapy
12.4.6.2.4. Chemotherapy
12.4.6.2.5. Immunotherapy
12.4.6.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
12.4.6.2.5.2. Cytokine
Treatment
12.4.6.2.5.3. Donor
lymphocyte infusion
12.4.6.2.5.4. Monoclonal
Antibody Treatment
12.4.6.2.5.5. Others
12.4.6.2.6. Targeted
Therapy
12.4.6.2.7. Hormone
Therapy
12.4.6.2.8. Stem
Cell Therapy
12.4.6.2.8.1. Autologous
stem cell transplantation
12.4.6.2.8.2. Allogeneic
stem cell transplantation
12.4.6.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
12.4.6.2.8.4. Graft-Versus-Host
Disease
12.4.6.2.9. Precision
Medicine
12.4.6.2.10. Others
12.4.7. Southeast
Asia
12.4.7.1. Southeast
Asia Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
12.4.7.1.1. Breast
Cancer
12.4.7.1.2. Prostate
Cancer
12.4.7.1.3. Basal
Cell Cancer
12.4.7.1.4. Skin
Cancer (Non-Melanoma)
12.4.7.1.5. Colorectal
cancer
12.4.7.1.6. Lung
Cancer
12.4.7.1.7. Renal
Cancer
12.4.7.1.8. Bladder
Cancer
12.4.7.1.9. Lymphoma
12.4.7.1.10. Others
12.4.7.2. Southeast
Asia Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
12.4.7.2.1. Drug
Therapies
12.4.7.2.1.1. Antimetabolites
12.4.7.2.1.2. Antitumor
Antibiotics
12.4.7.2.1.3. Asparagine-Specific
Enzymes
12.4.7.2.1.4. Bisphosphonates
and Biosimilars
12.4.7.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
12.4.7.2.1.6. Inhibitors
12.4.7.2.1.6.1. DNA-Repair Enzyme Inhibitors
12.4.7.2.1.6.2. Histone Deacetylase Inhibitors
12.4.7.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
12.4.7.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
12.4.7.2.1.6.5. Proteasome Inhibitors
12.4.7.2.1.6.6. Tyrosine Kinase Inhibitors
12.4.7.2.1.6.7. PD-1 and PD-L1 inhibitors
12.4.7.2.1.6.8. Others
12.4.7.2.1.7. Monoclonal
Antibodies
12.4.7.2.1.8. Others
12.4.7.2.2. Surgery
12.4.7.2.2.1. Open
Surgery
12.4.7.2.2.2. Minimally
Invasive Surgery
12.4.7.2.3. Radiation
Therapy
12.4.7.2.4. Chemotherapy
12.4.7.2.5. Immunotherapy
12.4.7.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
12.4.7.2.5.2. Cytokine
Treatment
12.4.7.2.5.3. Donor
lymphocyte infusion
12.4.7.2.5.4. Monoclonal
Antibody Treatment
12.4.7.2.5.5. Others
12.4.7.2.6. Targeted
Therapy
12.4.7.2.7. Hormone
Therapy
12.4.7.2.8. Stem
Cell Therapy
12.4.7.2.8.1. Autologous
stem cell transplantation
12.4.7.2.8.2. Allogeneic
stem cell transplantation
12.4.7.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
12.4.7.2.8.4. Graft-Versus-Host
Disease
12.4.7.2.9. Precision
Medicine
12.4.7.2.10. Others
12.4.7.3. Southeast
Asia Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Country
12.4.7.3.1. Indonesia
12.4.7.3.2. Thailand
12.4.7.3.3. Malaysia
12.4.7.3.4. Singapore
12.4.7.3.5. Rest
of Southeast Asia
12.4.8. Rest of
Asia Pacific
12.4.8.1. Rest
of Asia Pacific Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By
Indication
12.4.8.1.1. Breast
Cancer
12.4.8.1.2. Prostate
Cancer
12.4.8.1.3. Basal
Cell Cancer
12.4.8.1.4. Skin
Cancer (Non-Melanoma)
12.4.8.1.5. Colorectal
cancer
12.4.8.1.6. Lung
Cancer
12.4.8.1.7. Renal
Cancer
12.4.8.1.8. Bladder
Cancer
12.4.8.1.9. Lymphoma
12.4.8.1.10. Others
12.4.8.2. Rest
of Asia Pacific Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By
Treatment
12.4.8.2.1. Drug
Therapies
12.4.8.2.1.1. Antimetabolites
12.4.8.2.1.2. Antitumor
Antibiotics
12.4.8.2.1.3. Asparagine-Specific
Enzymes
12.4.8.2.1.4. Bisphosphonates
and Biosimilars
12.4.8.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
12.4.8.2.1.6. Inhibitors
12.4.8.2.1.6.1. DNA-Repair Enzyme Inhibitors
12.4.8.2.1.6.2. Histone Deacetylase Inhibitors
12.4.8.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
12.4.8.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
12.4.8.2.1.6.5. Proteasome Inhibitors
12.4.8.2.1.6.6. Tyrosine Kinase Inhibitors
12.4.8.2.1.6.7. PD-1 and PD-L1 inhibitors
12.4.8.2.1.6.8. Others
12.4.8.2.1.7. Monoclonal
Antibodies
12.4.8.2.1.8. Others
12.4.8.2.2. Surgery
12.4.8.2.2.1. Open
Surgery
12.4.8.2.2.2. Minimally
Invasive Surgery
12.4.8.2.3. Radiation
Therapy
12.4.8.2.4. Chemotherapy
12.4.8.2.5. Immunotherapy
12.4.8.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
12.4.8.2.5.2. Cytokine
Treatment
12.4.8.2.5.3. Donor
lymphocyte infusion
12.4.8.2.5.4. Monoclonal
Antibody Treatment
12.4.8.2.5.5. Others
12.4.8.2.6. Targeted
Therapy
12.4.8.2.7. Hormone
Therapy
12.4.8.2.8. Stem
Cell Therapy
12.4.8.2.8.1. Autologous
stem cell transplantation
12.4.8.2.8.2. Allogeneic
stem cell transplantation
12.4.8.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
12.4.8.2.8.4. Graft-Versus-Host
Disease
12.4.8.2.9. Precision
Medicine
12.4.8.2.10. Others
12.5. Key
Segment for Channeling Investments
12.5.1. By
Country
12.5.2. By
Indication
12.5.3. By
Treatment
13. Middle East and Africa Oncology Drugs Market Analysis and
Forecasts, 2019 - 2027
13.1. Overview
13.1.1. Middle
East and Africa Oncology Drugs Market Revenue (US$ Mn)
13.2. Middle
East and Africa Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By
Indication
13.2.1. Breast
Cancer
13.2.2. Prostate
Cancer
13.2.3. Basal
Cell Cancer
13.2.4. Skin
Cancer (Non-Melanoma)
13.2.5. Colorectal
cancer
13.2.6. Lung
Cancer
13.2.7. Renal
Cancer
13.2.8. Bladder
Cancer
13.2.9. Lymphoma
13.2.10. Others
13.3. Middle
East and Africa Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By
Treatment
13.3.1. Drug
Therapies
13.3.1.1. Antimetabolites
13.3.1.2. Antitumor
Antibiotics
13.3.1.3. Asparagine-Specific
Enzymes
13.3.1.4. Bisphosphonates
and Biosimilars
13.3.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
13.3.1.6. Inhibitors
13.3.1.6.1. DNA-Repair
Enzyme Inhibitors
13.3.1.6.2. Histone
Deacetylase Inhibitors
13.3.1.6.3. Janus-Associated
Kinase (JAK) Inhibitors
13.3.1.6.4. Phosphoinositide
3-kinase inhibitors (PI3K inhibitors)
13.3.1.6.5. Proteasome
Inhibitors
13.3.1.6.6. Tyrosine
Kinase Inhibitors
13.3.1.6.7. PD-1
and PD-L1 inhibitors
13.3.1.6.8. Others
13.3.1.7. Monoclonal
Antibodies
13.3.1.8. Others
13.3.2. Surgery
13.3.2.1. Open
Surgery
13.3.2.2. Minimally
Invasive Surgery
13.3.3. Radiation
Therapy
13.3.4. Chemotherapy
13.3.5. Immunotherapy
13.3.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
13.3.5.2. Cytokine
Treatment
13.3.5.3. Donor
lymphocyte infusion
13.3.5.4. Monoclonal
Antibody Treatment
13.3.5.5. Others
13.3.6. Targeted
Therapy
13.3.7. Hormone
Therapy
13.3.8. Stem
Cell Therapy
13.3.8.1. Autologous
stem cell transplantation
13.3.8.2. Allogeneic
stem cell transplantation
13.3.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
13.3.8.4. Graft-Versus-Host
Disease
13.3.9. Precision
Medicine
13.3.10. Others
13.4. Middle East
and Africa Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Country
13.4.1. Saudi
Arabia
13.4.1.1. Saudi
Arabia Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
13.4.1.1.1. Breast
Cancer
13.4.1.1.2. Prostate
Cancer
13.4.1.1.3. Basal
Cell Cancer
13.4.1.1.4. Skin
Cancer (Non-Melanoma)
13.4.1.1.5. Colorectal
cancer
13.4.1.1.6. Lung
Cancer
13.4.1.1.7. Renal
Cancer
13.4.1.1.8. Bladder
Cancer
13.4.1.1.9. Lymphoma
13.4.1.1.10. Others
13.4.1.2. Saudi
Arabia Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
13.4.1.2.1. Drug
Therapies
13.4.1.2.1.1. Antimetabolites
13.4.1.2.1.2. Antitumor
Antibiotics
13.4.1.2.1.3. Asparagine-Specific
Enzymes
13.4.1.2.1.4. Bisphosphonates
and Biosimilars
13.4.1.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
13.4.1.2.1.6. Inhibitors
13.4.1.2.1.6.1. DNA-Repair Enzyme Inhibitors
13.4.1.2.1.6.2. Histone Deacetylase Inhibitors
13.4.1.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
13.4.1.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
13.4.1.2.1.6.5. Proteasome Inhibitors
13.4.1.2.1.6.6. Tyrosine Kinase Inhibitors
13.4.1.2.1.6.7. PD-1 and PD-L1 inhibitors
13.4.1.2.1.6.8. Others
13.4.1.2.1.7. Monoclonal
Antibodies
13.4.1.2.1.8. Others
13.4.1.2.2. Surgery
13.4.1.2.2.1. Open
Surgery
13.4.1.2.2.2. Minimally
Invasive Surgery
13.4.1.2.3. Radiation
Therapy
13.4.1.2.4. Chemotherapy
13.4.1.2.5. Immunotherapy
13.4.1.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
13.4.1.2.5.2. Cytokine
Treatment
13.4.1.2.5.3. Donor
lymphocyte infusion
13.4.1.2.5.4. Monoclonal
Antibody Treatment
13.4.1.2.5.5. Others
13.4.1.2.6. Targeted
Therapy
13.4.1.2.7. Hormone
Therapy
13.4.1.2.8. Stem
Cell Therapy
13.4.1.2.8.1. Autologous
stem cell transplantation
13.4.1.2.8.2. Allogeneic
stem cell transplantation
13.4.1.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
13.4.1.2.8.4. Graft-Versus-Host
Disease
13.4.1.2.9. Precision
Medicine
13.4.1.2.10. Others
13.4.2. UAE
13.4.2.1. UAE
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
13.4.2.1.1. Breast
Cancer
13.4.2.1.2. Prostate
Cancer
13.4.2.1.3. Basal
Cell Cancer
13.4.2.1.4. Skin
Cancer (Non-Melanoma)
13.4.2.1.5. Colorectal
cancer
13.4.2.1.6. Lung
Cancer
13.4.2.1.7. Renal
Cancer
13.4.2.1.8. Bladder
Cancer
13.4.2.1.9. Lymphoma
13.4.2.1.10. Others
13.4.2.2. UAE
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
13.4.2.2.1. Drug
Therapies
13.4.2.2.1.1. Antimetabolites
13.4.2.2.1.2. Antitumor
Antibiotics
13.4.2.2.1.3. Asparagine-Specific
Enzymes
13.4.2.2.1.4. Bisphosphonates
and Biosimilars
13.4.2.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
13.4.2.2.1.6. Inhibitors
13.4.2.2.1.6.1. DNA-Repair Enzyme Inhibitors
13.4.2.2.1.6.2. Histone Deacetylase Inhibitors
13.4.2.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
13.4.2.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
13.4.2.2.1.6.5. Proteasome Inhibitors
13.4.2.2.1.6.6. Tyrosine Kinase Inhibitors
13.4.2.2.1.6.7. PD-1 and PD-L1 inhibitors
13.4.2.2.1.6.8. Others
13.4.2.2.1.7. Monoclonal
Antibodies
13.4.2.2.1.8. Others
13.4.2.2.2. Surgery
13.4.2.2.2.1. Open
Surgery
13.4.2.2.2.2. Minimally
Invasive Surgery
13.4.2.2.3. Radiation
Therapy
13.4.2.2.4. Chemotherapy
13.4.2.2.5. Immunotherapy
13.4.2.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
13.4.2.2.5.2. Cytokine
Treatment
13.4.2.2.5.3. Donor
lymphocyte infusion
13.4.2.2.5.4. Monoclonal
Antibody Treatment
13.4.2.2.5.5. Others
13.4.2.2.6. Targeted
Therapy
13.4.2.2.7. Hormone
Therapy
13.4.2.2.8. Stem
Cell Therapy
13.4.2.2.8.1. Autologous
stem cell transplantation
13.4.2.2.8.2. Allogeneic
stem cell transplantation
13.4.2.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
13.4.2.2.8.4. Graft-Versus-Host
Disease
13.4.2.2.9. Precision
Medicine
13.4.2.2.10. Others
13.4.3. Egypt
13.4.3.1. Egypt
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
13.4.3.1.1. Breast
Cancer
13.4.3.1.2. Prostate
Cancer
13.4.3.1.3. Basal
Cell Cancer
13.4.3.1.4. Skin
Cancer (Non-Melanoma)
13.4.3.1.5. Colorectal
cancer
13.4.3.1.6. Lung
Cancer
13.4.3.1.7. Renal
Cancer
13.4.3.1.8. Bladder
Cancer
13.4.3.1.9. Lymphoma
13.4.3.1.10. Others
13.4.3.2. Egypt
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
13.4.3.2.1. Drug
Therapies
13.4.3.2.1.1. Antimetabolites
13.4.3.2.1.2. Antitumor
Antibiotics
13.4.3.2.1.3. Asparagine-Specific
Enzymes
13.4.3.2.1.4. Bisphosphonates
and Biosimilars
13.4.3.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
13.4.3.2.1.6. Inhibitors
13.4.3.2.1.6.1. DNA-Repair Enzyme Inhibitors
13.4.3.2.1.6.2. Histone Deacetylase Inhibitors
13.4.3.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
13.4.3.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
13.4.3.2.1.6.5. Proteasome Inhibitors
13.4.3.2.1.6.6. Tyrosine Kinase Inhibitors
13.4.3.2.1.6.7. PD-1 and PD-L1 inhibitors
13.4.3.2.1.6.8. Others
13.4.3.2.1.7. Monoclonal
Antibodies
13.4.3.2.1.8. Others
13.4.3.2.2. Surgery
13.4.3.2.2.1. Open
Surgery
13.4.3.2.2.2. Minimally
Invasive Surgery
13.4.3.2.3. Radiation
Therapy
13.4.3.2.4. Chemotherapy
13.4.3.2.5. Immunotherapy
13.4.3.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
13.4.3.2.5.2. Cytokine
Treatment
13.4.3.2.5.3. Donor
lymphocyte infusion
13.4.3.2.5.4. Monoclonal
Antibody Treatment
13.4.3.2.5.5. Others
13.4.3.2.6. Targeted
Therapy
13.4.3.2.7. Hormone
Therapy
13.4.3.2.8. Stem
Cell Therapy
13.4.3.2.8.1. Autologous
stem cell transplantation
13.4.3.2.8.2. Allogeneic
stem cell transplantation
13.4.3.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
13.4.3.2.8.4. Graft-Versus-Host
Disease
13.4.3.2.9. Precision
Medicine
13.4.3.2.10. Others
13.4.4. Kuwait
13.4.4.1. Kuwait
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
13.4.4.1.1. Breast
Cancer
13.4.4.1.2. Prostate
Cancer
13.4.4.1.3. Basal
Cell Cancer
13.4.4.1.4. Skin
Cancer (Non-Melanoma)
13.4.4.1.5. Colorectal
cancer
13.4.4.1.6. Lung
Cancer
13.4.4.1.7. Renal
Cancer
13.4.4.1.8. Bladder
Cancer
13.4.4.1.9. Lymphoma
13.4.4.1.10. Others
13.4.4.2. Kuwait
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
13.4.4.2.1. Drug
Therapies
13.4.4.2.1.1. Antimetabolites
13.4.4.2.1.2. Antitumor
Antibiotics
13.4.4.2.1.3. Asparagine-Specific
Enzymes
13.4.4.2.1.4. Bisphosphonates
and Biosimilars
13.4.4.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
13.4.4.2.1.6. Inhibitors
13.4.4.2.1.6.1. DNA-Repair Enzyme Inhibitors
13.4.4.2.1.6.2. Histone Deacetylase Inhibitors
13.4.4.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
13.4.4.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
13.4.4.2.1.6.5. Proteasome Inhibitors
13.4.4.2.1.6.6. Tyrosine Kinase Inhibitors
13.4.4.2.1.6.7. PD-1 and PD-L1 inhibitors
13.4.4.2.1.6.8. Others
13.4.4.2.1.7. Monoclonal
Antibodies
13.4.4.2.1.8. Others
13.4.4.2.2. Surgery
13.4.4.2.2.1. Open
Surgery
13.4.4.2.2.2. Minimally
Invasive Surgery
13.4.4.2.3. Radiation
Therapy
13.4.4.2.4. Chemotherapy
13.4.4.2.5. Immunotherapy
13.4.4.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
13.4.4.2.5.2. Cytokine
Treatment
13.4.4.2.5.3. Donor
lymphocyte infusion
13.4.4.2.5.4. Monoclonal
Antibody Treatment
13.4.4.2.5.5. Others
13.4.4.2.6. Targeted
Therapy
13.4.4.2.7. Hormone
Therapy
13.4.4.2.8. Stem
Cell Therapy
13.4.4.2.8.1. Autologous
stem cell transplantation
13.4.4.2.8.2. Allogeneic
stem cell transplantation
13.4.4.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
13.4.4.2.8.4. Graft-Versus-Host
Disease
13.4.4.2.9. Precision
Medicine
13.4.4.2.10. Others
13.4.5. South
Africa
13.4.5.1. South
Africa Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
13.4.5.1.1. Breast
Cancer
13.4.5.1.2. Prostate
Cancer
13.4.5.1.3. Basal
Cell Cancer
13.4.5.1.4. Skin
Cancer (Non-Melanoma)
13.4.5.1.5. Colorectal
cancer
13.4.5.1.6. Lung
Cancer
13.4.5.1.7. Renal
Cancer
13.4.5.1.8. Bladder
Cancer
13.4.5.1.9. Lymphoma
13.4.5.1.10. Others
13.4.5.2. South
Africa Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
13.4.5.2.1. Drug
Therapies
13.4.5.2.1.1. Antimetabolites
13.4.5.2.1.2. Antitumor
Antibiotics
13.4.5.2.1.3. Asparagine-Specific
Enzymes
13.4.5.2.1.4. Bisphosphonates
and Biosimilars
13.4.5.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
13.4.5.2.1.6. Inhibitors
13.4.5.2.1.6.1. DNA-Repair Enzyme Inhibitors
13.4.5.2.1.6.2. Histone Deacetylase Inhibitors
13.4.5.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
13.4.5.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
13.4.5.2.1.6.5. Proteasome Inhibitors
13.4.5.2.1.6.6. Tyrosine Kinase Inhibitors
13.4.5.2.1.6.7. PD-1 and PD-L1 inhibitors
13.4.5.2.1.6.8. Others
13.4.5.2.1.7. Monoclonal
Antibodies
13.4.5.2.1.8. Others
13.4.5.2.2. Surgery
13.4.5.2.2.1. Open
Surgery
13.4.5.2.2.2. Minimally
Invasive Surgery
13.4.5.2.3. Radiation
Therapy
13.4.5.2.4. Chemotherapy
13.4.5.2.5. Immunotherapy
13.4.5.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
13.4.5.2.5.2. Cytokine
Treatment
13.4.5.2.5.3. Donor
lymphocyte infusion
13.4.5.2.5.4. Monoclonal
Antibody Treatment
13.4.5.2.5.5. Others
13.4.5.2.6. Targeted
Therapy
13.4.5.2.7. Hormone
Therapy
13.4.5.2.8. Stem
Cell Therapy
13.4.5.2.8.1. Autologous
stem cell transplantation
13.4.5.2.8.2. Allogeneic
stem cell transplantation
13.4.5.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
13.4.5.2.8.4. Graft-Versus-Host
Disease
13.4.5.2.9. Precision
Medicine
13.4.5.2.10. Others
13.4.6. Rest of
Middle East & Africa
13.4.6.1. Rest
of Middle East & Africa Oncology Drugs Market Revenue (US$ Mn) and
Forecasts, By Indication
13.4.6.1.1. Breast
Cancer
13.4.6.1.2. Prostate
Cancer
13.4.6.1.3. Basal
Cell Cancer
13.4.6.1.4. Skin
Cancer (Non-Melanoma)
13.4.6.1.5. Colorectal
cancer
13.4.6.1.6. Lung
Cancer
13.4.6.1.7. Renal
Cancer
13.4.6.1.8. Bladder
Cancer
13.4.6.1.9. Lymphoma
13.4.6.1.10. Others
13.4.6.2. Rest
of Middle East & Africa Oncology Drugs Market Revenue (US$ Mn) and
Forecasts, By Treatment
13.4.6.2.1. Drug
Therapies
13.4.6.2.1.1. Antimetabolites
13.4.6.2.1.2. Antitumor
Antibiotics
13.4.6.2.1.3. Asparagine-Specific
Enzymes
13.4.6.2.1.4. Bisphosphonates
and Biosimilars
13.4.6.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
13.4.6.2.1.6. Inhibitors
13.4.6.2.1.6.1. DNA-Repair Enzyme Inhibitors
13.4.6.2.1.6.2. Histone Deacetylase Inhibitors
13.4.6.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
13.4.6.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
13.4.6.2.1.6.5. Proteasome Inhibitors
13.4.6.2.1.6.6. Tyrosine Kinase Inhibitors
13.4.6.2.1.6.7. PD-1 and PD-L1 inhibitors
13.4.6.2.1.6.8. Others
13.4.6.2.1.7. Monoclonal
Antibodies
13.4.6.2.1.8. Others
13.4.6.2.2. Surgery
13.4.6.2.2.1. Open
Surgery
13.4.6.2.2.2. Minimally
Invasive Surgery
13.4.6.2.3. Radiation
Therapy
13.4.6.2.4. Chemotherapy
13.4.6.2.5. Immunotherapy
13.4.6.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
13.4.6.2.5.2. Cytokine
Treatment
13.4.6.2.5.3. Donor
lymphocyte infusion
13.4.6.2.5.4. Monoclonal
Antibody Treatment
13.4.6.2.5.5. Others
13.4.6.2.6. Targeted
Therapy
13.4.6.2.7. Hormone
Therapy
13.4.6.2.8. Stem
Cell Therapy
13.4.6.2.8.1. Autologous
stem cell transplantation
13.4.6.2.8.2. Allogeneic
stem cell transplantation
13.4.6.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
13.4.6.2.8.4. Graft-Versus-Host
Disease
13.4.6.2.9. Precision
Medicine
13.4.6.2.10. Others
13.5. Key
Segment for Channeling Investments
13.5.1. By
Country
13.5.2. By
Indication
13.5.3. By
Treatment
14. Latin America Oncology Drugs Market Analysis and Forecasts,
2019 - 2027
14.1. Overview
14.1.1. Latin
America Oncology Drugs Market Revenue (US$ Mn)
14.2. Latin
America Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
14.2.1. Breast
Cancer
14.2.2. Prostate
Cancer
14.2.3. Basal
Cell Cancer
14.2.4. Skin
Cancer (Non-Melanoma)
14.2.5. Colorectal
cancer
14.2.6. Lung
Cancer
14.2.7. Renal
Cancer
14.2.8. Bladder
Cancer
14.2.9. Lymphoma
14.2.10. Others
14.3. Latin
America Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
14.3.1. Drug
Therapies
14.3.1.1. Antimetabolites
14.3.1.2. Antitumor
Antibiotics
14.3.1.3. Asparagine-Specific
Enzymes
14.3.1.4. Bisphosphonates
and Biosimilars
14.3.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
14.3.1.6. Inhibitors
14.3.1.6.1. DNA-Repair
Enzyme Inhibitors
14.3.1.6.2. Histone
Deacetylase Inhibitors
14.3.1.6.3. Janus-Associated
Kinase (JAK) Inhibitors
14.3.1.6.4. Phosphoinositide
3-kinase inhibitors (PI3K inhibitors)
14.3.1.6.5. Proteasome
Inhibitors
14.3.1.6.6. Tyrosine
Kinase Inhibitors
14.3.1.6.7. PD-1
and PD-L1 inhibitors
14.3.1.6.8. Others
14.3.1.7. Monoclonal
Antibodies
14.3.1.8. Others
14.3.2. Surgery
14.3.2.1. Open
Surgery
14.3.2.2. Minimally
Invasive Surgery
14.3.3. Radiation
Therapy
14.3.4. Chemotherapy
14.3.5. Immunotherapy
14.3.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
14.3.5.2. Cytokine
Treatment
14.3.5.3. Donor
lymphocyte infusion
14.3.5.4. Monoclonal
Antibody Treatment
14.3.5.5. Others
14.3.6. Targeted
Therapy
14.3.7. Hormone
Therapy
14.3.8. Stem
Cell Therapy
14.3.8.1. Autologous
stem cell transplantation
14.3.8.2. Allogeneic
stem cell transplantation
14.3.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
14.3.8.4. Graft-Versus-Host
Disease
14.3.9. Precision
Medicine
14.3.10. Others
14.4. Latin
America Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Country
14.4.1. Brazil
14.4.1.1. Brazil
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
14.4.1.1.1. Breast
Cancer
14.4.1.1.2. Prostate
Cancer
14.4.1.1.3. Basal
Cell Cancer
14.4.1.1.4. Skin
Cancer (Non-Melanoma)
14.4.1.1.5. Colorectal
cancer
14.4.1.1.6. Lung
Cancer
14.4.1.1.7. Renal
Cancer
14.4.1.1.8. Bladder
Cancer
14.4.1.1.9. Lymphoma
14.4.1.1.10. Others
14.4.1.2. Brazil
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
14.4.1.2.1. Drug
Therapies
14.4.1.2.1.1. Antimetabolites
14.4.1.2.1.2. Antitumor
Antibiotics
14.4.1.2.1.3. Asparagine-Specific
Enzymes
14.4.1.2.1.4. Bisphosphonates
and Biosimilars
14.4.1.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
14.4.1.2.1.6. Inhibitors
14.4.1.2.1.6.1. DNA-Repair Enzyme Inhibitors
14.4.1.2.1.6.2. Histone Deacetylase Inhibitors
14.4.1.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
14.4.1.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
14.4.1.2.1.6.5. Proteasome Inhibitors
14.4.1.2.1.6.6. Tyrosine Kinase Inhibitors
14.4.1.2.1.6.7. PD-1 and PD-L1 inhibitors
14.4.1.2.1.6.8. Others
14.4.1.2.1.7. Monoclonal
Antibodies
14.4.1.2.1.8. Others
14.4.1.2.2. Surgery
14.4.1.2.2.1. Open
Surgery
14.4.1.2.2.2. Minimally
Invasive Surgery
14.4.1.2.3. Radiation
Therapy
14.4.1.2.4. Chemotherapy
14.4.1.2.5. Immunotherapy
14.4.1.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
14.4.1.2.5.2. Cytokine
Treatment
14.4.1.2.5.3. Donor
lymphocyte infusion
14.4.1.2.5.4. Monoclonal
Antibody Treatment
14.4.1.2.5.5. Others
14.4.1.2.6. Targeted
Therapy
14.4.1.2.7. Hormone
Therapy
14.4.1.2.8. Stem
Cell Therapy
14.4.1.2.8.1. Autologous
stem cell transplantation
14.4.1.2.8.2. Allogeneic
stem cell transplantation
14.4.1.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
14.4.1.2.8.4. Graft-Versus-Host
Disease
14.4.1.2.9. Precision
Medicine
14.4.1.2.10. Others
14.4.2. Argentina
14.4.2.1. Argentina
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Indication
14.4.2.1.1. Breast
Cancer
14.4.2.1.2. Prostate
Cancer
14.4.2.1.3. Basal
Cell Cancer
14.4.2.1.4. Skin
Cancer (Non-Melanoma)
14.4.2.1.5. Colorectal
cancer
14.4.2.1.6. Lung
Cancer
14.4.2.1.7. Renal
Cancer
14.4.2.1.8. Bladder
Cancer
14.4.2.1.9. Lymphoma
14.4.2.1.10. Others
14.4.2.2. Argentina
Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By Treatment
14.4.2.2.1. Drug
Therapies
14.4.2.2.1.1. Antimetabolites
14.4.2.2.1.2. Antitumor
Antibiotics
14.4.2.2.1.3. Asparagine-Specific
Enzymes
14.4.2.2.1.4. Bisphosphonates
and Biosimilars
14.4.2.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
14.4.2.2.1.6. Inhibitors
14.4.2.2.1.6.1. DNA-Repair Enzyme Inhibitors
14.4.2.2.1.6.2. Histone Deacetylase Inhibitors
14.4.2.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
14.4.2.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
14.4.2.2.1.6.5. Proteasome Inhibitors
14.4.2.2.1.6.6. Tyrosine Kinase Inhibitors
14.4.2.2.1.6.7. PD-1 and PD-L1 inhibitors
14.4.2.2.1.6.8. Others
14.4.2.2.1.7. Monoclonal
Antibodies
14.4.2.2.1.8. Others
14.4.2.2.2. Surgery
14.4.2.2.2.1. Open
Surgery
14.4.2.2.2.2. Minimally
Invasive Surgery
14.4.2.2.3. Radiation
Therapy
14.4.2.2.4. Chemotherapy
14.4.2.2.5. Immunotherapy
14.4.2.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
14.4.2.2.5.2. Cytokine
Treatment
14.4.2.2.5.3. Donor
lymphocyte infusion
14.4.2.2.5.4. Monoclonal
Antibody Treatment
14.4.2.2.5.5. Others
14.4.2.2.6. Targeted
Therapy
14.4.2.2.7. Hormone
Therapy
14.4.2.2.8. Stem
Cell Therapy
14.4.2.2.8.1. Autologous
stem cell transplantation
14.4.2.2.8.2. Allogeneic
stem cell transplantation
14.4.2.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
14.4.2.2.8.4. Graft-Versus-Host
Disease
14.4.2.2.9. Precision
Medicine
14.4.2.2.10. Others
14.4.3. Rest of
Latin America
14.4.3.1. Rest
of Latin America Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By
Indication
14.4.3.1.1. Breast
Cancer
14.4.3.1.2. Prostate
Cancer
14.4.3.1.3. Basal
Cell Cancer
14.4.3.1.4. Skin
Cancer (Non-Melanoma)
14.4.3.1.5. Colorectal
cancer
14.4.3.1.6. Lung
Cancer
14.4.3.1.7. Renal
Cancer
14.4.3.1.8. Bladder
Cancer
14.4.3.1.9. Lymphoma
14.4.3.1.10. Others
14.4.3.2. Rest
of Latin America Oncology Drugs Market Revenue (US$ Mn) and Forecasts, By
Treatment
14.4.3.2.1. Drug
Therapies
14.4.3.2.1.1. Antimetabolites
14.4.3.2.1.2. Antitumor
Antibiotics
14.4.3.2.1.3. Asparagine-Specific
Enzymes
14.4.3.2.1.4. Bisphosphonates
and Biosimilars
14.4.3.2.1.5. DNA-Damaging
Agents (Antineoplastics) and Alkylating Agents
14.4.3.2.1.6. Inhibitors
14.4.3.2.1.6.1. DNA-Repair Enzyme Inhibitors
14.4.3.2.1.6.2. Histone Deacetylase Inhibitors
14.4.3.2.1.6.3. Janus-Associated Kinase (JAK) Inhibitors
14.4.3.2.1.6.4. Phosphoinositide 3-kinase inhibitors (PI3K inhibitors)
14.4.3.2.1.6.5. Proteasome Inhibitors
14.4.3.2.1.6.6. Tyrosine Kinase Inhibitors
14.4.3.2.1.6.7. PD-1 and PD-L1 inhibitors
14.4.3.2.1.6.8. Others
14.4.3.2.1.7. Monoclonal
Antibodies
14.4.3.2.1.8. Others
14.4.3.2.2. Surgery
14.4.3.2.2.1. Open
Surgery
14.4.3.2.2.2. Minimally
Invasive Surgery
14.4.3.2.3. Radiation
Therapy
14.4.3.2.4. Chemotherapy
14.4.3.2.5. Immunotherapy
14.4.3.2.5.1. Chimeric
antigen receptor (CAR) T-cell therapy
14.4.3.2.5.2. Cytokine
Treatment
14.4.3.2.5.3. Donor
lymphocyte infusion
14.4.3.2.5.4. Monoclonal
Antibody Treatment
14.4.3.2.5.5. Others
14.4.3.2.6. Targeted
Therapy
14.4.3.2.7. Hormone
Therapy
14.4.3.2.8. Stem
Cell Therapy
14.4.3.2.8.1. Autologous
stem cell transplantation
14.4.3.2.8.2. Allogeneic
stem cell transplantation
14.4.3.2.8.3. Reduced-Intensity
Allogeneic Stem Cell Transplantation
14.4.3.2.8.4. Graft-Versus-Host
Disease
14.4.3.2.9. Precision
Medicine
14.4.3.2.10. Others
14.5. Key
Segment for Channeling Investments
14.5.1. By
Country
14.5.2. By
Indication
14.5.3. By
Treatment
15. Competitive Benchmarking
15.1. Market
Share Analysis, 2018
15.2. Global
Presence and Growth Strategies
15.2.1. Mergers
and Acquisitions
15.2.2. Product
Launches
15.2.3. Investments
Trends
15.2.4. R&D
Initiatives
16. Player Profiles
16.1. AstraZeneca
16.1.1. Company
Details
16.1.2. Company
Overview
16.1.3. Product
Offerings
16.1.4. Key Developments
16.1.5. Financial
Analysis
16.1.6. SWOT
Analysis
16.1.7. Business Strategies
16.2. Bayer Ag
16.2.1. Company
Details
16.2.2. Company
Overview
16.2.3. Product
Offerings
16.2.4. Key
Developments
16.2.5. Financial
Analysis
16.2.6. SWOT
Analysis
16.2.7. Business Strategies
16.3. CELEGENE CORPORATION
16.3.1. Company
Details
16.3.2. Company
Overview
16.3.3. Product
Offerings
16.3.4. Key
Developments
16.3.5. Financial
Analysis
16.3.6. SWOT
Analysis
16.3.7. Business Strategies
16.4. Eli Lily and Company
16.4.1. Company
Details
16.4.2. Company
Overview
16.4.3. Product
Offerings
16.4.4. Key
Developments
16.4.5. Financial
Analysis
16.4.6. SWOT
Analysis
16.4.7. Business Strategies
16.5. F. Hoffmann-La Roche Ltd
16.5.1. Company
Details
16.5.2. Company
Overview
16.5.3. Product
Offerings
16.5.4. Key
Developments
16.5.5. Financial
Analysis
16.5.6. SWOT
Analysis
16.5.7. Business Strategies
16.6. Johnson & Johnson Services, Inc.
16.6.1. Company
Details
16.6.2. Company
Overview
16.6.3. Product
Offerings
16.6.4. Key
Developments
16.6.5. Financial
Analysis
16.6.6. SWOT
Analysis
16.6.7. Business Strategies
16.7. Merck & Co., Inc.
16.7.1. Company
Details
16.7.2. Company
Overview
16.7.3. Product
Offerings
16.7.4. Key
Developments
16.7.5. Financial
Analysis
16.7.6. SWOT
Analysis
16.7.7. Business Strategies
16.8. Novartis AG
16.8.1. Company
Details
16.8.2. Company
Overview
16.8.3. Product
Offerings
16.8.4. Key
Developments
16.8.5. Financial
Analysis
16.8.6. SWOT Analysis
16.8.7. Business Strategies
16.9. Pfizer Inc.
16.9.1. Company
Details
16.9.2. Company
Overview
16.9.3. Product
Offerings
16.9.4. Key
Developments
16.9.5. Financial
Analysis
16.9.6. SWOT
Analysis
16.9.7. Business Strategies
16.10. Polaris Pharmaceuticals
16.10.1. Company
Details
16.10.2. Company
Overview
16.10.3. Product
Offerings
16.10.4. Key
Developments
16.10.5. Financial
Analysis
16.10.6. SWOT
Analysis
16.10.7. Business Strategies
16.11. Other
Market Participants
17. Key Findings
Note: This ToC is tentative
and can be changed according to the research study conducted during the course
of report completion.
**Exclusive for Multi-User
and Enterprise User.
At Absolute Markets Insights, we are engaged in building both global as well as country specific reports. As a result, the approach taken for deriving the estimation and forecast for a specific country is a bit unique and different in comparison to the global research studies. In this case, we not only study the concerned market factors & trends prevailing in a particular country (from secondary research) but we also tend to calculate the actual market size & forecast from the revenue generated from the market participants involved in manufacturing or distributing the any concerned product. These companies can also be service providers. For analyzing any country specifically, we do consider the growth factors prevailing under the states/cities/county for the same. For instance, if we are analyzing an industry specific to United States, we primarily need to study about the states present under the same(where the product/service has the highest growth). Similar analysis will be followed by other countries. Our scope of the report changes with different markets.
Our research study is mainly implement through a mix of both secondary and primary research. Various sources such as industry magazines, trade journals, and government websites and trade associations are reviewed for gathering precise data. Primary interviews are conducted to validate the market size derived from secondary research. Industry experts, major manufacturers and distributors are contacted for further validation purpose on the current market penetration and growth trends.
Prominent participants in our primary research process include:
- Key Opinion Leaders namely the CEOs, CSOs, VPs, purchasing managers, amongst others
- Research and development participants, distributors/suppliers and subject matter experts
Secondary Research includes data extracted from paid data sources:
- Reuters
- Factiva
- Bloomberg
- One Source
- Hoovers
Research Methodology
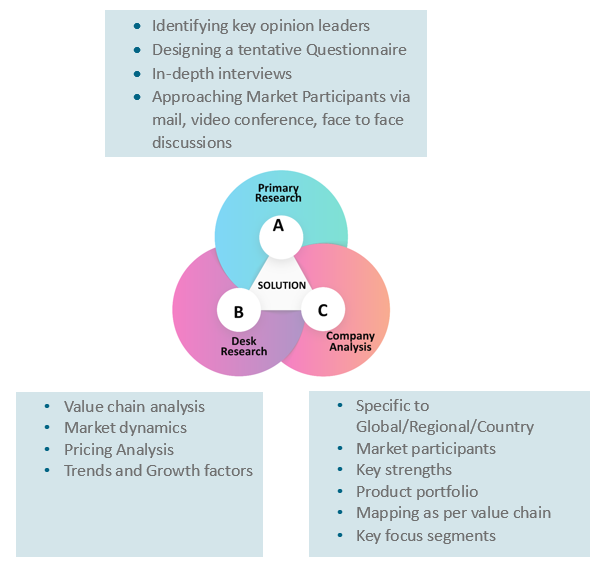
Key Inclusions

Reach to us
Call us on
+91-74002-42424
Drop us an email at
sales@absolutemarketsinsights.com
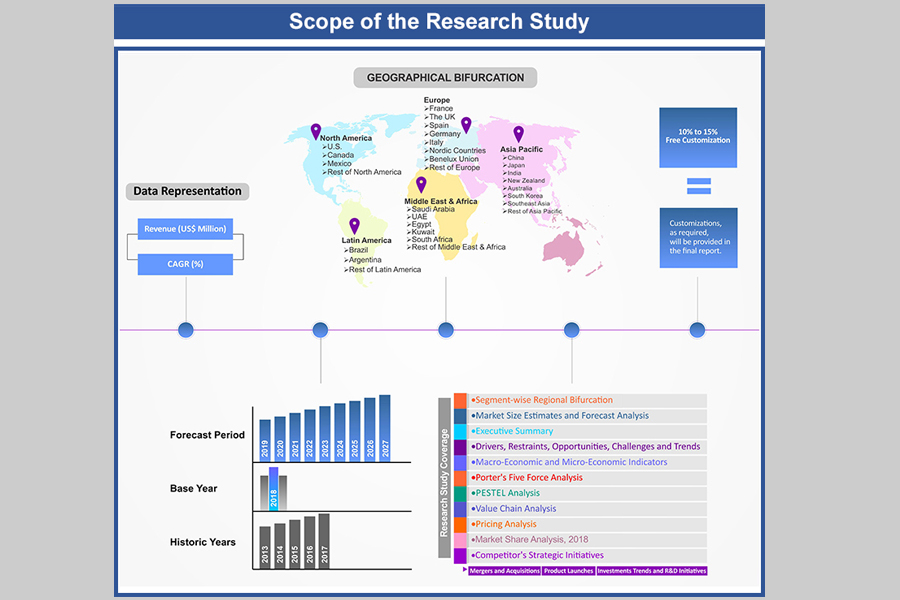
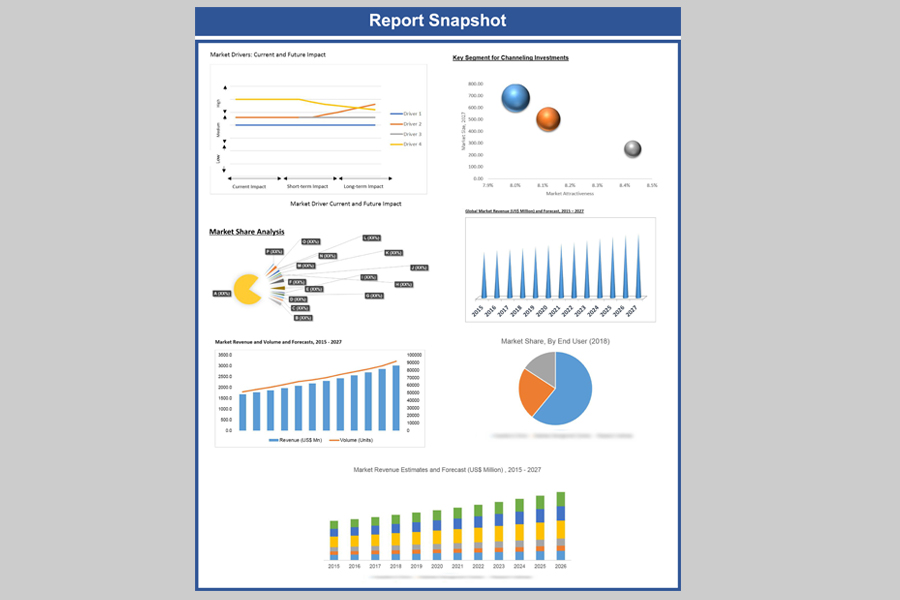
Why Absolute Markets Insights?
An effective strategy is the entity that influences a business to stand out of the crowd. An organization with a phenomenal strategy for success dependably has the edge over the rivals in the market. It offers the organizations a head start in planning their strategy. Absolute Market Insights is the new initiation in the industry that will furnish you with the lead your business needs. Absolute Market Insights is the best destination for your business intelligence and analytical solutions; essentially because our qualitative and quantitative sources of information are competent to give one-stop solutions. We inventively combine qualitative and quantitative research in accurate proportions to have the best report, which not only gives the most recent insights but also assists you to grow.